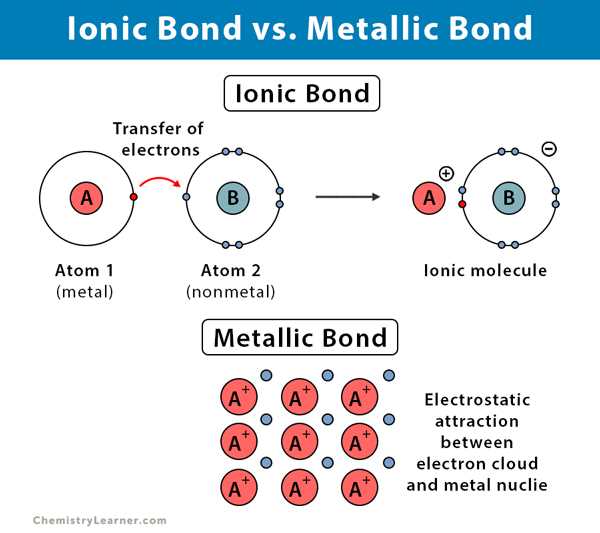
Understanding the nature of chemical bonding is crucial in the field of chemistry, as it helps explain the properties and behavior of substances. Two common types of chemical bonding are ionic and metallic bonding.
Ionic bonding occurs when there is a transfer of electrons between atoms, resulting in the formation of ions. One atom loses electrons to become positively charged (cation), while the other atom gains electrons to become negatively charged (anion). These oppositely charged ions are held together by electrostatic forces, forming a strong and stable ionic bond.
On the other hand, metallic bonding occurs between metal atoms. In this type of bonding, the valence electrons of metal atoms are delocalized, meaning they are free to move throughout the metal lattice. This creates a sea of electrons that hold the metal ions together. The positive metal ions are attracted to the negative charge of the electron sea, forming a cohesive and malleable structure.
So what are some key differences between ionic and metallic bonding? In ionic bonding, electrons are transferred between atoms, while in metallic bonding, electrons are delocalized. Ionic compounds typically have high melting and boiling points, as the electrostatic forces between ions are strong. In contrast, metallic compounds have variable melting points, as the strength of metallic bonding depends on factors such as the size and charge of the metal ions.
Ionic and Metallic Bonding: Explained and Answered
Ionic and metallic bonding are two types of chemical bonding that occur between atoms. These bonds play a crucial role in determining the physical and chemical properties of substances.
Ionic bonding occurs when there is a transfer of electrons from one atom to another, resulting in the formation of ions. One atom loses electrons to become a positively charged ion (cation), while the other atom gains electrons to become a negatively charged ion (anion). The oppositely charged ions are then held together by electrostatic forces, creating a strong bond. This type of bonding typically occurs between a metal and a non-metal atom, such as sodium and chlorine in sodium chloride (NaCl).
Metallic bonding, on the other hand, occurs within metals. In this type of bonding, the valence electrons of metal atoms are delocalized and free to move throughout the metal lattice. The positive metal ions are surrounded by a “sea” of electrons, creating a strong attraction between the positive ions and the delocalized electrons. This gives metals their characteristic properties, such as high conductivity, malleability, and ductility.
In conclusion, ionic bonding involves the transfer of electrons between atoms, leading to the formation of ions and strong electrostatic attractions. Metallic bonding occurs within metals, where the valence electrons are delocalized and create a strong attraction between positive ions and the “sea” of electrons. These two types of bonding have distinct characteristics and play a fundamental role in the properties of substances.
What is ionic bonding?
Ionic bonding is a type of chemical bonding that occurs between atoms when one atom donates electrons to another atom. This transfer of electrons results in the formation of positively charged ions (cations) and negatively charged ions (anions), which are then attracted to each other by electrostatic forces.
In ionic bonding, the atoms involved usually belong to elements with significantly different electronegativities. The atom with a lower electronegativity tends to donate its electrons to the atom with a higher electronegativity. This electron transfer leads to the formation of ionic compounds, such as salts.
During the process of ionic bonding, the atom that donates electrons becomes positively charged due to the loss of electrons, while the atom that accepts electrons becomes negatively charged. The resulting ions are held together in a regular, repeating pattern called a crystal lattice. This arrangement allows for the formation of strong bonds between the ions.
Ionic bonding is often associated with the formation of compounds with distinct properties, such as high melting and boiling points, as well as brittleness. These properties are a result of the strong electrostatic forces between the oppositely charged ions in the crystal lattice.
Overall, ionic bonding plays a crucial role in the formation of many types of compounds and materials, and it is a fundamental concept in the field of chemistry.
How does ionic bonding occur?
Ionic bonding occurs between atoms when one or more valence electrons are transferred from one atom to another. It typically involves a metal and a nonmetal element. The metal atom, which has a lower electronegativity, loses one or more electrons, becoming a positively charged ion or cation. The nonmetal atom, which has a higher electronegativity, gains the electrons, becoming a negatively charged ion or anion.
The attraction between these oppositely charged ions is what holds the ionic bond together. The positive and negative charges attract each other, forming a strong electrostatic attraction. This results in the formation of a crystal lattice structure, where the ions are arranged in a repeating pattern. The lattice structure of an ionic compound is what gives it its unique properties, such as high melting and boiling points, brittleness, and the ability to conduct electricity in the molten or aqueous state.
During the formation of an ionic bond, the metal atom becomes stable by losing electrons to achieve a noble gas electron configuration. The nonmetal atom becomes stable by gaining electrons to fill its valence shell. This transfer of electrons allows both atoms to achieve a more stable and energetically favorable state.
Overall, ionic bonding occurs through the transfer of electrons between a metal and a nonmetal atom, resulting in the formation of a crystal lattice structure held together by strong electrostatic attractions.
Properties of Ionic Compounds
Ionic compounds are formed through the transfer of electrons between atoms, resulting in the formation of positive and negative ions. These compounds have unique properties due to their ionic bonding.
High melting and boiling points: Ionic compounds have high melting and boiling points because the strong electrostatic attractions between the positive and negative ions require a significant amount of energy to break the bonds and convert the solid structure into a liquid or gas.
Crystal lattice structure: Ionic compounds often have crystalline structures, where the positive and negative ions are arranged in a repeating pattern. This structure contributes to the high melting and boiling points of these compounds.
Conductivity of electricity: When in a molten state or dissolved in water, ionic compounds can conduct electricity. This is because the movement of ions allows the flow of electric current. However, in their solid state, ionic compounds do not conduct electricity because the ions are locked in place.
Brittle nature: Ionic compounds are often brittle and can easily break or shatter when subjected to external forces. This is due to the regular arrangement of ions in the crystal lattice structure. When a force is applied, the ions of like charge come into close proximity, causing repulsion and leading to the breaking of the crystal lattice.
Dissolution in water: Many ionic compounds dissolve readily in water due to the attraction between the polar water molecules and the charged ions. This ability to dissolve in water allows for easy separation and transport of ions, making ionic compounds important in various chemical and biological processes.
Examples of ionic compounds

Ionic compounds are compounds formed between a metal and a non-metal. They are characterized by their high melting and boiling points, as well as their ability to conduct electricity when dissolved in water or melted. Here are some examples of common ionic compounds:
- Sodium chloride (NaCl): This is a classic example of an ionic compound. It is made up of sodium ions (Na+) and chloride ions (Cl-) arranged in a crystal lattice structure. Sodium chloride is commonly known as table salt.
- Potassium iodide (KI): This compound is formed between potassium ions (K+) and iodide ions (I-). It is often used in medical applications, such as in thyroid treatments.
- Copper(II) sulfate (CuSO4): This compound consists of copper ions (Cu2+) and sulfate ions (SO42-). It is commonly used as a fungicide and in educational experiments.
- Magnesium oxide (MgO): This compound is made up of magnesium ions (Mg2+) and oxide ions (O2-). It is commonly used as a refractory material and in the production of magnesium metal.
These are just a few examples of the many ionic compounds that exist. Overall, ionic compounds play important roles in various fields, including industry, medicine, and everyday life.
What is metallic bonding?
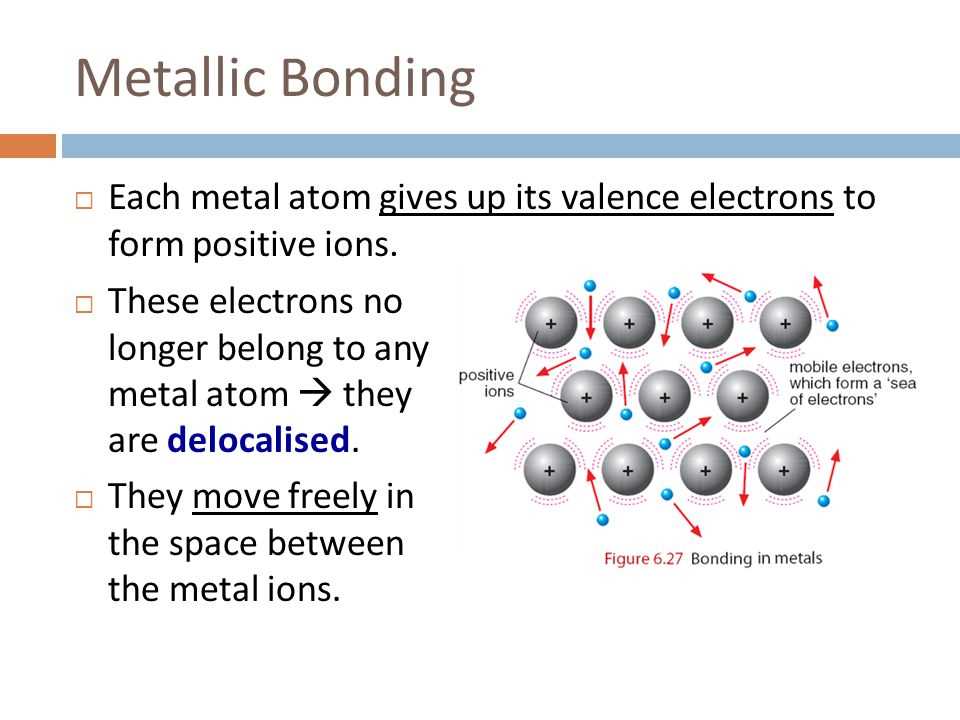
Metallic bonding is a type of chemical bonding that occurs between atoms of metallic elements. In metallic bonding, the outermost electrons of the metal atoms are delocalized and are free to move throughout the entire crystal lattice of the metal. This delocalization of electrons gives metals their unique properties, such as high electrical and thermal conductivity, malleability, and ductility.
In metallic bonding, the metal atoms form a lattice structure, with the delocalized electrons forming a “sea” of electron cloud surrounding the positively charged metal ions. This electron cloud is responsible for the characteristic properties of metals. When a voltage is applied to a metal, the delocalized electrons can easily move through the lattice, creating an electric current. Similarly, when heat is applied, the delocalized electrons can transfer thermal energy very effectively.
The strength of metallic bonding is determined by factors such as the number of delocalized electrons per atom and the size of the metal ions. Metals with more delocalized electrons and smaller ions tend to have stronger metallic bonding. This is why metals like copper and gold, which have a high number of delocalized electrons per atom, exhibit higher tensile strengths and melting points compared to metals like sodium and potassium.
In summary, metallic bonding is a type of chemical bonding that occurs between atoms of metallic elements. It involves the delocalization of electrons, creating a sea of electron cloud surrounding the metal ions. This delocalization gives metals their characteristic properties, such as high conductivity and malleability.
The nature of metals
Metallic bonding is a type of chemical bonding that occurs between atoms of metallic elements. In a metallic bond, atoms share their outermost electrons, forming a common electron cloud or sea of electrons. This is what gives metals their unique properties such as conductivity, malleability, and ductility.
This sharing of electrons in metallic bonding allows for the formation of a lattice structure, where positive metal ions are surrounded by this sea of delocalized electrons. The positive ions are held together by the attraction of their opposite charges to the negative charge of the electron cloud. This is what gives metals their high melting and boiling points, as the metallic bonds are strong and require a significant amount of energy to break.
Metals also have high electrical and thermal conductivity due to the mobility of the delocalized electrons. These electrons are free to move throughout the metal structure, allowing for the easy flow of electric current and the transfer of thermal energy. This is why metals are commonly used in electrical wiring and as heat conductors.
In addition, metals are generally malleable and ductile, meaning they can be easily shaped into thin sheets or drawn into wires. This is due to the ability of the metallic bonds to move and rearrange without breaking. The delocalized electrons can flow and fill in the gaps created during deformation, allowing the metal to maintain its structure.
In summary, the nature of metals is defined by their metallic bonding, which involves the sharing of electrons to form a sea of delocalized electrons. This bonding gives metals their unique properties such as conductivity, malleability, and ductility, making them essential materials in various industries and applications.
Properties of metals
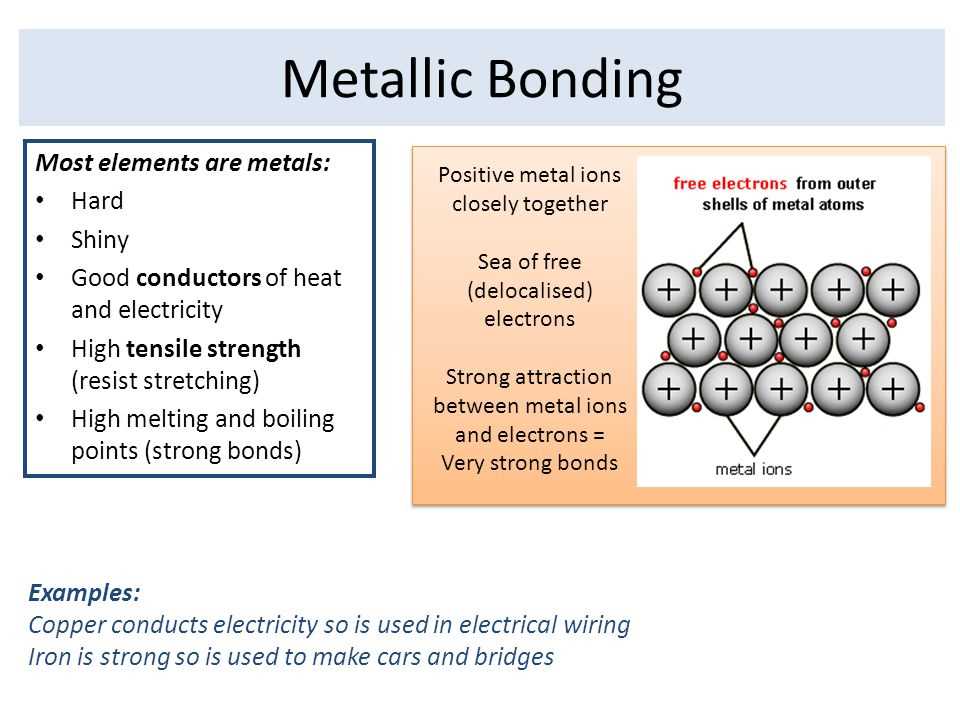
Metallic elements are characterized by a range of unique properties that set them apart from nonmetals. These properties include their high thermal and electrical conductivity, malleability, ductility, and luster.
Thermal conductivity: Metals are excellent conductors of heat. This means that they are able to transfer heat energy easily and quickly. This property makes metals ideal for applications where efficient heat transfer is necessary, such as in electrical wiring or cooking utensils.
Electrical conductivity: Similar to their high thermal conductivity, metals also have exceptional electrical conductivity. This means that they allow the flow of electric current without significant resistance. This property is crucial for the functioning of electrical circuits and devices.
Malleability: Metals can be easily hammered or pressed into different shapes without breaking. This property is known as malleability and is due to the presence of delocalized electrons that allow for the movement of metal atoms without disrupting the overall structure.
Ductility: Ductility is the property of metals to be stretched into thin wires without breaking. This property is also a result of the presence of delocalized electrons. Ductile metals are commonly used in electrical wiring and other applications where the ability to form long, thin shapes is important.
Luster: Metals have a characteristic shine or luster, which is due to the way they reflect light. This property is a result of the arrangement of metal atoms and their ability to absorb and re-emit light in a way that creates a shiny appearance. The luster of metals is often used for decorative purposes.
Overall, these unique properties of metals make them invaluable for a wide range of applications in various industries, from construction and electronics to transportation and jewelry-making.