
Welcome to the Chem 101 final exam study guide! This guide has been carefully curated to help you prepare for your upcoming chemistry exam. Here, you will find a comprehensive overview of the key topics and concepts that will be covered on the exam.
In this study guide, we will cover a range of important topics, including chemical reactions, atomic structure, periodic trends, and thermodynamics. Each topic will be broken down into manageable sections, with clear explanations and examples to aid your understanding.
As you work through this study guide, it is important to actively engage with the material. Take notes, ask questions, and complete practice problems to solidify your knowledge. Remember, the more you actively participate in the learning process, the better prepared you will be for the exam.
Whether you are new to chemistry or simply looking to brush up on your skills, this study guide is a valuable resource for anyone preparing for a Chem 101 final exam. So, let’s dive in and start mastering the key concepts that will lead to your success on exam day!
Understanding the Importance of the Chem 101 Final Exam

The Chem 101 final exam holds great significance for students pursuing a degree in chemistry or related fields. This comprehensive assessment is designed to test their understanding of the fundamental concepts and principles covered throughout the course. It serves as a pivotal moment, determining students’ mastery of the subject, their ability to critically think, solve problems, and apply their knowledge in real-world scenarios.
The Chem 101 final exam covers a wide range of topics, including atomic structure, chemical bonding, stoichiometry, thermodynamics, and chemical reactions. By requiring students to demonstrate their knowledge and skills in these areas, the exam serves as a summative evaluation that measures their overall comprehension and retention of the course material.
The outcome of the Chem 101 final exam can have numerous implications for students’ academic and professional pathways. A strong performance on the exam can not only boost students’ confidence in their ability to excel in the field of chemistry but also open doors for advanced studies and research opportunities. It may also serve as a strong foundation for success in subsequent courses that build upon the concepts covered in Chem 101.
On the other hand, a poor performance on the Chem 101 final exam can have repercussions, potentially leading to lower grades in the course or even the need to retake it. It can also indicate gaps in understanding that may hinder students’ progress in future chemistry-related courses. Recognizing the importance of the final exam, students are motivated to invest time and effort in their studies, engage in thorough revision, seek clarification on challenging topics, and practice problem-solving to enhance their chances of success.
In conclusion, the Chem 101 final exam is a critical assessment that enables students to demonstrate their understanding of foundational chemistry concepts. It plays a key role in determining their academic and professional trajectory in the field. By recognizing its importance, students are inspired to dedicate themselves to their studies and strive for excellence in this crucial evaluation.
In the world of chemistry, the final exam for Chem 101 is a crucial milestone that can greatly impact your academic success.

First and foremost, the Chem 101 final exam is an opportunity to showcase your understanding and mastery of the fundamental concepts and principles of chemistry. Throughout the course, you have learned about atoms, molecules, chemical reactions, and various other topics that lay the foundation for more advanced studies in chemistry. The final exam allows you to demonstrate your ability to apply this knowledge and think critically about chemical problems.
Moreover, the Chem 101 final exam serves as a comprehensive assessment of your learning and retention. Chemistry is a cumulative subject, meaning that concepts build upon one another. The final exam tests your understanding of not only the recent material covered, but also the concepts and skills learned earlier in the semester. It evaluates how well you have been able to synthesize and connect different aspects of chemistry, and how effectively you can recall and apply this knowledge.
To succeed on the Chem 101 final exam, it is important to review and consolidate your understanding of the course material. This may involve going over lecture notes, textbook readings, and completing practice problems. Additionally, seeking help from professors or participating in study groups can provide valuable insights and clarification on challenging topics.
Ultimately, excelling on the Chem 101 final exam can have a significant impact on your academic success. It not only demonstrates your competency in chemistry, but can also contribute to your overall GPA and academic standing. Additionally, a strong performance on the final exam can provide a solid foundation for future chemistry courses and career opportunities in fields such as medicine, pharmacy, or chemical research. Therefore, dedicating time and effort to prepare for and perform well on the Chem 101 final exam is essential for achieving your academic goals in chemistry and beyond.
Reviewing key concepts in general chemistry
As you prepare for your general chemistry final exam, it is important to review and understand the key concepts that have been covered throughout the course. Here are some topics that you should focus on:
Atomic structure:
Make sure you understand the basic structure of an atom, including the different subatomic particles (protons, neutrons, and electrons) and their charges. Review the periodic table and understand how to read and interpret the information it provides, such as atomic number, atomic mass, and the arrangement of elements.
Chemical bonding:
Review the different types of chemical bonds, including covalent, ionic, and metallic bonds. Understand how these bonds are formed and how they contribute to the properties of compounds. Review Lewis dot structures and understand how to determine the number of valence electrons for different elements.
Chemical reactions:
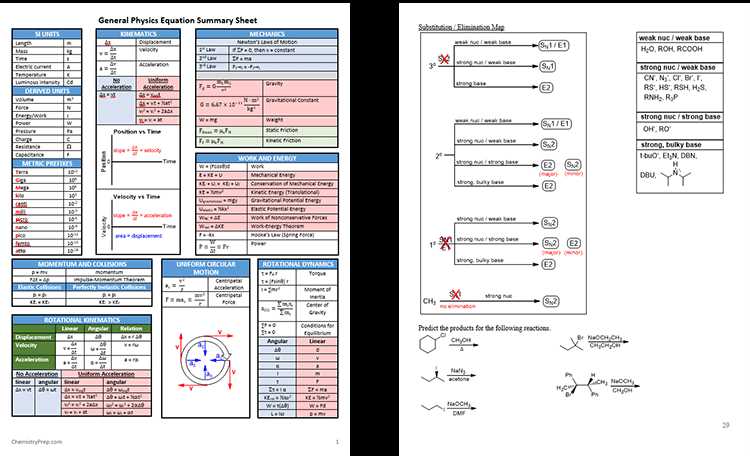
Refresh your understanding of different types of chemical reactions, such as synthesis, decomposition, single displacement, and double displacement reactions. Review balancing chemical equations and understanding the concept of stoichiometry.
States of matter and phase changes:
Review the properties of solids, liquids, and gases, including their physical and chemical properties. Understand the concepts of temperature, pressure, and volume and how they relate to the behavior of substances in different states. Review phase diagrams and the different types of phase changes, such as melting, boiling, and sublimation.
Acids and bases:

Understand the concepts of pH, pOH, and the pH scale. Review the properties of acids and bases, including their reactions and their behavior in water. Understand the concept of acid-base equilibrium and how to calculate the pH of a solution.
By reviewing these key concepts, you will be well-prepared for your general chemistry final exam. Good luck!
Brushing up on the foundational principles of general chemistry
As you prepare for your Chem 101 final exam, it’s important to review and refresh your understanding of the foundational principles of general chemistry. These principles serve as the building blocks for more advanced concepts and help to explain the behavior of matter and chemical reactions.
Atomic structure: Understanding the structure of atoms is crucial in chemistry. Recall the basic components of an atom, including protons, neutrons, and electrons. Review the periodic table to familiarize yourself with the different elements and their atomic numbers, which determine the number of protons in an atom.
- Protons: Positively charged particles found in the nucleus of an atom.
- Neutrons: Neutral particles found in the nucleus of an atom.
- Electrons: Negatively charged particles that orbit the nucleus in energy levels or shells.
Chemical bonding: Understanding how atoms bond together is essential in understanding the behavior and properties of compounds. Review the different types of chemical bonds, including ionic bonds, covalent bonds, and metallic bonds.
- Ionic bonds: Formed when there is a transfer of electrons from one atom to another, resulting in the formation of ions with opposite charges that are held together by electrostatic attraction.
- Covalent bonds: Formed when atoms share electrons, creating a strong bond between the atoms involved.
- Metallic bonds: Found in metals, where the electrons are delocalized and free to move throughout the metal lattice.
Chemical reactions: Familiarize yourself with the different types of chemical reactions, including synthesis, decomposition, combustion, and displacement reactions. Understand the concept of balancing chemical equations and how to interpret and predict the products of a reaction using stoichiometry.
| Reaction Type | General Equation | Example |
|---|---|---|
| Synthesis | A + B → AB | 2Na + Cl2 → 2NaCl |
| Decomposition | AB → A + B | 2H2O → 2H2 + O2 |
| Combustion | Fuel + Oxygen → Carbon Dioxide + Water | CH4 + 2O2 → CO2 + 2H2O |
| Displacement | A + BC → AC + B | Zn + 2HCl → ZnCl2 + H2 |
By reviewing and understanding these foundational principles, you will be better prepared to tackle the more complex concepts and problem-solving tasks that may appear on your Chem 101 final exam.
Examining Important Theories and Laws in Chemistry

In the study of chemistry, there are several important theories and laws that form the foundation of the field. These theories and laws help scientists understand the behavior of matter and the interactions between different substances. By examining these theories and laws, we can gain a deeper insight into the principles that govern chemical reactions and processes.
One of the most fundamental theories in chemistry is the Atomic Theory, which states that all matter is composed of tiny, indivisible particles called atoms. This theory was first proposed by John Dalton in the early 19th century and has since been supported by numerous experimental observations. The Atomic Theory provides a framework for understanding the composition, structure, and properties of all substances.
Another important theory in chemistry is the Quantum Theory, which describes the behavior of particles at the atomic and subatomic levels. This theory incorporates principles from quantum mechanics to explain phenomena such as electron orbital arrangements, energy levels, and the quantized nature of certain properties, such as angular momentum. The Quantum Theory has revolutionized our understanding of the behavior and properties of matter on a very small scale.
In addition to theories, there are also several fundamental laws in chemistry that provide a basis for understanding chemical reactions and processes. For example, the Law of Conservation of Mass states that mass is neither created nor destroyed in a chemical reaction, but is instead conserved. This law allows scientists to balance chemical equations and predict the amounts of reactants and products in a given reaction.
Another important law is the Law of Definite Proportions, which states that a chemical compound always contains the same elements in the same proportions by mass. This law helps in determining the empirical formula of a compound and understanding the stoichiometry of chemical reactions. By examining these theories and laws, chemists are able to understand and predict the behavior of matter, leading to advancements in various fields such as medicine, materials science, and environmental studies.
Exploring essential topics covered in Chem 101
Chemistry 101, often called Chem 101, is an introductory course that covers the foundational topics in chemistry. It provides students with a solid understanding of the basic principles and concepts of this scientific discipline. In this article, we will explore some of the essential topics covered in Chem 101.
Atomic structure: Chem 101 begins with an exploration of atomic structure. Students learn about the subatomic particles that make up an atom, including protons, neutrons, and electrons. They also study the periodic table, which organizes elements based on their atomic number, mass, and electron configuration.
Chemical bonding: Another important topic covered in Chem 101 is chemical bonding. Students learn about different types of chemical bonds, such as ionic, covalent, and metallic bonds. They also study the concept of Lewis structures and learn how to predict the shapes and polarity of molecules.
- Ionic bonds: Ionic bonds are formed when one or more electrons are transferred from one atom to another. This results in the formation of positively and negatively charged ions, which are attracted to each other.
- Covalent bonds: Covalent bonds are formed when atoms share electrons. This sharing of electrons allows atoms to achieve a stable electron configuration.
- Metallic bonds: Metallic bonds are formed between metal atoms. In this type of bond, electrons are free to move throughout the metal lattice, creating a sea of delocalized electrons.
Chemical reactions: Chem 101 also covers the fundamental concepts of chemical reactions. Students learn about the different types of chemical reactions, such as synthesis, decomposition, single replacement, and double replacement. They also study stoichiometry, which involves balancing chemical equations and calculating the quantities of reactants and products involved in a reaction.
States of matter: Lastly, Chem 101 introduces students to the three states of matter: solid, liquid, and gas. They learn about the properties and behavior of each state, as well as the phase transitions between them, such as melting, boiling, and condensation.
In conclusion, Chem 101 covers a wide range of essential topics in chemistry, including atomic structure, chemical bonding, chemical reactions, and states of matter. By understanding these foundational concepts, students can build a strong foundation for further studies in chemistry.