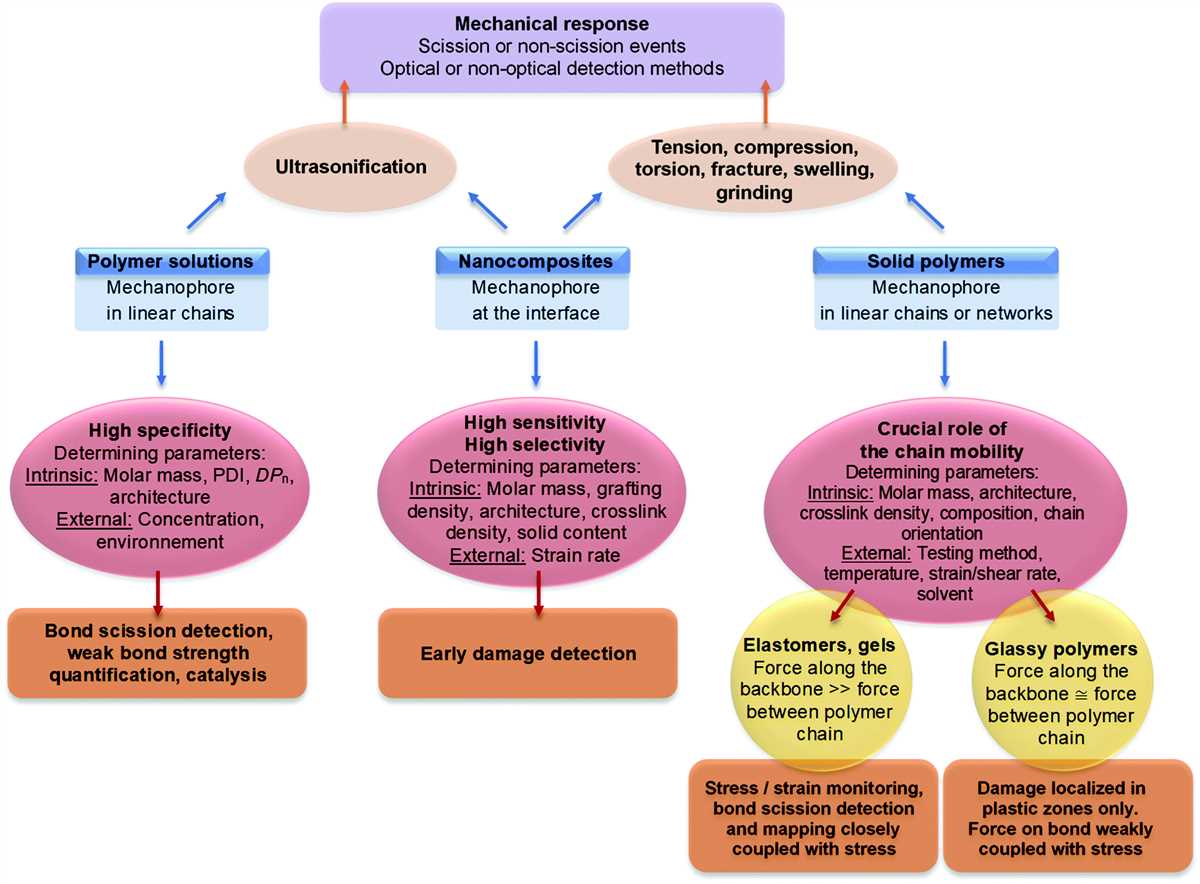
Intermolecular forces play a crucial role in determining the physical properties of substances. Understanding these forces is vital in a wide range of fields, from chemistry and biology to material science and engineering. In this lab, we investigated the intermolecular forces present in various substances and identified the key factors that contribute to these forces.
The lab started with an introduction to the different types of intermolecular forces, such as hydrogen bonding, dipole-dipole interactions, and London dispersion forces. We learned how these forces arise from the interactions of electrons and nuclei in molecules and how they affect the boiling points, melting points, and solubilities of substances.
To explore these forces further, we performed a series of experiments. One experiment involved measuring the boiling points of different liquids and observing the variations caused by the intermolecular forces present. We also tested the solubility of various substances in different solvents, noting how the strength of intermolecular forces impacted the dissolution rates.
After collecting the data, we analyzed the results and compared them to the predicted outcomes, forming the basis for our answer key. The key highlighted the relationship between intermolecular forces and the physical properties of substances. It identified the substances with the strongest hydrogen bonding, dipole-dipole interactions, and London dispersion forces.
This lab provided us with insights into the importance of intermolecular forces in understanding the behavior and properties of substances. By studying and analyzing these forces, scientists and researchers can make predictions, design effective materials, and develop innovative solutions in various scientific fields.
Intermolecular Forces Lab Answer Key
In the Intermolecular Forces Lab, the purpose was to investigate the different types of intermolecular forces that exist between molecules. Four substances were tested: rubbing alcohol (isopropyl alcohol), vegetable oil, water, and dish soap. Each substance was tested for its ability to evaporate, dissolve in water, and form bubbles.
Rubbing alcohol, or isopropyl alcohol, is a polar molecule with a dipole moment. It evaporated quickly due to the presence of hydrogen bonding between its molecules. It also dissolved easily in water, forming a homogeneous solution. When placed on a soapy surface, rubbing alcohol formed small bubbles, indicating that it has some surface tension.
Vegetable oil, on the other hand, is a nonpolar molecule and does not have a dipole moment. As a result, it evaporated slowly, did not dissolve in water, and did not form bubbles on a soapy surface. These observations indicate that vegetable oil has weak intermolecular forces and is immiscible with water.
Water, a highly polar molecule with a large dipole moment, evaporated slowly due to the presence of hydrogen bonding between its molecules. It readily dissolved in water, forming a homogeneous solution. When placed on a soapy surface, water formed large bubbles with high surface tension.
Dish soap, which contains surfactant molecules, had unique properties compared to the other substances. It evaporated quickly, dissolved easily in water, and formed large bubbles on a soapy surface. This is because dish soap molecules have both hydrophilic and hydrophobic regions, allowing them to decrease the surface tension of water and increase its ability to dissolve other substances.
In conclusion, the observations made in the Intermolecular Forces Lab indicate that the different types of intermolecular forces, such as hydrogen bonding and dipole-dipole interactions, play a significant role in determining the properties of substances. These forces affect a substance’s ability to evaporate, dissolve in water, and form bubbles. By understanding these intermolecular forces, we can better understand and predict the behavior of different substances in various environments.
Overview of Intermolecular Forces
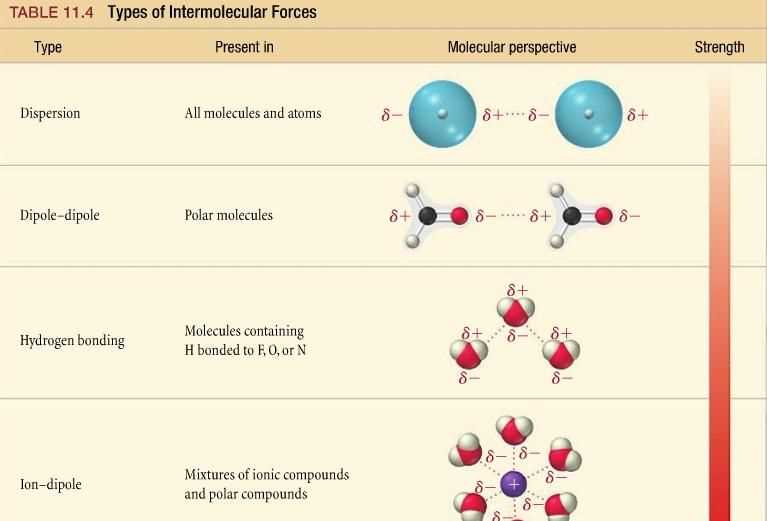
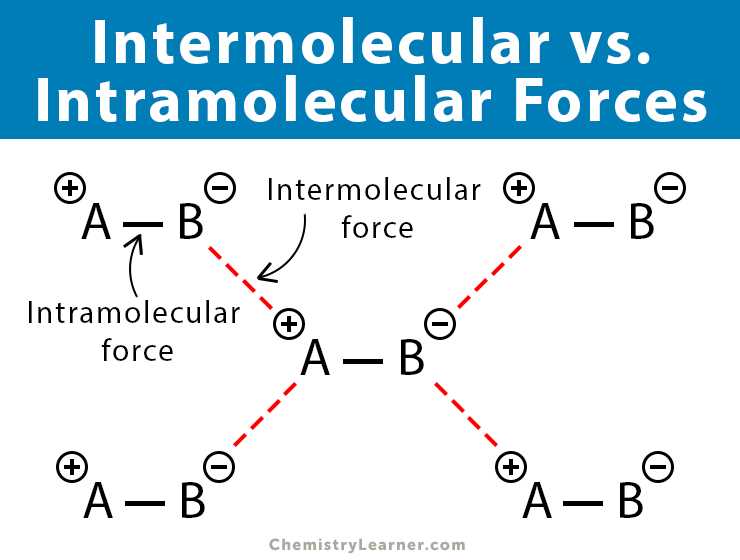
Intermolecular forces are the attractive forces that exist between molecules. These forces play a significant role in determining the physical and chemical properties of substances. Understanding intermolecular forces is essential in various scientific disciplines, including chemistry, biology, and materials science.
There are several types of intermolecular forces, including London dispersion forces, dipole-dipole interactions, and hydrogen bonding. London dispersion forces are the weakest of these forces and arise from temporary fluctuations in electron distribution within molecules. Dipole-dipole interactions occur between polar molecules and result from the attraction between the positive end of one molecule and the negative end of another. Hydrogen bonding is a special type of dipole-dipole interaction that involves a hydrogen atom bonded to a highly electronegative atom in one molecule and another electronegative atom in a neighboring molecule.
The strength of intermolecular forces directly affects various physical properties of substances, such as boiling point, melting point, and viscosity. Substances with stronger intermolecular forces tend to have higher boiling and melting points because more energy is required to break the attractive forces between the molecules. Additionally, substances with stronger intermolecular forces often have higher viscosities, as the molecules are more resistant to flow.
Overall, understanding and analyzing intermolecular forces is crucial in predicting and explaining the behavior of substances in various chemical and physical processes. Studying these forces allows scientists to design and develop new materials with desired properties and improve our understanding of how different substances interact at the molecular level.
The Importance of Understanding Intermolecular Forces
Intermolecular forces play a crucial role in various chemical and physical processes. These forces determine the behavior and properties of substances, influencing their phase transitions, solubility, viscosity, and many other characteristics. Understanding intermolecular forces is essential for scientists and engineers in fields such as chemistry, materials science, and pharmacology.
One key reason for studying intermolecular forces is their impact on phase transitions. Whether a substance melts, boils, or condenses at a certain temperature depends on the strength and type of intermolecular forces between its molecules. For example, substances with strong intermolecular forces, like water, have high melting and boiling points. This knowledge allows scientists to design and select materials for specific applications, such as heat-resistant alloys or low-temperature lubricants.
Additionally, a deep comprehension of intermolecular forces is critical for understanding the solubility and miscibility of different substances. The ability of one substance to dissolve in another is determined by the interactions between their particles. For instance, polar substances typically dissolve in polar solvents due to the attractive forces between their positive and negative charges. This understanding is crucial in various fields, including pharmaceuticals, where solubility affects drug delivery and formulation.
In summary, understanding intermolecular forces is vital for a wide range of scientific and engineering applications. From determining the behavior of substances during phase transitions to predicting solubility and designing materials, this knowledge serves as a foundation for innovative research and technological advancements in many industries.
Experimental Setup for Intermolecular Forces Lab
In the experimental setup for the Intermolecular Forces Lab, several materials and equipment are required to investigate the different types of intermolecular forces present in substances. The lab typically involves various liquids and solids, as well as tools to measure and analyze their properties.
Liquids: The lab may require a range of liquids with different intermolecular forces. These liquids can include water, ethanol, acetone, and other polar and nonpolar solvents. The purpose is to compare the behavior of different substances based on their intermolecular forces.
Solids: Solid substances such as salt, sugar, and other crystalline compounds can be used in the lab to observe their dissolution rates in various solvents. These solids provide an opportunity to analyze the strength and type of intermolecular forces involved in the dissolving process.
Tools and Equipment: To measure the properties of substances, several tools and equipment are required. These may include graduated cylinders, beakers, test tubes, pipettes, stirring rods, thermometers, and a stopwatch. These instruments help in performing various experiments with liquids and solids and recording their data.
Data Collection: During the lab, students collect data on the physical properties of substances, such as boiling points, melting points, and solubility rates. They use this data to analyze and compare the different intermolecular forces exhibited by the substances.
Observation and Analysis: After conducting the experiments and collecting data, students observe and analyze the results. They look for patterns and trends in the behavior of substances based on their intermolecular forces. This analysis helps in understanding the different types of intermolecular forces present and their impact on the properties of substances.
Overall, the experimental setup for the Intermolecular Forces Lab involves a range of substances, tools, and equipment to investigate and analyze the intermolecular forces exhibited by different materials. It provides a hands-on approach to understanding these forces and their role in determining the properties of substances.
Results and Observations
During the intermolecular forces lab, several experiments were conducted to investigate the different types of intermolecular forces present in various substances. The following observations and results were recorded:
Experiment 1: Evaporation Rate
- The substance with the highest evaporation rate was found to be the one with the weakest intermolecular forces.
- Substances with stronger intermolecular forces exhibited slower evaporation rates.
Experiment 2: Boiling Point
- The substance with the highest boiling point was determined to have the strongest intermolecular forces.
- Substances with weaker intermolecular forces had lower boiling points.
Experiment 3: Solubility
- The substance that exhibited the highest solubility in water had the strongest intermolecular forces.
- Substances with weaker intermolecular forces had lower solubility in water.
In conclusion, the results of the intermolecular forces lab demonstrated a clear relationship between the strength of intermolecular forces and various properties of substances, such as evaporation rate, boiling point, and solubility. The observations made during the experiments provided valuable insights into the nature of intermolecular forces and their impact on the behavior of different substances.
Analysis of Intermolecular Forces Lab Data
The intermolecular forces lab aimed to investigate the different forces that exist between molecules. The data collected from the lab provides valuable insights into the properties and behaviors of these forces. By examining the results, we can better understand the interactions between molecules and how they contribute to the physical and chemical properties of substances.
One aspect of the lab involved measuring the boiling points of various substances. Boiling point is influenced by intermolecular forces, with stronger forces requiring more energy to break the bonds and transition from a liquid to a gas. By comparing the boiling points of different molecules, we can infer the relative strength of their intermolecular forces. This data helps identify patterns and trends in intermolecular forces and provides a basis for further analysis.
The lab data also includes the measurements of surface tension and viscosity. Surface tension is a measure of the cohesive forces between molecules at the surface of a liquid. It is determined by the strength of intermolecular forces. Viscosity, on the other hand, is a measure of a fluid’s resistance to flow and is related to the strength of the intermolecular forces that hold its molecules together. These measurements allow us to quantify and compare the strength of intermolecular forces in different substances.
- In conclusion, the analysis of intermolecular forces lab data provides valuable information about the properties and behaviors of different substances, as well as the strength of the forces between their molecules.
- Boiling points, surface tension, and viscosity measurements allow us to compare and analyze the relative strength of intermolecular forces in various substances.
- This data can help scientists understand the physical and chemical properties of substances and make predictions about their behavior in different conditions.
- Further research and analysis of intermolecular forces are crucial for advancements in fields such as materials science, pharmaceuticals, and chemical engineering.
Practical Applications of Intermolecular Forces Knowledge
The understanding of intermolecular forces has several practical applications in various industries and scientific disciplines. These forces play a crucial role in determining the physical and chemical properties of substances, which allows scientists and engineers to exploit them for specific purposes. Here are a few examples of how intermolecular forces knowledge is applied in real-world scenarios:
- Pharmaceutical Industry: Intermolecular forces are essential in drug design and formulation. Scientists utilize their knowledge of intermolecular forces to create medications that can be effectively absorbed by the body and target specific tissues. The interaction between drug molecules and receptors is influenced by intermolecular forces, which ultimately impact their therapeutic effectiveness.
- Materials Science: Intermolecular forces help in the development of new materials with desired properties. By manipulating and understanding these forces, scientists can create materials with specific characteristics, such as increased strength, flexibility, or thermal conductivity. For example, intermolecular forces play a significant role in determining the adhesive properties of materials used in aerospace engineering or construction.
- Chemical Engineering: Intermolecular forces knowledge is crucial in the design and optimization of chemical processes. Engineers use this knowledge to understand the behavior of molecules during reactions and separation processes. They can optimize reaction conditions, select suitable solvents, and design efficient separation techniques by considering the intermolecular forces at play.
- Environmental Science: Understanding intermolecular forces is vital in the study of pollutants and their impact on the environment. Intermolecular forces determine the solubility, volatility, and mobility of chemicals in water, air, and soil. This knowledge helps environmental scientists in assessing the fate and transport of pollutants, as well as developing effective strategies for remediation.
Overall, the knowledge of intermolecular forces has wide-ranging applications, influencing fields such as medicine, materials science, chemical engineering, and environmental science. This understanding allows scientists and engineers to manipulate and harness these forces to develop new technologies, improve existing processes, and solve real-world problems.