Understanding the concepts of Bronsted-Lowry acids and bases is essential for a deeper comprehension of acid-base reactions. This worksheet provides answers to questions that will help solidify your understanding of these fundamental concepts.
The worksheet begins with a series of definitions and explanations, allowing you to review the basic terms associated with Bronsted-Lowry acids and bases. It then moves on to a series of practice questions that test your knowledge of acid and base strength, conjugate acid-base pairs, and the identification of acids and bases in given reactions.
By working through this worksheet and checking your answers against the provided solutions, you can identify any areas where you may need further study. This will help you build a strong foundation in the concepts of Bronsted-Lowry acids and bases, which are important not only in chemistry but also in biology and environmental science.
Bronsted Lowry Acids and Bases Worksheet Answers
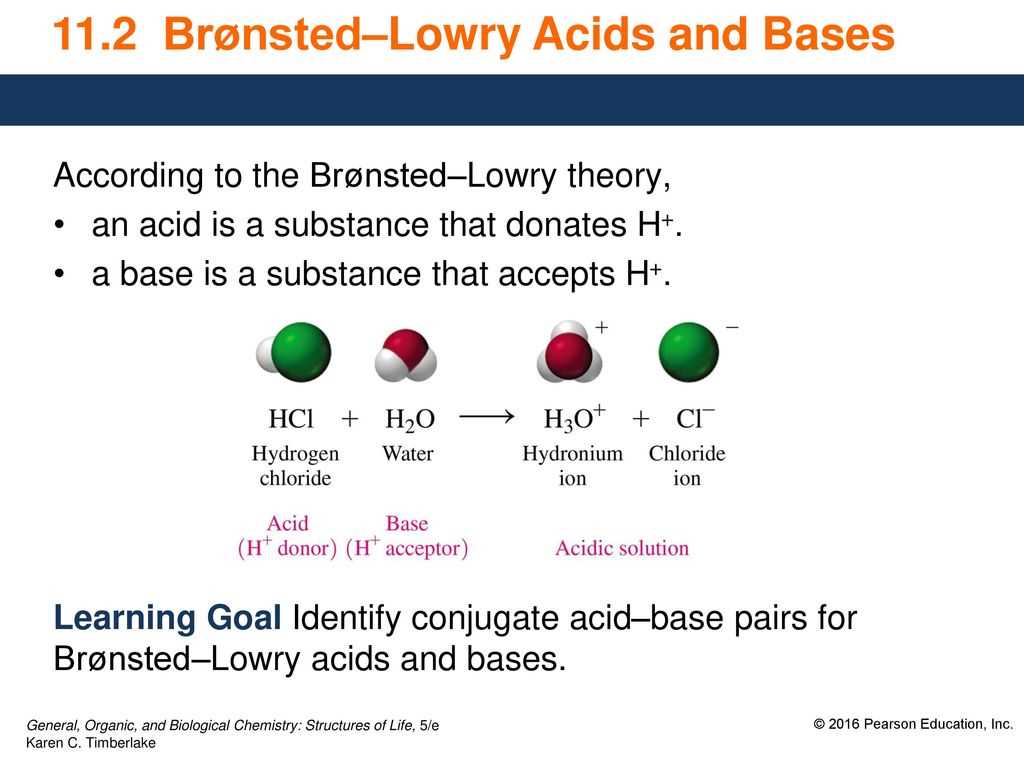
In the study of acids and bases, the Bronsted-Lowry theory is an important concept to understand. This theory defines an acid as a substance that donates a proton (H+) and a base as a substance that accepts a proton.
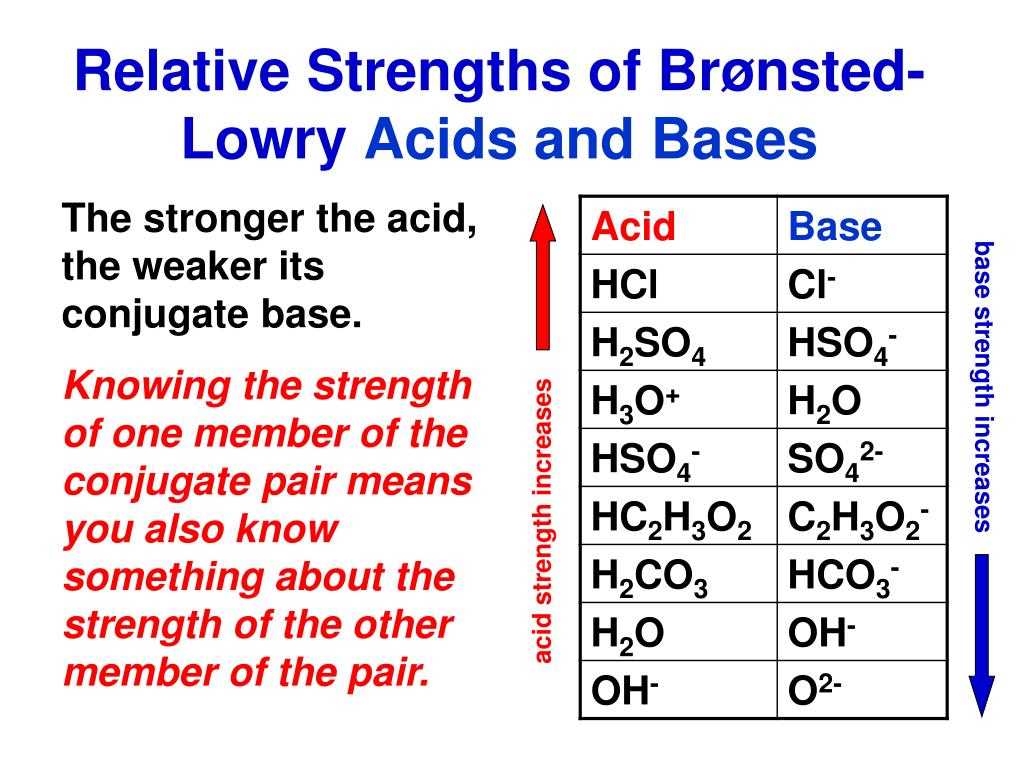
When working with Bronsted-Lowry acids and bases, it is crucial to be able to identify the acid and base in a chemical reaction. One way to do this is by examining the reactants and products of the reaction. For example, in the equation HCl + H2O -> H3O+ + Cl-, HCl is the acid because it donates a proton to H2O, which acts as the base by accepting the proton.
In a worksheet on Bronsted-Lowry acids and bases, you may be asked to identify the acid and base in a series of reactions or classify various substances as acids or bases. It is important to know the properties of acids and bases in order to correctly answer these questions.
Acids typically have a sour taste, can conduct electricity, and react with metals to produce hydrogen gas. Bases, on the other hand, usually have a bitter taste, feel slippery to the touch, and can conduct electricity. By understanding these properties, you can determine whether a given substance is an acid or a base.
When completing a Bronsted-Lowry acids and bases worksheet, it is helpful to have a list of common acids and bases, as well as their chemical formulas. This can assist you in correctly identifying the acid and base in a reaction and classifying substances. For example, hydrochloric acid (HCl) is a common acid, while sodium hydroxide (NaOH) is a common base.
Overall, understanding the Bronsted-Lowry theory and being able to identify acids and bases in chemical reactions is essential for success in studying acids and bases. By practicing with worksheet questions and knowing the properties of acids and bases, you can confidently answer questions on Bronsted-Lowry acids and bases worksheets.
What are Bronsted-Lowry Acids and Bases?

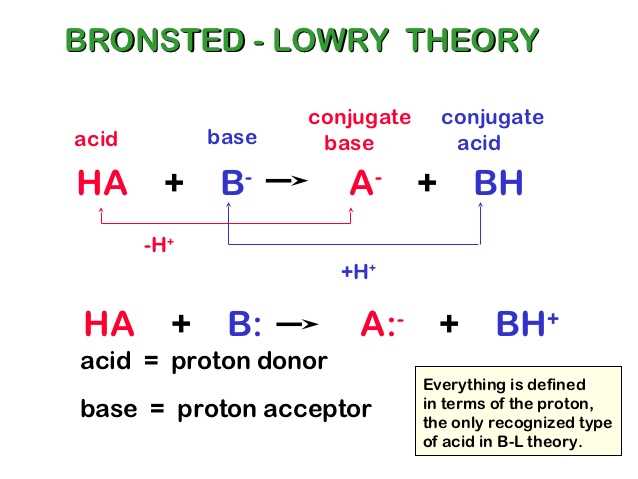
Bronsted-Lowry acids and bases are a fundamental concept in the field of chemistry. They are named after Johannes Nicolaus Bronsted and Thomas Martin Lowry, who independently proposed the theory of acids and bases in the early 20th century. According to the Bronsted-Lowry theory, an acid is a substance that donates a proton (H+) and a base is a substance that accepts a proton.
Unlike the more traditional Arrhenius theory, the Bronsted-Lowry theory does not require the presence of water as a solvent for acids and bases to react. It focuses on the transfer of protons between species in a chemical reaction. In this theory, an acid-base reaction involves the transfer of a proton from the acid to the base. The acid that loses a proton becomes its conjugate base, while the base that accepts a proton becomes its conjugate acid.
One key aspect of the Bronsted-Lowry theory is the concept of acid-base equilibria. The strength of an acid or base is determined by its ability to donate or accept protons. Strong acids completely dissociate in water, while weak acids only partially dissociate. Similarly, strong bases completely dissociate to form hydroxide ions (OH-), while weak bases do not completely dissociate.
The Bronsted-Lowry theory provides a crucial framework for understanding the behavior of acids and bases in various chemical reactions. It allows chemists to predict and explain the outcomes of acid-base reactions, as well as to design and optimize chemical processes in industries such as pharmaceuticals, agriculture, and environmental science.
How to Identify Bronsted-Lowry Acids and Bases?
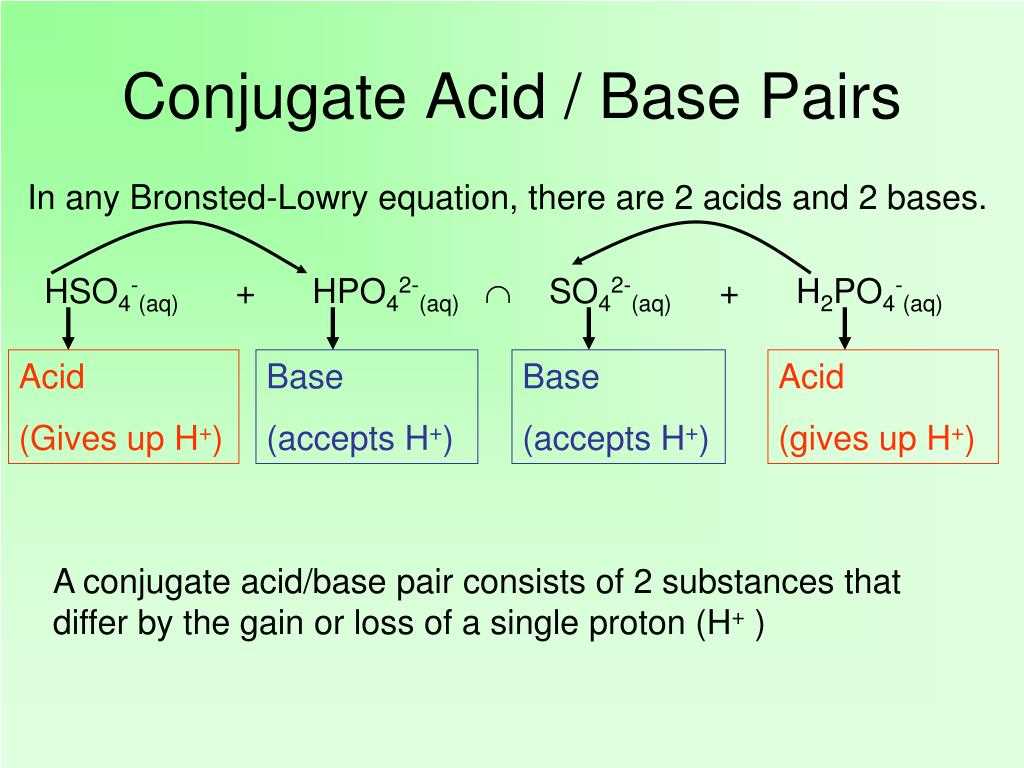
Identifying Bronsted-Lowry acids and bases requires an understanding of the theory behind this classification. According to the Bronsted-Lowry acid-base theory, an acid is a substance that can donate a proton (H+) to another substance, while a base is a substance that can accept a proton. To identify Bronsted-Lowry acids and bases, one can consider the reactions in which these substances are involved.
When a substance donates a proton, it is acting as an acid. For example, in the reaction: HCl + H2O -> H3O+ + Cl-, HCl is the acid because it donates a proton to water, which acts as the base by accepting the proton. Similarly, in the reaction: NH3 + H2O -> NH4+ + OH-, NH3 is the base as it accepts a proton from water, which acts as the acid.
Another way to identify Bronsted-Lowry acids and bases is by looking at their chemical formulas. Acids typically have a hydrogen atom at the beginning of their formula, followed by one or more other elements. Bases, on the other hand, often contain hydroxide (OH-) or amine (-NH2) groups. It is important to note that not all compounds with hydrogen are acids, and not all compounds with hydroxide or amine groups are bases. The classification depends on their ability to donate or accept protons.
To summarize, to identify Bronsted-Lowry acids and bases, one should consider their ability to donate or accept protons in a reaction. This can be determined by examining the reactions they are involved in or analyzing their chemical formulas for characteristic functional groups. Understanding this classification is essential in many areas of chemistry, as acids and bases play key roles in various chemical reactions and reactions.
Bronsted-Lowry Acids and Bases Worksheet Questions
Bronsted-Lowry theory defines acids as substances that donate protons (H+) and bases as substances that accept protons. This theory provides a more comprehensive understanding of acids and bases compared to the Arrhenius definition, which only focuses on the presence of H+ or OH- ions in solution.
The Bronsted-Lowry acid-base reaction involves the transfer of protons from one species to another. In this reaction, the acid donates a proton to the base, forming a conjugate acid-base pair. The acid that loses a proton becomes a conjugate base, while the base that gains a proton becomes a conjugate acid.
Worksheet Questions
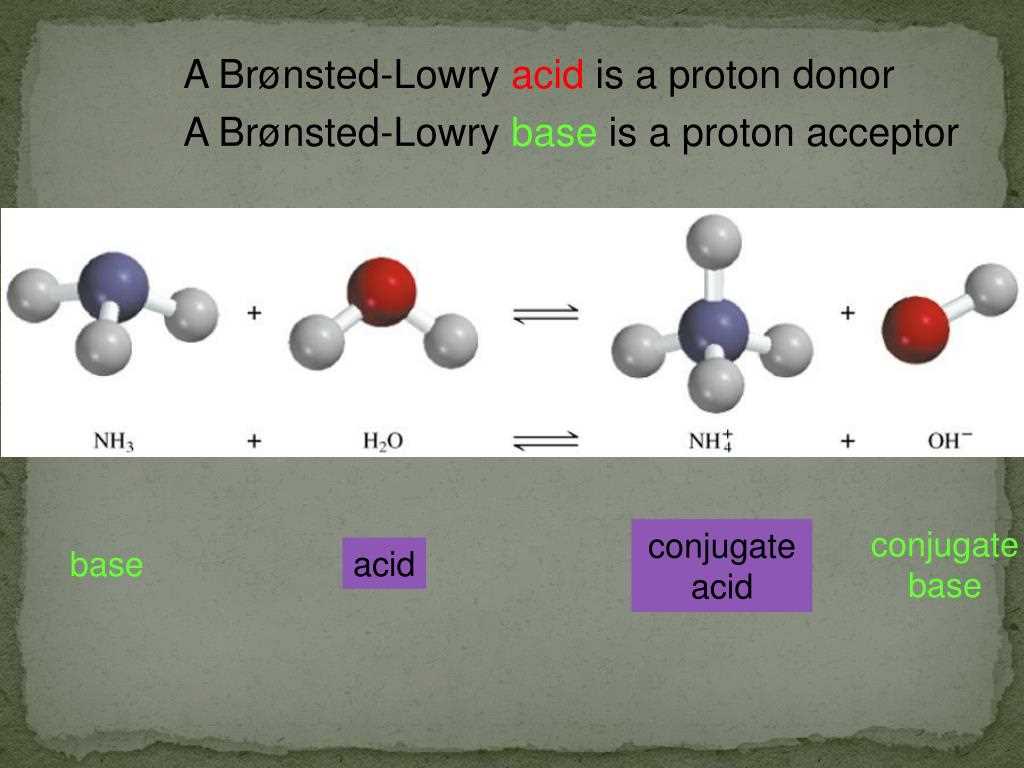
- Identify the acid, base, conjugate acid, and conjugate base in the following reaction: HCl + H2O → H3O+ + Cl-
- Classify the following substances as acids or bases according to the Bronsted-Lowry definition:
- NaOH
- CH3COOH
- NH3
- H2SO4
- Write the balanced equation for the following acid-base reaction: H2SO4 + NH3 → (NH4)2SO4
- Determine whether the following acid-base reactions are exothermic or endothermic:
- HCl + NaOH → NaCl + H2O
- NH3 + HCl → NH4Cl
- Explain the concept of amphoteric substances and provide an example.
These worksheet questions help students practice applying the Bronsted-Lowry theory to identify acids, bases, conjugate acid-base pairs, and balanced equations for acid-base reactions. They also encourage students to think about the energetics of acid-base reactions and the concept of amphoteric substances.
Worksheet Answers for Bronsted-Lowry Acids and Bases
Here are the answers to the worksheet on Bronsted-Lowry acids and bases.
Question 1: Identify the acids and bases in the following reactions:
- HCl + H2O → H3O+ + Cl-
- NH3 + H2O → NH4+ + OH-
- H2SO4 + OH- → HSO4- + H2O
Answer:
- In the reaction HCl + H2O → H3O+ + Cl-, HCl is the acid and H2O is the base.
- In the reaction NH3 + H2O → NH4+ + OH-, NH3 is the base and H2O is the acid.
- In the reaction H2SO4 + OH- → HSO4- + H2O, H2SO4 is the acid and OH- is the base.
Question 2: Write the balanced chemical equation for the following acid-base reactions:
- Hydrochloric acid reacts with sodium hydroxide to produce water and sodium chloride.
- Nitric acid reacts with ammonia to produce ammonium nitrate.
- Phosphoric acid reacts with calcium hydroxide to produce water and calcium phosphate.
Answer:
- HCl + NaOH → H2O + NaCl
- HNO3 + NH3 → NH4NO3
- H3PO4 + Ca(OH)2 → H2O + Ca3(PO4)2
Question 3: Classify each of the following substances as an acid or a base:
- Vinegar
- Baking soda
- Lemon juice
- Ammonia
Answer:
- Vinegar – Acid
- Baking soda – Base
- Lemon juice – Acid
- Ammonia – Base
I hope these answers help you understand the concepts of Bronsted-Lowry acids and bases more clearly. If you have any more questions, feel free to ask!
Explanation of Worksheet Answers
Below are the answers to the Bronsted-Lowry acids and bases worksheet:
- Question 1: The acid is HCl and the base is NH3.
- Question 2: The acid is HNO3 and the base is OH–.
- Question 3: The acid is H2SO4 and the base is H2O.
- Question 4: The acid is H3PO4 and the base is H2O.
- Question 5: The acid is HCOOH and the base is HCOO–.
By identifying the acids and bases in each equation, you can determine the products formed when they react. Acids donate protons (H+ ions) to bases, forming conjugate bases and conjugate acids.
Understanding the concept of Bronsted-Lowry acids and bases is crucial in many areas of chemistry, including acid-base reactions, equilibrium, and pH calculations. This worksheet provides practice in identifying acids and bases in various chemical equations.