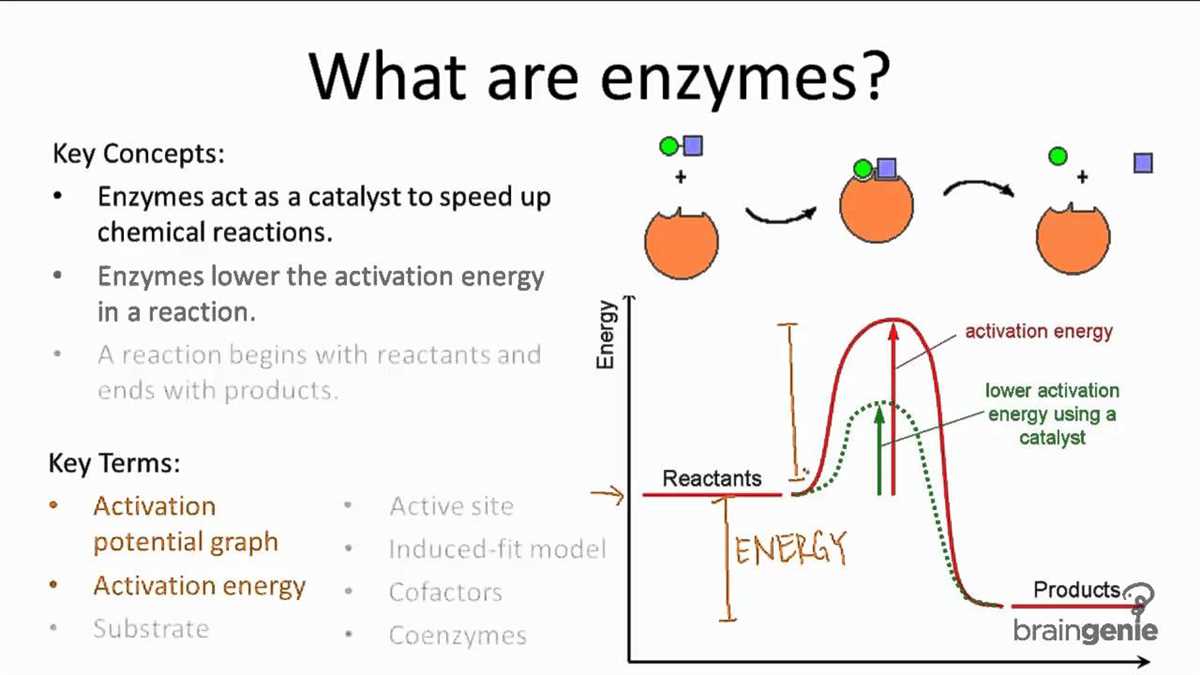
In Chapter 2 Lesson 4 of our chemistry course, we will be exploring the fascinating world of chemical reactions and enzymes. Chemical reactions are the processes by which substances are transformed into new substances. This can involve the rearrangement of atoms, the breaking and forming of bonds, and the release or absorption of energy. Enzymes, on the other hand, are specialized proteins that act as catalysts, speeding up chemical reactions without being consumed in the process.
Chemical reactions are essential for life as they occur inside living organisms constantly. From the digestion of food to the production of energy, or even the synthesis of DNA, all these processes involve chemical reactions. Understanding how chemical reactions work and the factors that influence them is crucial in the field of biochemistry.
Enzymes, as mentioned earlier, play a vital role in catalyzing chemical reactions. They are incredibly efficient and specific, allowing reactions to occur at a significantly faster rate. Enzymes accomplish this by lowering the activation energy required for a reaction to proceed, making it easier for molecules to reach the transition state and form new products.
In this lesson, we will study the different types of chemical reactions, such as synthesis, decomposition, single and double displacement, and combustion. We will also delve into the factors that affect reaction rates, including temperature, concentration, surface area, and the presence of catalysts. By the end of this lesson, you will have a comprehensive understanding of chemical reactions and enzymes, and be able to explain their significance in various biological processes.
Chapter 2 Lesson 4 Chemical Reactions and Enzymes Answer Key
In Chapter 2 Lesson 4, we discussed chemical reactions and enzymes. A chemical reaction is a process in which substances undergo chemical changes to form different substances. This process involves breaking and forming chemical bonds. Chemical reactions can be classified into different types, such as synthesis reactions, decomposition reactions, and exchange reactions.
Enzymes play a crucial role in chemical reactions. They are biological molecules that act as catalysts, increasing the rate of chemical reactions without being consumed or permanently altered. Enzymes lower the activation energy required for a reaction to occur, allowing it to happen more quickly. Enzymes are highly specific and can only catalyze specific reactions due to their unique structure.
The answer key for Chapter 2 Lesson 4 includes information about important concepts and terms related to chemical reactions and enzymes. It provides the correct answers to questions and exercises given in the lesson. The answer key helps students check their understanding and accuracy in solving problems related to chemical reactions and enzymes.
- Synthesis reactions: A type of chemical reaction where two or more substances combine to form a more complex substance.
- Decomposition reactions: A type of chemical reaction where a complex substance breaks down into two or more simpler substances.
- Exchange reactions: A type of chemical reaction where atoms or groups of atoms are exchanged between different molecules.
- Enzymes: Biological catalysts that speed up chemical reactions by lowering the activation energy required for the reaction to occur.
- Activation energy: The energy required for a chemical reaction to start.
Understanding chemical reactions and enzymes is essential in various scientific fields, including biology, chemistry, and biochemistry. By studying these concepts, scientists can better understand the complex processes that occur in living organisms and apply this knowledge to various industries, such as pharmaceuticals, agriculture, and environmental science.
Understanding Chemical Reactions
Chemical reactions are fundamental processes in chemistry that involve the transformation of one or more substances into different substances. These reactions occur when there is a rearrangement of atoms in molecules, resulting in the formation of new compounds or the breaking apart of existing compounds. Understanding chemical reactions is crucial in various fields, including medicine, environmental science, and industrial processes.
Chemical reactions are represented by chemical equations, which describe the reactants and products involved. Reactants are the starting materials that undergo the reaction, and products are the end result of the reaction. These equations also provide information about the stoichiometry of the reaction, including the relative amounts of each reactant and product.
Types of Chemical Reactions
There are several different types of chemical reactions that can occur. These include precipitation reactions, acid-base reactions, redox reactions, and combustion reactions. In a precipitation reaction, two aqueous solutions are combined, resulting in the formation of an insoluble solid called a precipitate. Acid-base reactions occur when an acid and a base react to form a salt and water. Redox reactions involve the transfer of electrons between reactants, leading to the formation of oxidized and reduced species. Combustion reactions involve the rapid combination of oxygen with a fuel, releasing energy in the form of heat and light.
Factors Affecting Chemical Reactions
Several factors can affect the rate and outcome of a chemical reaction. These include temperature, concentration, surface area, and the presence of catalysts. Increasing the temperature generally increases the rate of reaction, as it provides more thermal energy for molecules to collide and react. Higher concentration of reactants also leads to an increased rate of reaction due to more frequent collisions. Increased surface area can also speed up chemical reactions, as it provides more contact between reactants. Catalysts are substances that increase the rate of reaction without being consumed in the process. They achieve this by providing an alternative reaction pathway with lower activation energy.
Understanding chemical reactions is essential for scientists and engineers to develop new materials, understand biological processes, and optimize industrial processes. By studying the factors that influence reactions, it is possible to control and manipulate chemical reactions for desired outcomes, such as the production of specific products or the elimination of pollutants. Chemical reactions are at the heart of many important scientific and technological advancements, and continued research in this field is vital for further progress.
Different Types of Chemical Reactions
Chemical reactions can be classified into several different types based on the changes that occur during the reaction. These types include synthesis reactions, decomposition reactions, single displacement reactions, double displacement reactions, and combustion reactions.
Synthesis reactions are reactions in which two or more reactants combine to form a single product. This type of reaction is often represented by the general equation: A + B → AB. Synthesis reactions are commonly seen in nature, such as the formation of water through the combination of hydrogen and oxygen.
Decomposition reactions are the opposite of synthesis reactions, where a single reactant breaks down into two or more products. The general equation for a decomposition reaction is: AB → A + B. An example of a decomposition reaction is the breakdown of hydrogen peroxide into water and oxygen gas.
Single displacement reactions, also known as substitution reactions, involve the exchange of one element with another in a compound. The general equation for a single displacement reaction is: A + BC → AC + B. An example of a single displacement reaction is the reaction between zinc and hydrochloric acid, where zinc displaces hydrogen from the acid.
Double displacement reactions occur when there is an exchange of ions between two compounds. The general equation for a double displacement reaction is: AB + CD → AD + CB. A common example of a double displacement reaction is the reaction between sodium chloride and silver nitrate, which forms sodium nitrate and silver chloride.
Combustion reactions, also known as burning reactions, involve the rapid combination of a substance with oxygen, producing heat and often light. The general equation for a combustion reaction is: fuel + oxygen → carbon dioxide + water. An example of a combustion reaction is the burning of gasoline in a car engine, where the fuel (gasoline) combines with oxygen in the air to produce carbon dioxide and water vapor.
Overall, understanding the different types of chemical reactions is crucial in understanding how substances interact and transform, and it allows scientists to predict the products of a reaction based on its type.
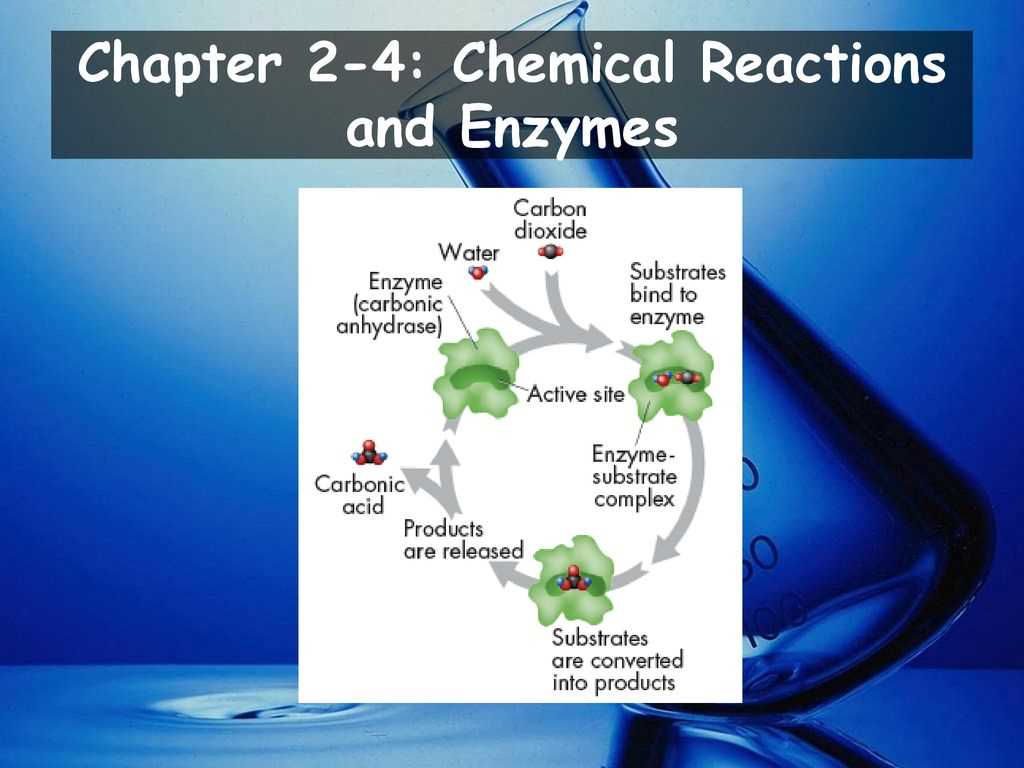
Factors Affecting Chemical Reactions
Chemical reactions are influenced by various factors that can either speed up or slow down the reaction rate. These factors include:
- Temperature: Increasing the temperature generally increases the rate of a chemical reaction. This is because higher temperatures provide more energy to the reacting particles, increasing their collision frequency and energy. As a result, more successful collisions occur, leading to a faster reaction rate. However, extremely high temperatures can also denature enzymes and change the nature of the reaction.
- Concentration: Higher concentration of reactants tends to increase the reaction rate. This is because the chances of reactant particles colliding with each other are higher when their concentration is higher. Consequently, the frequency of successful collisions increases, leading to a faster reaction.
- Surface Area: Increasing the surface area of solid reactants can accelerate the reaction rate. This is because exposing more surface area to the reactants allows for a greater number of collisions to occur. For example, a powdered substance will react more quickly than a solid block of the same substance due to its increased surface area.
- Catalysts: Catalysts are substances that can speed up a chemical reaction without being consumed in the process. They work by lowering the activation energy required for the reaction to occur. By providing an alternative reaction pathway with a lower energy barrier, catalysts increase the rate of reaction. Enzymes are biological catalysts that play a crucial role in many biochemical reactions.
- Presence of Light: Some reactions are sensitive to the presence of light. In these cases, light can either accelerate or inhibit the reaction rate. This phenomenon is known as photochemical reactions.
Understanding these factors and how they influence chemical reactions is important for various fields, including industrial processes, environmental sciences, and biochemistry. By manipulating these factors, scientists can optimize reaction conditions to achieve desired reaction rates and outcomes.
The Role of Enzymes in Chemical Reactions
Enzymes play a crucial role in chemical reactions by acting as catalysts. A catalyst is a substance that speeds up a reaction by lowering the activation energy required for the reaction to occur. In other words, enzymes help to facilitate chemical reactions by making them happen faster and more efficiently.
Enzymes are proteins with specific shapes and active sites. The active site is where the enzyme binds to the substrate, which is the molecule that the enzyme acts upon. The binding of the enzyme to the substrate forms an enzyme-substrate complex. This complex allows the enzyme to catalyze the conversion of the substrate into a product. Once the reaction is complete, the product is released and the enzyme returns to its original state, ready to catalyze another reaction.
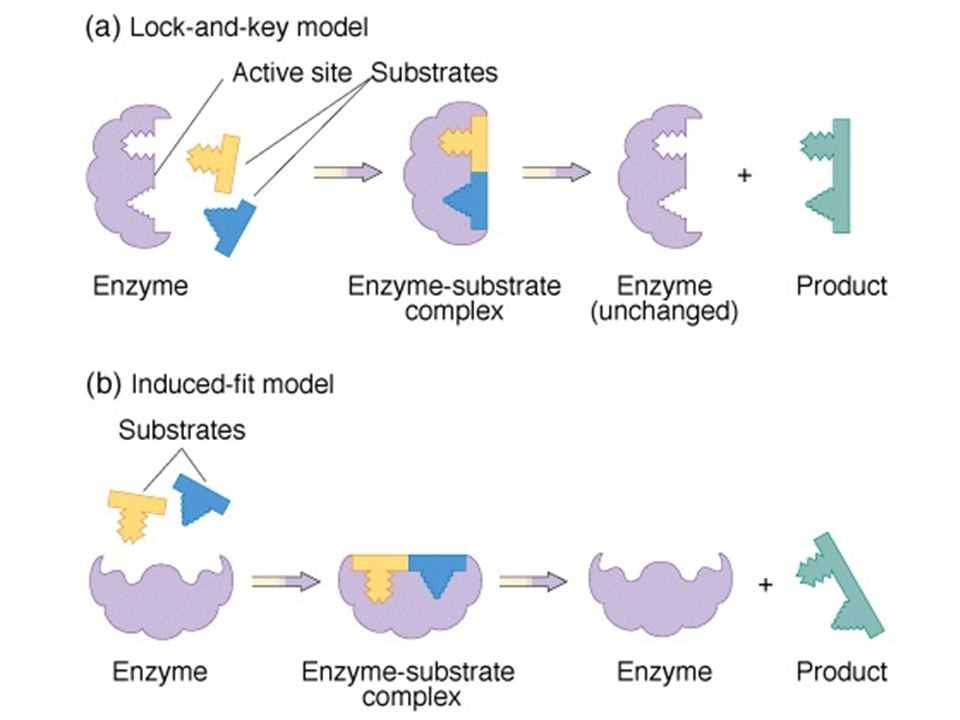
Enzymes are highly specific and can usually only catalyze one type of reaction. This specificity is due to the unique shape of the enzyme’s active site, which only fits certain substrates. The enzyme-substrate interaction is often compared to a lock and key, where the enzyme is the lock and the substrate is the key. The lock can only be opened by the correct key, and similarly, the enzyme can only bind to the appropriate substrate.
Enzymes are essential for life as they regulate the metabolic processes in cells. Without enzymes, chemical reactions would occur too slowly to sustain life. Enzymes greatly increase the rate of reactions, allowing them to occur at a viable pace. They also ensure that reactions happen under specific conditions and prevent unwanted side reactions. Overall, enzymes are a crucial component of biological systems, enabling the complex chemistry of life to take place.
Characteristics of Enzymes
Enzymes are biological catalysts that play a crucial role in various biochemical reactions within living organisms. They are protein molecules that have specific functional groups that allow them to bind to substrates and facilitate the conversion of these substrates into products. Enzymes are highly specific in their actions, meaning that each enzyme can only catalyze a particular reaction or group of reactions. This specificity is due to the unique arrangement of amino acids in the enzyme’s active site, which is the region where the substrate binds and the reaction occurs.
Enzymes have several key characteristics that make them essential for living systems. Firstly, enzymes can greatly accelerate the rate of chemical reactions, often by millions or billions of times. This acceleration is achieved by reducing the activation energy required for the reaction to occur. By lowering the energy barrier, enzymes allow reactions to happen more readily, enabling the efficient metabolism of nutrients and the synthesis of essential molecules.
Another important characteristic of enzymes is their sensitivity to environmental conditions. Factors such as temperature, pH, and the presence of certain ions can affect the activity of enzymes. Each enzyme has an optimal range of conditions in which it functions most efficiently. Outside of this range, the enzyme’s activity may be reduced or completely inhibited. This sensitivity to environmental conditions allows organisms to regulate enzymatic activity and adapt to different physiological conditions.
Enzymes also exhibit the property of being reusable. Once an enzyme has catalyzed a reaction, it is not consumed or altered in the process. Instead, the enzyme can dissociate from the products and bind to another substrate, starting the catalytic cycle anew. This property allows enzymes to carry out multiple reactions without being depleted, making them highly efficient catalysts.
In summary, enzymes are protein molecules that have unique functional groups and active sites, allowing them to catalyze specific reactions. They greatly accelerate reaction rates, are sensitive to environmental conditions, and can be reused multiple times. These characteristics make enzymes essential for the efficient functioning and regulation of biochemical processes within living organisms.
Enzyme Activity and Regulation
The activity of enzymes is crucial for the proper functioning of biological systems. Enzymes catalyze chemical reactions, increasing the rate at which they occur. They play a vital role in various physiological processes, such as digestion, metabolism, and DNA replication.
Enzyme activity can be regulated in multiple ways to ensure that the right reactions occur at the right time and in the right place. One important regulatory mechanism is allosteric regulation, where molecules bind to specific sites on an enzyme, altering its activity. This allosteric regulation can either activate or inhibit the enzyme, depending on the nature of the molecules involved.
Another method of regulating enzyme activity is through the process of phosphorylation. Phosphorylation involves the addition of a phosphate group to an enzyme, which can either activate or deactivate it. This process is often regulated by protein kinases and phosphatases, which add or remove phosphate groups, respectively.
Enzyme activity is also influenced by environmental factors, such as pH and temperature. Each enzyme has an optimal pH at which it functions most effectively. Deviations from this optimal pH can impair enzyme activity. Similarly, temperature affects enzyme activity, with most enzymes having an optimal temperature at which they work best.
In conclusion, the activity of enzymes is essential for the proper functioning of biological systems. Regulation of enzyme activity through allosteric regulation, phosphorylation, and environmental factors ensures that the right reactions occur when and where they are needed. Understanding enzyme activity and regulation is crucial for understanding biochemical processes and developing potential therapeutic interventions.