
If you are studying chemistry or physics, you may have come across the concept of Charles’ Law. This law states that as the temperature of a gas increases, its volume also increases, as long as the pressure remains constant. Understanding and applying this law is crucial in many scientific experiments and calculations.
To test your knowledge and understanding of Charles’ Law, you may have been given a worksheet with various questions and problems to solve. These questions typically involve manipulating the variables of temperature and volume and applying the principles of Charles’ Law to find the unknown values.
When solving a Charles’ Law worksheet, it is important to carefully read and understand each question. Pay attention to the given information and identify the variables provided. You may need to convert the temperature from Celsius to Kelvin, as Charles’ Law is typically expressed in Kelvin. Then, use the formula V1/T1 = V2/T2, where V1 and V2 represent the initial and final volumes, and T1 and T2 represent the initial and final temperatures.
Understanding Charles Law and Its Worksheet Answers
The concept of Charles Law is an important topic in the study of gases and their behavior. It describes the relationship between the volume and temperature of a gas, assuming that the pressure and amount of gas remain constant. According to Charles Law, the volume of a gas is directly proportional to its temperature in Kelvin.
Charles Law can be mathematically expressed as V1/T1 = V2/T2, where V1 and T1 represent the initial volume and temperature, and V2 and T2 represent the final volume and temperature. This equation allows us to predict changes in volume when the temperature of a gas is altered.
Worksheet answers related to Charles Law usually involve solving for an unknown variable in the equation. Students may be given the initial volume, temperature, and one of the final variables (volume or temperature), and they need to determine the missing value. This requires applying the equation and rearranging it to solve for the unknown.
Additionally, Charles Law worksheet answers may involve interpreting and analyzing data from experiments or word problems. Students may be given data points representing the volume and temperature of a gas under different conditions, and they have to identify patterns or trends that align with Charles Law. This helps to reinforce the understanding of how temperature affects the volume of a gas.
In summary, understanding Charles Law and its worksheet answers is crucial in mastering the concept of gas behavior. It allows students to manipulate the equation, solve for unknown variables, and interpret experimental data to reinforce their understanding of this fundamental principle. By practicing with worksheet exercises, students can enhance their problem-solving skills and apply Charles Law to real-world scenarios.
What Is Charles Law?
Charles Law, also known as Gay-Lussac’s law, is a fundamental principle in the field of thermodynamics that describes the relationship between the temperature and volume of a gas. It states that the volume of a gas is directly proportional to its absolute temperature, assuming that the pressure and the amount of gas remain constant.
This law was formulated by French chemist Joseph Louis Gay-Lussac in the early 19th century but is commonly credited to British scientist John Charles who performed similar experiments around the same time. The law is widely used in various scientific applications, especially in the study of gases and their behavior.
In mathematical terms, Charles Law can be expressed as V₁/T₁ = V₂/T₂, where V₁ and T₁ represent the initial volume and temperature of the gas, while V₂ and T₂ represent the final volume and temperature. This equation shows that as the temperature of a gas increases, its volume also increases proportionally, and vice versa.
Charles Law is based on the kinetic theory of gases, which assumes that gases consist of tiny particles in constant motion. According to this theory, when the temperature of a gas increases, the particles move faster, resulting in an increase in their kinetic energy. Consequently, the particles collide more frequently with the container walls, leading to an increase in pressure and volume.
Charles Law has numerous practical applications in various fields, including chemistry, physics, and engineering. It is particularly useful in areas such as gas systems design, thermodynamic calculations, and the study of heat transfer and fluid dynamics. By understanding and applying Charles Law, scientists and engineers can make more accurate predictions and calculations related to gases and their behavior.
What Does Charles Law State?
Charles Law, also known as the law of volumes, is one of the fundamental gas laws in thermodynamics. It states that the volume of a gas is directly proportional to its absolute temperature when pressure is held constant. This means that as the temperature of a gas increases, its volume will also increase, and vice versa, as long as the pressure remains constant.
A simple way to express Charles Law mathematically is through the equation: V1/T1 = V2/T2, where V1 and V2 represent the initial and final volumes of the gas, and T1 and T2 represent the initial and final temperatures of the gas in kelvin. This equation demonstrates the direct relationship between volume and temperature.
Charles Law is a consequence of the kinetic theory of gases, which states that gas molecules are in constant random motion. When the temperature of a gas increases, the kinetic energy of its molecules also increases. As a result, the molecules move faster and collide more frequently with the walls of the container, leading to an increase in volume. On the other hand, when the temperature decreases, the gas molecules move slower, resulting in fewer collisions with the container walls and a decrease in volume.
Charles Law has numerous applications in various fields, including weather forecasting, scuba diving, and hot air ballooning. It helps explain the behavior of gases under different temperature conditions and allows scientists and engineers to make accurate predictions and calculations related to gas volumes. Understanding Charles Law is essential for mastering the principles of thermodynamics and gas behavior.
Overall, Charles Law states that as the temperature of a gas increases or decreases, its volume will change proportionally as long as the pressure remains constant. This relationship between temperature and volume is a fundamental concept in the study of gases and has practical applications in many scientific and technological fields.
How Does Charles Law Relate to the Ideal Gas Law?
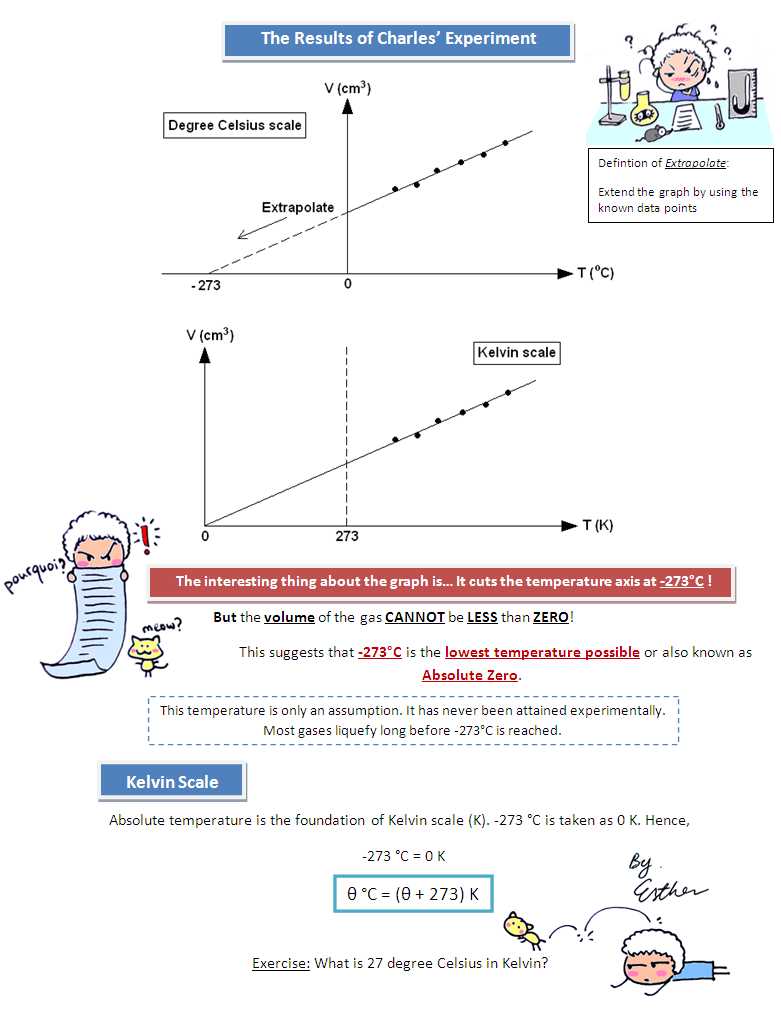
Charles Law is a fundamental principle in the field of thermodynamics that describes the relationship between the volume and temperature of an ideal gas. According to Charles Law, when the temperature of an ideal gas is increased, its volume will also increase, as long as the pressure is held constant. This can be expressed mathematically as V1/T1=V2/T2, where V1 and V2 are the initial and final volumes of the gas, and T1 and T2 are the initial and final temperatures.
Charles Law is one of the important components of the Ideal Gas Law, which is a mathematical equation that relates the pressure, volume, temperature, and number of moles of an ideal gas. The Ideal Gas Law equation is expressed as PV = nRT, where P is the pressure, V is the volume, n is the number of moles of gas, R is the ideal gas constant, and T is the temperature in Kelvin. The Ideal Gas Law combines the principles of Charles Law, Boyle’s Law, and Avogadro’s Law to describe the behavior of ideal gases.
By understanding Charles Law, we can better comprehend the behavior of ideal gases in various experimental conditions. The relationship between volume and temperature established by Charles Law is a crucial factor in determining the properties and behavior of gases, especially under conditions of constant pressure. The Ideal Gas Law provides a comprehensive equation that incorporates Charles Law along with other gas laws, enabling scientists and researchers to make accurate predictions and calculations in the field of thermodynamics.
How to Solve Charles Law Problems?

Solving Charles Law problems involves understanding the relationship between temperature and volume of a gas at constant pressure. Here are the steps to solve Charles Law problems:
- Identify the variables: Determine the known and unknown variables in the problem. The known variables typically include the initial volume and temperature, as well as any changes in volume or temperature. The unknown variable is usually the final volume or temperature.
- Convert units: Make sure all the units are consistent. Convert any given units to the same unit system (e.g., Celsius to Kelvin or Fahrenheit to Kelvin) to ensure accurate calculations.
- Apply the Charles Law equation: Charles Law is represented by the equation V1/T1 = V2/T2, where V1 and T1 are the initial volume and temperature, and V2 and T2 are the final volume and temperature. Use this equation to relate the known and unknown variables.
- Perform the calculations: Substitute the known values into the Charles Law equation and solve for the unknown variable. Rearrange the equation if necessary to isolate the unknown variable.
- Check the solution: Verify that the solution makes sense in the context of the problem. Ensure that the units are correct and that the answer aligns with any given constraints or conditions.
By following these steps, you can effectively solve Charles Law problems and determine the relationship between temperature and volume in a gas system.
Worksheet Answers for Charles Law Problems

Worksheet answers for Charles Law problems can help students understand and apply the relationship between temperature and volume of a gas. By providing a set of problems and their corresponding answers, students can practice solving Charles Law equations and gain confidence in their understanding of the topic.
Example Problem 1: A gas at 25°C has a volume of 2.5 L. What will be its volume when the temperature is increased to 50°C?
Answer: Using Charles Law, we know that the volume of a gas is directly proportional to its temperature. Therefore, we can set up the following equation:
V₁/T₁ = V₂/T₂
2.5 L / 25°C = V₂ / 50°C
Simplifying the equation, we get:
2.5 L * 50°C = V₂ * 25°C
125 L°C = 25 V₂
V₂ = 125 L°C / 25
V₂ = 5 L
Therefore, the volume of the gas will be 5 L when the temperature is increased to 50°C.
Example Problem 2: A gas at 5 L and 10°C is compressed to a volume of 2 L. If the temperature remains constant, what will be the new temperature?
Answer: Since the temperature remains constant, we can use the formula:
V₁/T₁ = V₂/T₂
5 L / 10°C = 2 L / T₂
Simplifying the equation, we get:
5 L * T₂ = 2 L * 10°C
T₂ = 20°C
Therefore, the new temperature will be 20°C.
In summary, worksheet answers for Charles Law problems provide students with the opportunity to practice and apply their knowledge of the relationship between temperature and volume of a gas. By solving a variety of problems, students can strengthen their understanding of Charles Law and improve their problem-solving skills.
Importance of Understanding Charles Law

The understanding of Charles Law is of great significance in the field of thermodynamics and gas behavior. This empirical gas law, formulated by French scientist Jacques Charles in the late 18th century, states that the volume of a gas is directly proportional to its temperature, given that the pressure and amount of gas remain constant. Charles Law can be expressed mathematically as:
V = k * T
Where V represents the volume of the gas, T represents the temperature of the gas in Kelvin, and k is a constant.
Having a thorough understanding of Charles Law allows scientists and engineers to predict and manipulate the behavior of gases under different conditions. Here are a few reasons why understanding Charles Law is important:
- Prediction of Gas Volume Changes: Charles Law enables scientists and engineers to predict how the volume of a gas will change with a change in temperature, assuming constant pressure and amount of gas. This is particularly useful when designing and operating systems that involve the handling or storage of gases, such as in industrial processes or gas cylinders.
- Temperature Control in Gas-based Systems: Charles Law helps in controlling the temperature of a gas-based system. By manipulating the volume of a gas, the temperature can be controlled to achieve desired outcomes, such as maintaining a specific temperature range in a chemical reaction or adjusting the temperature in heating or cooling systems.
- Understanding the Behavior of Gases: Charles Law provides insights into the behavior of gases at different temperatures. It helps in understanding how gases expand or contract when exposed to varying temperatures and how this behavior can impact other properties of gases, such as pressure and density.
- Applications in Various Fields: Understanding Charles Law has numerous applications in various fields, including chemistry, physics, engineering, and meteorology. It allows for accurate modeling and simulation of gas-based processes, analysis of gas behavior in different environments, and optimization of systems involving gases.
In conclusion, Charles Law is a fundamental concept in the study of gases and has wide-ranging applications in different fields. It helps in predicting and controlling gas volume changes, temperature control in gas-based systems, understanding gas behavior, and finding applications in various scientific and engineering disciplines. Therefore, a clear understanding of Charles Law is crucial for anyone working with gases or studying thermodynamics.