
Oxidation reduction, also known as redox, is a fundamental concept in chemistry that involves the transfer of electrons between chemical species. It is an essential process in many chemical reactions, including combustion, corrosion, and metabolism. Understanding how oxidation reduction works is crucial for students studying chemistry.
An oxidation reduction worksheet is a valuable tool that helps students practice and reinforce their understanding of redox reactions. It typically contains a series of questions and problems that require students to identify the oxidizing and reducing agents, balance oxidation reduction equations, and calculate oxidation numbers. Having an answer key for these worksheets is essential for students to check their work and ensure they are on the right track.
The oxidation reduction worksheet answer key provides the correct answers and explanations for each question and problem in the worksheet. It allows students to compare their answers to the correct ones and learn from any mistakes they may have made. Moreover, the answer key helps students grasp the underlying concepts and principles of oxidation reduction, enhancing their overall understanding of the topic.
Oxidation Reduction Worksheet Answer Key
Oxidation reduction reactions, also known as redox reactions, are chemical reactions in which there is a transfer of electrons between two species. In these reactions, one species loses electrons and is oxidized, while the other species gains electrons and is reduced. This transfer of electrons allows for the conversion of different chemical species into new compounds.
Understanding these redox reactions is crucial in chemistry, as they play a major role in various processes such as combustion, corrosion, and metabolism. To master the concept of oxidation reduction reactions, students often solve oxidation reduction worksheet problems to practice and reinforce their understanding of the topic.
Answer Key:
Here is an example answer key for an oxidation reduction worksheet:
- Question 1: Identify the oxidizing agent and reducing agent in the following reaction: 2Na + Cl2 -> 2NaCl
- Answer: The oxidizing agent is Cl2, as it is being reduced by gaining electrons. The reducing agent is Na, as it is being oxidized by losing electrons.
- Question 2: Balance the following redox reaction: Fe2O3 + Al -> Al2O3 + Fe
- Answer: The balanced equation for the reaction is 2Fe2O3 + 6Al -> 3Al2O3 + 4Fe.
- Question 3: Calculate the oxidation number of sulfur in H2SO4.
- Answer: The oxidation number of sulfur in H2SO4 is +6.
By practicing with oxidation reduction worksheet problems and reviewing the answer key, students can enhance their understanding of redox reactions and strengthen their problem-solving skills in chemistry.
Overview
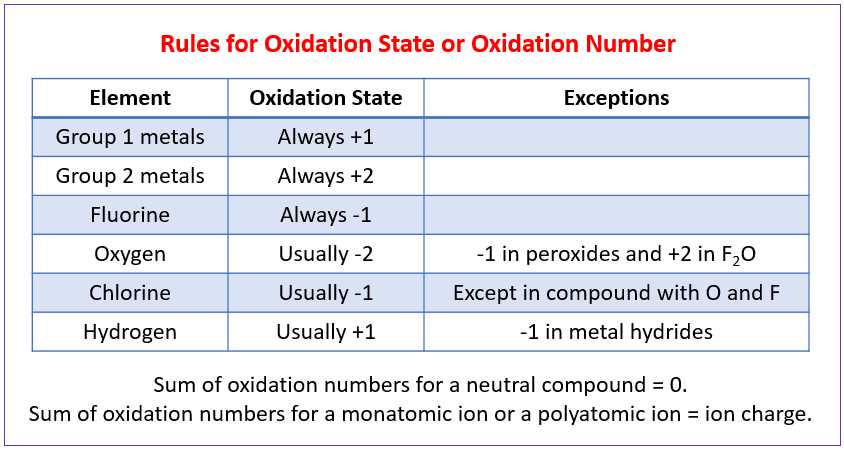
This worksheet provides an answer key for the oxidation-reduction reactions. Oxidation-reduction, also known as redox reactions, involve the transfer of electrons between reactants. In these reactions, one species loses electrons (oxidation) while another species gains electrons (reduction).
The worksheet includes a series of questions and problems that require students to identify the oxidation state of elements, balance redox equations, and determine the oxidizing and reducing agents. The answer key provided here helps students check their work and understand the correct solutions.
Contents:
- Identification of oxidation state: Students are asked to determine the oxidation state of elements in various compounds and ions.
- Redox equation balancing: Students must balance redox equations by ensuring that the number of electrons lost in the oxidation half-reaction is equal to the number of electrons gained in the reduction half-reaction.
- Identification of oxidizing and reducing agents: Students learn to identify the species that undergoes oxidation and reduction in a reaction.
- Calculating oxidation numbers: Students are required to calculate the oxidation numbers of elements in compounds and ions.
The answer key provides step-by-step solutions to each question and problem, making it a valuable resource for students studying oxidation-reduction reactions. By using this answer key, students can check their understanding of the topic and improve their problem-solving skills in this area of chemistry.
What are oxidation and reduction?

Oxidation and reduction are two fundamental processes in chemistry that involve the transfer of electrons between chemical species. These processes are key components in many chemical reactions and play a crucial role in various biological and industrial processes.
Oxidation refers to the loss of electrons by a substance, resulting in an increase in its oxidation state. It is characterized by an increase in the number of oxygen atoms or a decrease in the number of hydrogen atoms in a molecule. Oxidation reactions are commonly associated with the formation of oxides, such as when iron rusts or when metal is exposed to air and undergoes corrosion.
Reduction, on the other hand, refers to the gain of electrons by a substance. It is the opposite of oxidation and results in a decrease in the oxidation state of a substance. Reduction reactions often involve the addition of hydrogen atoms or the removal of oxygen atoms from a molecule. One example of a reduction reaction is the conversion of iron(III) oxide to iron metal in the process of extracting iron from its ore.
Oxidation and reduction always occur together in chemical reactions, and are collectively referred to as redox reactions. In these reactions, one substance is oxidized while another is simultaneously reduced. The species that undergoes oxidation is called the reducing agent, as it causes the reduction of another substance. Conversely, the species that undergoes reduction is called the oxidizing agent, as it causes the oxidation of another substance.
The importance of understanding oxidation and reduction
Oxidation and reduction are fundamental concepts in chemistry and have significant implications in various fields of science and everyday life. Understanding these processes is crucial for students and professionals alike, as they play a pivotal role in numerous chemical reactions and processes.
Oxidation refers to the loss of electrons by a substance, resulting in an increase in its oxidation state. This process is often accompanied by the addition of oxygen or the removal of hydrogen atoms. In contrast, reduction involves the gain of electrons by a substance, leading to a decrease in its oxidation state. These two processes are interconnected and often occur simultaneously, forming what is known as a redox reaction.
One of the primary reasons why understanding oxidation and reduction is important is their role in energy production. Many energy conversion systems, such as batteries and fuel cells, rely on redox reactions to generate electricity. By understanding the principles of oxidation and reduction, scientists and engineers can optimize these systems for improved efficiency and performance.
Additionall , an understanding of oxidation and reduction is essential in the study of organic chemistry. Many organic compounds undergo oxidation and reduction reactions, which can drastically alter their properties and functionality. By recognizing these reactions and their mechanisms, chemists can synthesize new compounds or modify existing ones to create desired products.
Furthermore, oxidation and reduction are vital in environmental science and the study of natural processes. For instance, the oxidation of pollutants in the air can lead to the formation of smog and contribute to air pollution. Understanding these processes allows scientists to develop strategies and technologies to mitigate the impact of pollution and protect the environment.
In conclusion, mastering the concepts of oxidation and reduction is crucial for anyone studying or working in the field of chemistry. These processes are fundamental to various chemical reactions, energy production, organic synthesis, and environmental science. By understanding oxidation and reduction, scientists and professionals can unlock new possibilities in research, technology, and the development of sustainable solutions.
How to balance oxidation-reduction reactions
Oxidation-reduction reactions, also known as redox reactions, involve the transfer of electrons between species. Balancing these reactions can sometimes be challenging, but with a systematic approach, it becomes easier to achieve a balanced equation. There are several steps one can follow to balance oxidation-reduction reactions effectively.
Step 1: Identify the oxidation states
The first step in balancing redox reactions is to identify the oxidation state of each element present in the equation. This can be done by assigning oxidation numbers based on a set of rules. It is essential to determine which elements are being oxidized (losing electrons) and which are being reduced (gaining electrons).
Step 2: Write half-reactions
Next, write separate half-reactions for the oxidation and reduction processes. In the oxidizing half-reaction, write the reactant species on the left side and the products on the right side, including the electrons gained. In the reducing half-reaction, write the reactant species on the left side and the products on the right side, including the electrons lost.
Step 3: Balance atoms
The next step is to balance the number of atoms in each half-reaction. Begin by balancing the atoms other than oxygen and hydrogen. Then, balance the oxygen atoms by adding water molecules to the side deficient in oxygen. Finally, balance the hydrogen atoms by adding hydrogen ions (H+) to the side lacking hydrogen.
Step 4: Balance charges
After balancing the atoms, it is necessary to balance the charges in each half-reaction. Add electrons to the side with a more positive charge to equalize the charges on both sides of the equation. The number of electrons added should be equal to the difference in oxidation states.
Step 5: Combine the half-reactions
The final step is to combine the balanced half-reactions into one overall balanced equation. Multiply each half-reaction by a common factor to ensure that the number of electrons gained equals the number of electrons lost. Cancel out any common species on both sides of the equation and simplify if necessary.
By following these steps, one can successfully balance oxidation-reduction reactions and obtain a balanced equation that accurately represents the transfer of electrons between species.
Oxidation Reduction Worksheet Answer Key: Practice Problems and Answer Key
Now that we have covered the basics of oxidation-reduction reactions and balanced redox equations, let’s test our understanding with some practice problems. Below, you will find a list of problems along with their solutions:
Practice Problem 1:
Balance the following equation using the half-reaction method:
KMnO4 + HCl → KCl + MnCl2 + H2O + Cl2
Solution:
Step 1: Identify the species being oxidized and reduced:
– Mn in KMnO4 is being reduced
– Cl in HCl is being oxidized
Step 2: Write the half-reactions:
Reduction Half-Reaction:
MnO4– → Mn2+
Oxidation Half-Reaction:
Cl– → Cl2
Step 3: Balance the atoms:
Reduction Half-Reaction:
2MnO4– → 2Mn2+
Oxidation Half-Reaction:
2Cl– → Cl2
Step 4: Balance the charges:
Reduction Half-Reaction:
5e– + 8H+ + 2MnO4– → 2Mn2+ + 4H2O
Oxidation Half-Reaction:
10Cl– → 5Cl2 + 10e–
Step 5: Combine the half-reactions:
10Cl– + 8H+ + 2MnO4– → 5Cl2 + 2Mn2+ + 4H2O
Practice Problem 2:
Balance the following equation using the oxidation number method:
Cr2O72- + Fe2+ → Cr3+ + Fe3+
Solution:
| Element | Oxidation Number |
|---|---|
| Cr in Cr2O72- | +6 → +3 |
| Fe in Fe2+ | +2 → +3 |
To balance the charges, we need three Fe atoms on the reactant side, so we multiply the Fe2+ half-reaction by 3. Similarly, we need two Cr atoms on the product side, so we multiply the Cr2O72- half-reaction by 2. The balanced equation is:
2Cr2O72- + 6Fe2+ → 2Cr3+ + 6Fe3+
Summary:
Through these practice problems, we have applied our knowledge of oxidation-reduction reactions and balanced redox equations. By using either the half-reaction method or the oxidation number method, we were able to balance complex chemical equations. Practice like this is essential in mastering the skills needed to tackle more challenging oxidation-reduction problems.
Remember to break down reactions into half-reactions, balance the atoms and charges, and then combine the half-reactions to obtain a balanced equation. Keep practicing, and soon you will become proficient at balancing redox equations!