
Welcome to the answer key for the Spectroscopy Lab. In this lab, you were introduced to the world of spectroscopy, a powerful technique used to study and analyze the interaction of matter with electromagnetic radiation. By using different types of spectroscopic instruments, you were able to examine the absorption and emission spectra of various substances. Now, it’s time to review your findings and understand the key concepts behind spectroscopy.
1. Absorption Spectra: In spectroscopy, absorption spectra are obtained by measuring the amount of light absorbed by a substance at different wavelengths. During the lab, you observed absorption spectra for a variety of colored solutions. The key takeaway from this part of the lab is that each substance has a unique absorption spectrum, which can be used to identify unknown substances.
2. Emission Spectra: Emission spectra are obtained by measuring the light emitted by a substance when it transitions from a higher energy state to a lower energy state. During the lab, you observed the emission spectra of different gases. The key concept to remember here is that each element has a unique emission spectrum, allowing scientists to identify the presence of specific elements in a sample.
Spectroscopy Lab Answer Key: Unlocking the Secrets of Light
The field of spectroscopy is a powerful tool that allows scientists to analyze and understand the properties of light. In this lab, we explored the fundamentals of spectroscopy and learned how to interpret spectra to extract valuable information.
Spectroscopy involves the study of the interaction between light and matter. By breaking down light into its component wavelengths, we can identify the unique patterns and signatures that different elements and compounds produce. This information can then be used to determine the composition, structure, and behavior of various substances.
The key to unlocking the secrets of light lies in the interpretation of spectra. During the lab, we were provided with a spectrogram, which is a visual representation of the spectra obtained from different light sources. By closely examining the spectrogram, we were able to identify the presence of specific elements or compounds by matching their known spectral lines.
For example, we observed a set of sharp, discrete lines in the spectrogram, corresponding to the hydrogen Balmer series. This indicated the presence of hydrogen gas in the light source. Similarly, by analyzing the position and intensity of the spectral lines, we were able to infer the temperature and composition of stars, as well as detect the presence of certain pollutants in environmental samples.
The spectroscopy lab provided us with a hands-on experience in unraveling the mysteries of light.
To enhance our understanding, we also learned about the different types of spectroscopy techniques, such as absorbance spectroscopy and emission spectroscopy, and their applications in various scientific fields. These techniques have revolutionized the way we analyze materials, diagnose diseases, and explore the universe.
In conclusion, the spectroscopy lab was an enlightening experience that demonstrated the power of light analysis. By unlocking the secrets of light through spectroscopy, scientists can gain valuable insights into the composition, structure, and behavior of matter. Whether it is studying distant stars or analyzing everyday substances, spectroscopy plays a crucial role in advancing our understanding of the world around us.
Understanding Spectroscopy: A Brief Overview
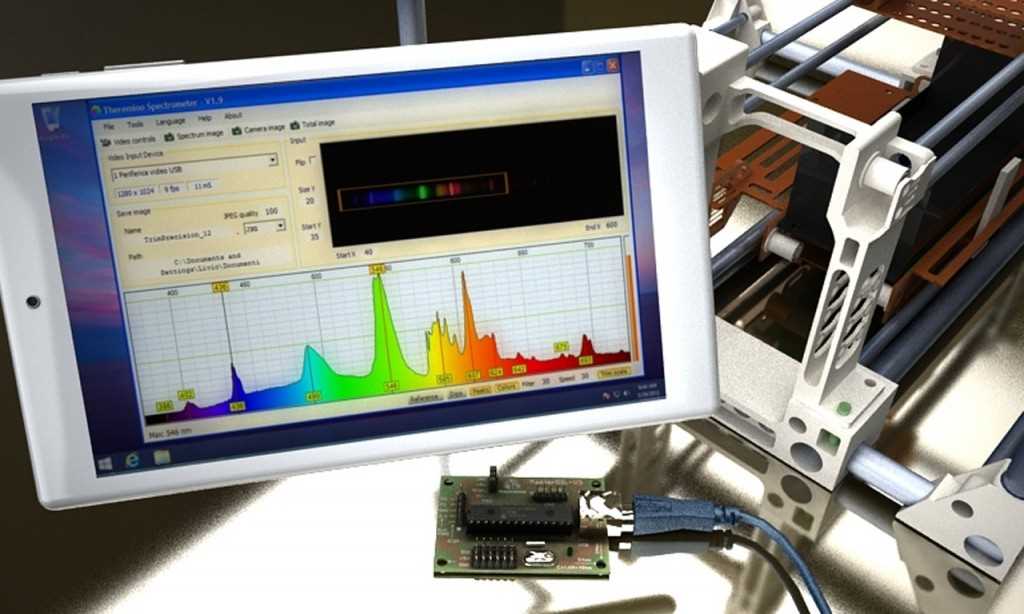
Spectroscopy is a powerful scientific technique used to study the interaction between matter and electromagnetic radiation, such as light. By analyzing the way that different materials absorb, emit, or scatter light, spectroscopy provides valuable information about their composition, structure, and properties. Spectroscopy is widely used in various fields, including chemistry, physics, biology, and astronomy.
One of the key concepts in spectroscopy is the spectrum, which is the distribution of electromagnetic radiation as a function of its wavelength or frequency. The spectrum can be separated into different regions, such as ultraviolet, visible, and infrared, depending on the range of wavelengths or frequencies being studied. Each region of the spectrum provides unique information about the interaction between matter and electromagnetic radiation.
Spectroscopy can be performed using different techniques, such as absorption spectroscopy, emission spectroscopy, and scattering spectroscopy. In absorption spectroscopy, the sample absorbs specific wavelengths of light, which results in the appearance of dark absorption lines or bands in the spectrum. By analyzing these absorption lines or bands, scientists can determine the presence of specific compounds or elements in the sample. In emission spectroscopy, the sample emits light at specific wavelengths, which results in the appearance of bright emission lines or bands in the spectrum. This technique is used to study the energy levels and electron transitions in atoms, molecules, and other particles. In scattering spectroscopy, the sample scatters light in different directions, leading to changes in the intensity and polarization of the incident light. Scattering spectroscopy is used to study the size, shape, and refractive index of particles in suspensions or solutions.
Overall, spectroscopy is a versatile and powerful tool that allows scientists to gain insights into the properties and behavior of various materials. By understanding the principles and techniques of spectroscopy, scientists can uncover new knowledge and make significant advancements in numerous scientific disciplines.
The Role of Spectroscopy in Scientific Research
Spectroscopy plays a crucial role in scientific research as it provides valuable information about the interaction of matter with electromagnetic radiation. It is a powerful tool used by scientists across various disciplines to study the composition, structure, and properties of different materials.
One of the key applications of spectroscopy is in the field of chemistry, where it is used to identify and analyze the chemical composition of substances. Spectroscopic techniques such as infrared spectroscopy, nuclear magnetic resonance (NMR) spectroscopy, and mass spectrometry are commonly used in organic and inorganic chemistry to determine the structure and purity of compounds. These techniques allow scientists to identify functional groups, detect impurities, and quantify the amount of a particular substance present.
Spectroscopy also plays a vital role in physics and astronomy. In physics, spectroscopy is used to study the interaction of atoms and molecules with light, providing insights into the fundamental principles of quantum mechanics and atomic physics. In astronomy, spectroscopy helps scientists determine the chemical composition, temperature, and velocity of celestial objects such as stars and galaxies. By analyzing the light emitted or absorbed by these objects, astronomers can gain insights into their physical properties and understand the processes happening within them.
Furthermore, spectroscopy is widely used in the field of biochemistry and biology. It allows researchers to investigate the structure and function of biomolecules such as proteins, nucleic acids, and carbohydrates. Techniques like ultraviolet-visible (UV-Vis) spectroscopy and fluorescence spectroscopy are used to study the absorption and emission of light by biological molecules, providing information about their structural changes, interactions, and enzymatic activities.
In summary, spectroscopy is an essential analytical tool in scientific research. Its applications span across various scientific disciplines and enable scientists to gain a deeper understanding of the properties and behavior of matter. Whether it is in chemistry, physics, astronomy, or biology, spectroscopy continues to contribute to new discoveries and advancements in scientific knowledge.
Interpreting the Spectroscopy Lab Results

In the spectroscopy lab, we performed various experiments using different types of spectroscopes to analyze the composition and properties of substances. The results obtained from these experiments can provide valuable insights into the molecular structure and behavior of the substances.
One of the main outcomes of the lab was the identification of different elements present in the samples. By examining the emission or absorption spectra, we were able to match the observed patterns with known spectral lines of elements. This allowed us to determine the elemental composition of the samples and confirm the presence of specific elements.
Additionally, the spectroscopy lab results provided information about the molecules and compounds present in the samples. By analyzing the vibrational and rotational energy levels observed in the spectra, we could infer the presence of specific chemical bonds and functional groups. This allowed us to identify the types of molecules and compounds present, as well as gain insights into their chemical structures.
The spectroscopy lab results also gave us information about the energy levels and transitions within the samples. By examining the intensity and positions of absorption or emission peaks, we could determine the energy required for electronic transitions and the energy levels of different electronic states. This information is crucial for understanding the electronic properties and behavior of the substances under study.
Common Challenges in Spectroscopy and How to Overcome Them
Spectroscopy is a powerful technique used in various scientific fields to analyze the electronic, vibrational, and rotational properties of atoms and molecules. However, it is not without its challenges. This article discusses some common challenges encountered in spectroscopy experiments and suggests possible solutions to overcome them.
1. Signal-to-Noise Ratio
One of the most common challenges in spectroscopy is achieving a high signal-to-noise ratio. This is crucial for obtaining accurate and reliable data. Noise in spectroscopy can arise from various sources, including electronic noise, background interference, and sample impurities.
To overcome this challenge, several strategies can be employed. First, one can use a technique called signal averaging, where multiple measurements are taken and averaged to reduce random noise. Another approach is to increase the intensity of the signal by using stronger light sources or increasing the sample concentration. Additionally, using appropriate filters and shielding can help minimize background interference.
2. Calibration and Instrument Drift
Calibration is essential in spectroscopy to ensure accurate measurements. Over time, instruments can experience drift, leading to inaccuracies in the acquired data. This can be caused by changes in environmental conditions, variations in detector performance, or instrument aging.
To overcome calibration and instrument drift challenges, regular calibration checks should be performed using appropriate standard samples. Additionally, instruments should be regularly serviced and maintained to minimize drift. Calibrations should also be performed at different wavelengths and intensities to account for potential nonlinearities in the system.
3. Spectral Interference

Spectral interference can arise when multiple analytes or compounds have overlapping absorption or emission spectra. This can lead to difficulties in accurately identifying and quantifying individual components in a mixture.
To overcome spectral interference, advanced spectral deconvolution techniques can be employed. These techniques analyze the spectra and mathematically separate the contributions of different components. Additionally, using selective wavelength regions or employing techniques such as derivative spectroscopy or wavelength modulation spectroscopy can help enhance the specificity of measurements.
In conclusion, while spectroscopy is a powerful tool, it is not immune to challenges. By understanding these challenges and implementing appropriate strategies, researchers can overcome them and obtain accurate and reliable spectroscopic data.
Application of Spectroscopy in Various Fields
Spectroscopy, the study of the interaction between matter and electromagnetic radiation, has found numerous applications in various fields. This powerful technique allows scientists to analyze and identify different substances based on their unique spectral signatures. In this article, we will explore some of the key areas where spectroscopy plays a crucial role.
1. Chemistry: Spectroscopy is widely used in chemistry to determine the structure, composition, and concentration of chemical compounds. It helps chemists study the interaction of light with molecules, providing valuable information about their electronic and vibrational states. This knowledge is essential for drug discovery, environmental analysis, and materials science.
2. Medicine: In medicine, spectroscopy is used for various diagnostic and therapeutic purposes. For example, infrared spectroscopy can identify the biochemical composition of tissues, aiding in the early detection of diseases such as cancer. Magnetic resonance spectroscopy (MRS) is used to study metabolic processes in the body, helping doctors monitor brain function and diagnose neurological disorders.
- Pharmaceuticals:
- Spectroscopy is instrumental in pharmaceutical analysis, allowing researchers to determine the purity and quality of drugs through techniques such as nuclear magnetic resonance (NMR) spectroscopy and mass spectrometry. It also helps in formulation development and drug stability testing.
- Environmental Science:
- Environmental scientists rely on spectroscopy to analyze air, water, and soil samples for pollutants and contaminants. Techniques such as flame atomic absorption spectroscopy (FAAS) and inductively coupled plasma-mass spectrometry (ICP-MS) are used to measure trace elements and heavy metals.
- Astronomy:
- Spectroscopy has revolutionized our understanding of the universe by allowing astronomers to study the light emitted by celestial objects. It helps in determining the composition, temperature, and motion of stars, galaxies, and other astronomical phenomena.
In conclusion, spectroscopy is a versatile tool that finds applications in various fields, ranging from chemistry and medicine to pharmaceuticals, environmental science, and astronomy. Its ability to provide detailed information about the properties of matter makes it an indispensable technique for scientists and researchers.
Conclusion

In conclusion, spectroscopy has played a crucial role in various scientific fields, including chemistry, physics, biology, and astronomy. It has provided valuable insights into the composition, structure, and behavior of matter at a molecular and atomic level.
Over the years, spectroscopy techniques have evolved, and new innovations have emerged to enhance the precision and capabilities of spectroscopic analysis. Some of the future trends and innovations in spectroscopy include:
- Advancements in instrumentation: The development of more sophisticated and sensitive spectrometers will enable scientists to perform measurements with greater accuracy and resolution.
- Miniaturization and portability: There is a growing demand for portable spectroscopy devices that can be used in fieldwork or remote locations. Miniaturization of spectrometers will make them more accessible and convenient for on-site analysis.
- Multidimensional spectroscopy: Techniques like two-dimensional infrared spectroscopy and multidimensional nuclear magnetic resonance (NMR) spectroscopy will provide a more comprehensive understanding of molecular interactions and dynamics.
- Advances in data analysis: With the increasing volume of data generated by spectroscopy experiments, there is a need for advanced data analysis techniques, such as artificial intelligence and machine learning algorithms, to extract meaningful insights from the data.
- Integration with other analytical techniques: Combining spectroscopy with other techniques, such as mass spectrometry, microscopy, or chromatography, will enable scientists to obtain more comprehensive information about samples.
In summary, spectroscopy will continue to be a vital tool for scientific research and discovery. The future holds exciting possibilities for the development of new spectroscopic techniques and applications, leading to breakthroughs in various fields and contributing to advancements in technology, medicine, and environmental science.