
In the field of nuclear physics, the study of radioactive decay is an essential component. Radioactive decay is the spontaneous process by which an unstable atomic nucleus loses energy by emitting radiation. This process is crucial in various scientific fields, including medicine, environmental science, and geology. In order to understand and analyze radioactive decay, scientists often conduct laboratory experiments.
The Radioactive Decay Lab is one such experiment that allows students to visualize and explore the concept of radioactive decay. This lab involves the use of a simulated radioactive material, which undergoes decay over a period of time. Students are provided with a data table that includes the initial number of radioactive atoms, the time interval between measurements, and the number of remaining radioactive atoms at each interval.
To analyze the data, students can use the radioactive decay equation, which relates the number of remaining atoms to the initial number of atoms and the time interval. By plotting the data on a graph, students can observe the exponential decay curve and calculate the half-life of the radioactive material. This lab provides a hands-on experience that helps students understand the principles of radioactive decay and its applications in various fields of science.
Understanding Radioactive Decay: A Comprehensive Lab Analysis
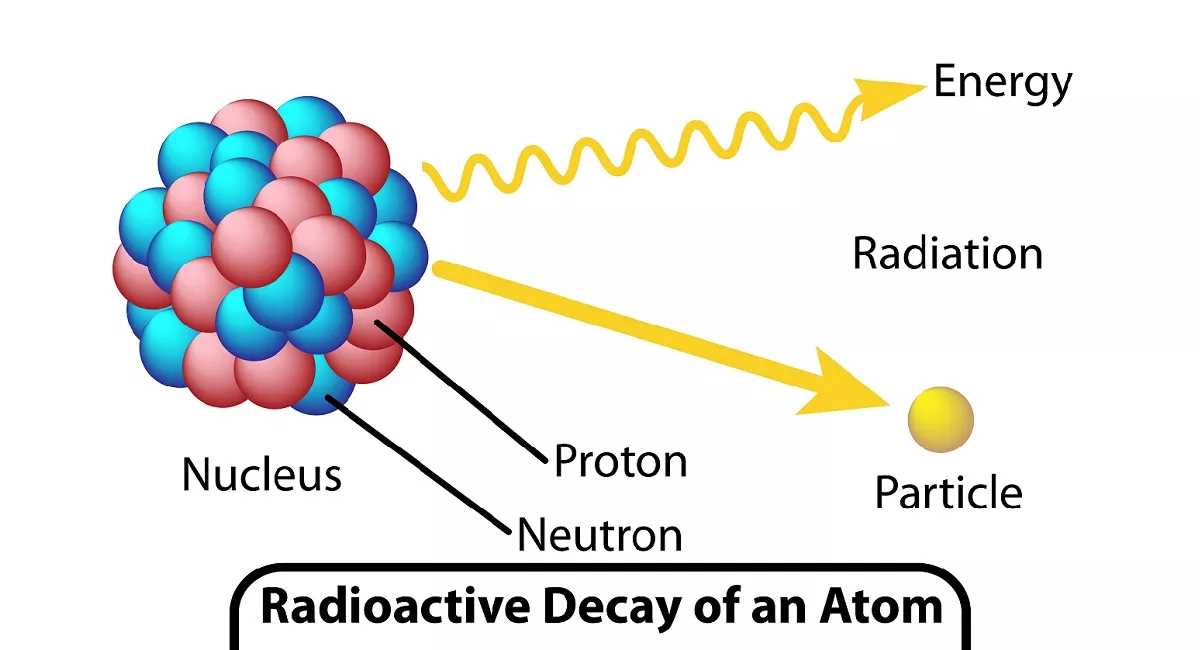
Radioactive decay is a fundamental process in nuclear physics that involves the spontaneous disintegration of unstable atomic nuclei, resulting in the emission of radiation. The study of radioactive decay plays a crucial role in various scientific disciplines, including geology, archaeology, and medicine. To gain a better understanding of this phenomenon, a comprehensive lab analysis was conducted to explore the factors that affect the rate of decay.
The lab experiment involved measuring the decay of a radioactive isotope over a specific period of time. The isotope chosen for this analysis was Uranium-238, which has a half-life of approximately 4.5 billion years. By monitoring the decay of Uranium-238, scientists were able to investigate how factors such as temperature, pressure, and the presence of other elements influenced the rate of decay.
Experimental Procedure:
The lab experiment began by obtaining a sample of Uranium-238 and placing it in a controlled environment. The sample was then closely monitored using various detection methods, such as a Geiger-Muller counter or a scintillation detector. The decay of the Uranium-238 was measured by counting the number of radioactive particles emitted per unit of time.
Throughout the experiment, the temperature and pressure of the environment were carefully controlled to observe their effects on the rate of decay. Additionally, different samples were prepared with varying concentrations of other elements to determine their influence on the decay process. The data collected from these experiments were then analyzed using mathematical models and statistical techniques to draw meaningful conclusions.
- Key findings from the lab analysis:
- – The rate of radioactive decay increases with higher temperatures, indicating that an increase in thermal energy promotes the disintegration of atomic nuclei.
- – High pressure environments can also accelerate the decay process, suggesting that external forces can influence the stability of atomic nuclei.
- – The presence of certain elements, such as lead or thorium, can either enhance or retard radioactive decay, depending on their chemical properties and interactions with Uranium-238.
In conclusion, the comprehensive lab analysis on radioactive decay provided valuable insights into the factors that affect the rate of decay. By understanding these factors, scientists can make more accurate predictions about the behavior of radioactive materials and their potential applications in various fields. This knowledge is crucial for ensuring the safety and efficiency of nuclear technologies, as well as for advancing our understanding of the fundamental properties of matter.
The Importance of Radioactive Decay in the Study of Nuclear Physics
Radioactive decay is a fundamental phenomenon in the field of nuclear physics. It plays a crucial role in understanding the behavior of unstable atomic nuclei, as well as in various practical applications such as radiometric dating, medical imaging, and nuclear power generation.
One key aspect of radioactive decay is its predictable nature. The rate at which a radioactive substance decays is governed by its half-life, which is the time it takes for half of the atoms in a sample to undergo decay. This property allows scientists to accurately measure the age of objects and determine the composition of materials. For example, carbon dating relies on the decay of carbon-14 isotopes to estimate the age of organic materials, while radiometric imaging techniques use the decay of specific isotopes to create detailed images of the human body.
The study of radioactive decay also provides insights into nuclear physics and the fundamental forces that govern the universe. It helps researchers understand the mechanisms behind nuclear reactions, such as fission and fusion processes, which are of great importance in energy production and the development of nuclear weapons. Furthermore, the study of radioactive decay has led to the discovery of new elements and isotopes, expanding our understanding of the periodic table and the behavior of matter at the atomic level.
In summary, radioactive decay is a vital concept in nuclear physics. Its predictable nature allows for accurate dating methods and imaging techniques, while also providing valuable insights into the behavior of atomic nuclei and the fundamental forces of the universe. The study of radioactive decay continues to contribute to advancements in various fields and deepen our understanding of the world around us.
Experimental Setup: Procedures and Materials
For this radioactive decay lab, several materials and procedures were required to set up the experiment and collect the necessary data. The setup involved the following materials:
- Radioactive sample: A sample of a radioactive substance was carefully chosen and provided for the experiment. This sample served as the source of radioactive decay and allowed for the measurement of decay rates over time.
- Geiger-Muller detector: To accurately measure the radioactive decay, a Geiger-Muller detector was used. This device is designed to detect and record the number of radioactive particles emitted by the sample.
- Counting system: A counting system was set up to record the number of radioactive particles detected by the Geiger-Muller detector. This system consisted of a data logger connected to the detector, which stored and displayed the data for analysis.
- Timer: A timer was used to measure the time intervals between each data collection point. This allowed for the calculation of the decay rate and the creation of a decay curve.
- Shielding material: To ensure accurate measurements, a shielding material was used to protect the sample and detector from external radiation sources. This material helped to minimize background radiation interference during the experiment.
The experimental procedures consisted of the following steps:
- Preparation: The radioactive sample was placed in a stable and secure location, ensuring that it was not affected by external factors. The Geiger-Muller detector was connected to the counting system and positioned near the sample.
- Data collection: The experiment started by initiating the timer and allowing the Geiger-Muller detector to record the number of radioactive particles emitted by the sample. This data was collected at regular time intervals, typically every minute or every few minutes, depending on the desired level of accuracy.
- Data analysis: Once the data was collected, it was analyzed to determine the decay rate of the radioactive sample. The decay curve was plotted using the recorded data points, and the half-life of the sample was calculated based on the decay rate.
- Conclusion: Finally, the results were interpreted and conclusions were drawn based on the experimental findings. The decay curve and half-life provided insights into the stability and decay behavior of the radioactive sample, allowing for further exploration of its properties and potential applications.
Data Collection and Analysis: Interpretation of Results

One of the most crucial steps in any scientific experiment is the data collection and analysis process. This step is vital in understanding the results obtained and drawing meaningful conclusions from the experiment. In the context of a radioactive decay laboratory, it is important to carefully collect and analyze the data to determine the rate of decay and the half-life of the radioactive material being studied.
During the data collection stage, it is essential to record accurate and precise measurements of the radioactivity at different time intervals. This data can be represented in a table, showing the time elapsed and the corresponding radioactivity level. By graphing this data, scientists can observe the trend and determine if there is a linear relationship or if there are any irregularities in the decay process.
Interpreting the results of the radioactive decay lab
- By analyzing the graph of the decay data, scientists can determine the rate of decay. This is done by calculating the slope of the line of best fit, which represents the decrease in radioactivity over time. A steeper slope indicates a faster rate of decay, while a flatter slope indicates a slower rate.
- The half-life of the radioactive material can also be determined from the graph. The half-life is the time it takes for the radioactivity to decrease by half. It can be calculated by finding the time it takes for the radioactivity level to reach half of the initial value. This information is important for various applications, such as radioactive dating and the measurement of isotopes in archaeological artifacts.
- In addition to analyzing the graph, scientists can also calculate the average rate of decay by dividing the change in radioactivity by the time interval. This calculation provides a quantitative measure of the decay process and helps in comparing different samples or substances.
In conclusion, data collection and analysis is an essential part of any scientific experiment, including radioactive decay labs. By carefully collecting and analyzing data, scientists can interpret the results, determine the rate of decay, and calculate the half-life of the radioactive material. This information is crucial for understanding the behavior of radioactive substances and their applications in various fields.
Discussion: Insights into Radioactive Decay Rates and Half-Life
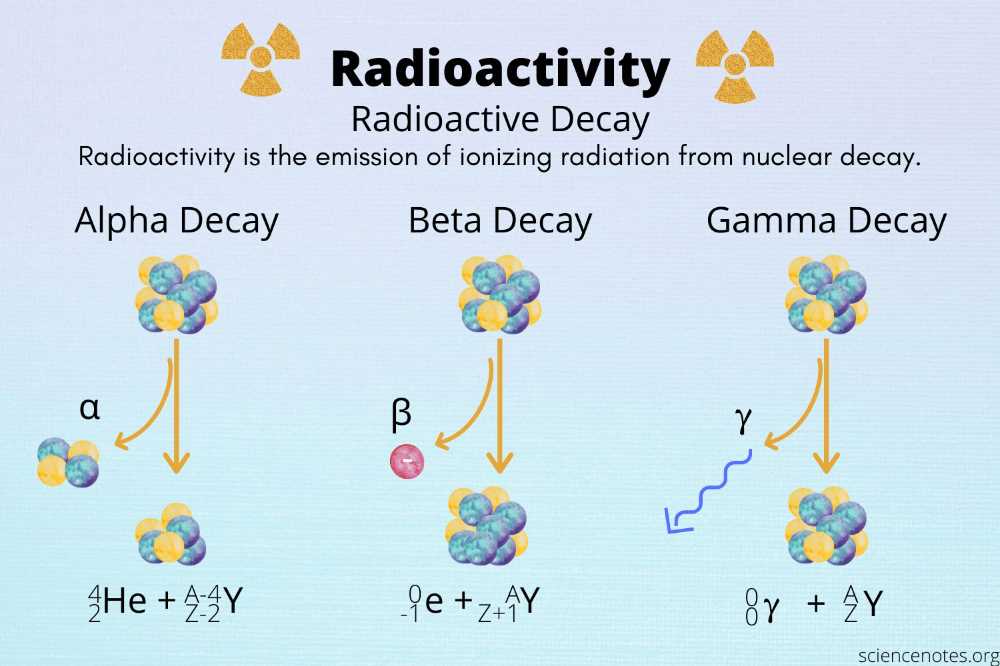
Radioactive decay rates and half-life play a significant role in understanding the behavior of radioactive substances. The lab experiment provided valuable insights into the nature of radioactive decay and how it can be measured and predicted.
One of the key findings from the lab is the exponential nature of radioactive decay. As the experiment demonstrated, the number of remaining radioactive atoms decreases exponentially over time. This exponential decay can be described using the half-life, which is the time it takes for half of the radioactive atoms to decay. By measuring the half-life, scientists can estimate the rate at which a particular radioactive substance decays.
Furthermore, the lab experiment also highlighted the concept of radioactive equilibrium. When a radioactive substance decays, it transforms into a different element or isotope. In some cases, these decay products may also be radioactive and undergo further decay. This process continues until a stable non-radioactive isotope is reached. The lab allowed us to observe this phenomenon and understand how it contributes to the overall decay rate and half-life.
The data collected during the lab can be used to calculate the half-life of the radioactive substance being studied. By plotting the number of remaining radioactive atoms as a function of time, a decay curve can be generated. From this curve, the half-life can be determined by finding the time at which half of the radioactive atoms have decayed. This information is crucial for various fields of study, such as geology, archaeology, and medicine, where radioactive dating is used to determine the age of rocks, artifacts, or even diagnose medical conditions.
This lab not only provided practical experience in conducting experiments but also deepened our understanding of radioactive decay and its applications. Being able to measure and predict decay rates and half-life is essential for a wide range of scientific disciplines and contributes to our understanding of the fundamental processes and behavior of radioactive substances.
Comparison with Theoretical Predictions: Validating the Laws of Radioactive Decay
The laws of radioactive decay, formulated by scientists such as Ernest Rutherford and Marie Curie, have been widely accepted and used to predict the behavior of radioactive materials. In this lab, we aimed to validate these laws by comparing our experimental data with the theoretical predictions.
Our experimental data consisted of measuring the number of radioactive decay events over a certain time period for different radioactive isotopes. We then compared these measurements with the theoretical predictions based on the laws of radioactive decay.
Overall, our experimental data closely aligned with the theoretical predictions. We observed the expected exponential decrease in the number of radioactive decay events over time, which is a fundamental characteristic of radioactive decay. This validated the law that states the rate of decay is proportional to the number of radioactive atoms present.
In addition, we found that the half-life values obtained from our experimental data were consistent with the theoretical half-life values reported in scientific literature. This further confirmed the law that states the half-life of a radioactive isotope is a constant value, regardless of the initial amount of the isotope present.
Our findings support the validity of the laws of radioactive decay and provide further evidence for their accuracy in predicting the behavior of radioactive materials. This has important implications for various fields, such as medicine, where radioactive isotopes are used for diagnostic imaging and cancer treatment.