
Intermolecular forces play a crucial role in determining the physical and chemical properties of matter. These forces, which exist between molecules, are responsible for phenomena such as boiling point, solubility, and surface tension. Understanding the different types of intermolecular forces and their effects is essential for comprehending the behavior and characteristics of substances in various states.
In the Pogil activity on intermolecular forces, students are introduced to the key concepts and principles behind these forces. By examining different substances and analyzing their properties, students are prompted to think critically and apply their knowledge to real-world examples. The answer key for this activity provides a comprehensive guide to help students navigate through the questions and reinforce their understanding of the topic.
Through the Pogil activity, students will explore various types of intermolecular forces, including London dispersion forces, dipole-dipole forces, and hydrogen bonding. They will learn how these forces arise from the interactions between molecules and their electron distribution. The answer key for this activity will give students the opportunity to check their answers and ensure they have grasped the fundamental concepts correctly.
By utilizing the Pogil intermolecular forces answer key, students will be able to deepen their understanding of the topic and build a solid foundation in the study of matter and its properties. This resource serves as a valuable tool for educators, providing them with guidance and support to facilitate meaningful classroom discussions and further engage students in the exploration of intermolecular forces.
Pogil InterMolecular Forces Answer Key: Understanding the Basics
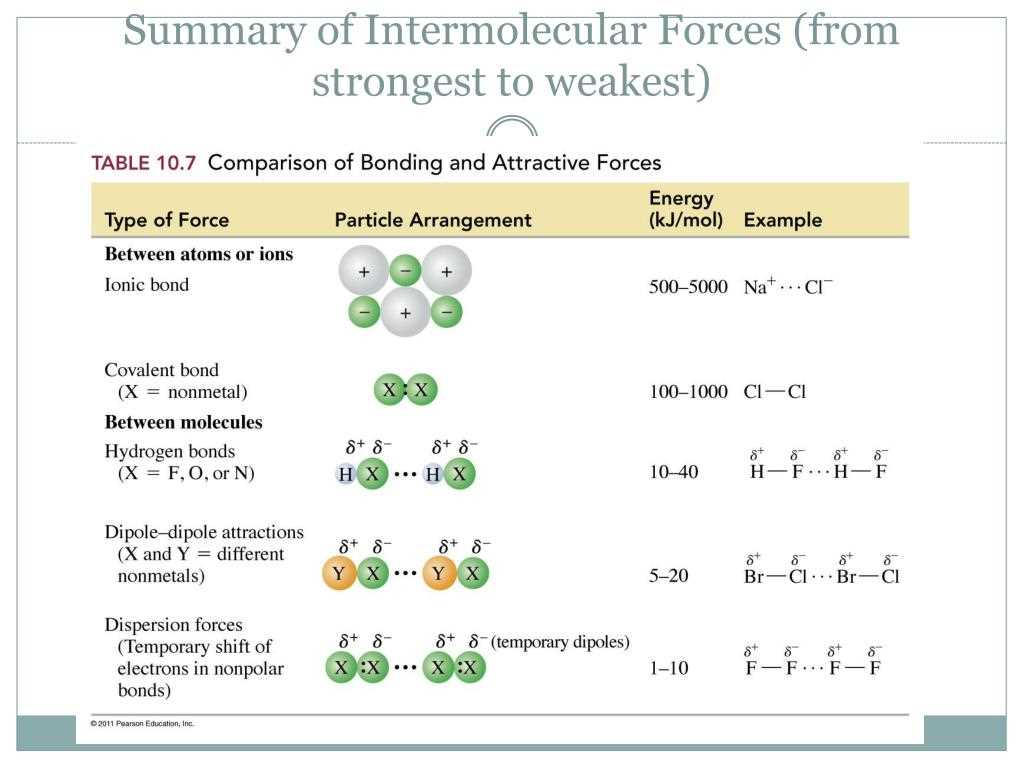
The Pogil InterMolecular Forces Answer Key is a valuable resource for students learning about the basics of intermolecular forces in chemistry. Intermolecular forces are the forces of attraction between molecules that determine many of their physical properties, such as boiling point, melting point, and solubility.
This answer key provides students with the necessary tools to understand and solve problems related to intermolecular forces. It includes a comprehensive list of answers to the questions and activities in the Pogil worksheet, helping students verify their understanding and learn from their mistakes.
Key Concepts Covered:
- Van der Waals forces: These are weak intermolecular forces that arise from the temporary fluctuations in electron distribution within molecules.
- Dipole-dipole forces: These are stronger intermolecular forces that occur between polar molecules due to the positive and negative ends of their molecular dipoles attracting each other.
- Hydrogen bonding: This is a type of dipole-dipole force that occurs between a hydrogen atom bonded to an electronegative atom (such as oxygen, nitrogen, or fluorine) and an unshared electron pair on another electronegative atom.
The Pogil InterMolecular Forces Answer Key also provides explanations for the different types of intermolecular forces, examples of molecules that exhibit each type of force, and practice problems to reinforce the concepts. By using this answer key, students can enhance their understanding of intermolecular forces and confidently solve problems related to these forces in chemistry.
Types of InterMolecular Forces

Intermolecular forces are the forces of attraction between molecules that determine their physical properties, such as boiling point, melting point, and solubility. There are several types of intermolecular forces, including:
- London Dispersion Forces: These forces are the weakest intermolecular forces. They occur between all molecules and are caused by the temporary shifting of electron clouds, resulting in temporary dipoles. The strength of London dispersion forces increases with the size of the molecules and the number of electrons.
- Dipole-Dipole Forces: These forces occur between molecules that have permanent dipoles. A dipole is a separation of positive and negative charges within a molecule. The positive end of one molecule is attracted to the negative end of another, resulting in a dipole-dipole interaction. The strength of dipole-dipole forces increases with the electronegativity difference between the atoms in the molecule.
- Hydrogen Bonding: Hydrogen bonding is a special type of dipole-dipole interaction. It occurs when hydrogen is bonded to a highly electronegative atom, such as nitrogen, oxygen, or fluorine. The hydrogen atom in the molecule has a partial positive charge and is attracted to the partial negative charge on the electronegative atom. Hydrogen bonding is stronger than regular dipole-dipole interactions.
These intermolecular forces play a crucial role in determining the physical and chemical properties of substances. They affect the boiling and melting points of substances, as well as their solubilities. Understanding the different types of intermolecular forces is important in understanding molecular interactions and the behavior of substances in various conditions.
The Importance of InterMolecular Forces in Chemistry

Intermolecular forces play a crucial role in several aspects of chemistry. These forces are responsible for determining the physical and chemical properties of substances and are essential for understanding molecular interactions.
One of the primary functions of intermolecular forces is to hold molecules together in a solid or a liquid. These forces are responsible for the attractive interactions between neighboring molecules that allow them to form a cohesive substance. For example, in the case of water, hydrogen bonding–a type of intermolecular force–holds the water molecules together, giving water its unique properties such as high boiling point and surface tension.
In addition to holding substances together, intermolecular forces are also involved in several chemical reactions. For instance, in reactions involving acids and bases, intermolecular forces can influence the strength of the acid or base. Strong intermolecular forces between acid molecules can lead to a stronger acid, while weaker intermolecular forces may result in a weaker acid.
Furthermore, intermolecular forces also play a role in the solubility of substances. The strength and type of intermolecular forces between a solute and a solvent determine whether the solute will dissolve or not. For example, polar solutes are more likely to dissolve in polar solvents due to the similar intermolecular forces between the molecules.
In summary, intermolecular forces are crucial in chemistry as they determine the physical and chemical properties of substances, influence chemical reactions, and affect solubility. Understanding these forces is essential for studying molecular interactions and predicting the behavior of different substances.
Properties and Effects of InterMolecular Forces

Intermolecular forces are the attractive forces that exist between molecules. These forces play a crucial role in determining the physical properties of substances, such as boiling point, melting point, surface tension, and solubility. Understanding the properties and effects of intermolecular forces is essential in various fields, including chemistry, physics, and materials science.
1. Boiling Point and Melting Point: Intermolecular forces affect the boiling point and melting point of substances. The strength of these forces determines how tightly the molecules are held together. Substances with stronger intermolecular forces will require more energy to break the bonds and change from a solid to a liquid or from a liquid to a gas, resulting in higher boiling and melting points.
2. Surface Tension: Intermolecular forces also play a role in surface tension, which is the measure of the attraction between molecules at the surface of a liquid. Substances with stronger intermolecular forces will have higher surface tension because the molecules at the surface are more closely packed together.
3. Solubility: Intermolecular forces affect the solubility of substances in different solvents. Like dissolves like – substances with similar intermolecular forces are more likely to dissolve in each other. For example, polar substances dissolve in polar solvents due to the attraction between the positive and negative charges, while nonpolar substances dissolve in nonpolar solvents.
In summary, intermolecular forces have a significant impact on the physical properties and behavior of substances. By understanding and manipulating these forces, scientists can develop new materials with specific properties, find suitable solvents for dissolving certain substances, and explain phenomena such as boiling and melting points. The study of intermolecular forces is an essential aspect of modern science and has numerous applications in various industries.
Pogil Activity on InterMolecular Forces: Step-by-Step Guide
Intermolecular forces play a significant role in the properties and behavior of substances. Understanding these forces is crucial in comprehending various phenomena, such as boiling and melting points, solubility, and even the behavior of biological molecules.
In this Pogil activity on intermolecular forces, we will explore the different types of intermolecular forces and their impact on molecular properties. The activity is designed to provide a step-by-step guide to help you understand the concepts and apply them to various scenarios.
Step 1: Start by reviewing the provided background information on intermolecular forces. This will give you a solid foundation before diving into the activity. Take note of key concepts and definitions to aid in your understanding.
Step 2: Next, analyze the given scenarios and identify the type of intermolecular force at play. These scenarios may involve substances in different states, such as solids, liquids, or gases. Determine the forces involved based on the nature of the molecules and their interactions.
Step 3: With a clear understanding of the intermolecular forces involved, proceed to answer the specific questions provided for each scenario. These questions will prompt you to apply your knowledge to explain observed behaviors or predict outcomes based on the forces at play.
Step 4: After completing the questions, compare your answers with the provided answer key. Take note of any discrepancies and review the corresponding explanations to further solidify your understanding of intermolecular forces.
By following this step-by-step guide, you will gain a comprehensive understanding of intermolecular forces and their role in shaping molecular properties. This activity will strengthen your ability to analyze and predict the behavior of various substances, making it an essential tool in your study of chemistry.
Understanding the Pogil Activity Answer Key

When completing a Pogil activity, students are often provided with an answer key to check their work and assess their understanding of the material. The Pogil activity answer key serves as a guide for students to compare their own answers to the correct solutions. It allows students to identify any errors or misconceptions they may have and helps them learn from their mistakes. The answer key provides a valuable tool for self-assessment and self-correction, allowing students to take ownership of their learning.
The Pogil activity answer key typically includes detailed explanations and step-by-step solutions for each question or problem in the activity. This helps students understand the reasoning behind the correct answers and allows them to apply the concepts learned to similar problems in the future. The answer key may also include additional notes or tips to further enhance students’ understanding of the topic.
Using the Pogil activity answer key effectively requires active engagement and critical thinking. Students should not simply use the answer key as a shortcut to obtain the correct answers, but rather as a tool to deepen their understanding of the material. They should carefully review the answer key, compare it to their own work, and analyze any differences or mistakes. This process helps students identify their weaknesses and areas for improvement, allowing them to develop a stronger grasp of the subject matter.
The Pogil activity answer key can be a powerful learning resource when used properly. It helps foster independent learning and encourages students to take responsibility for their own progress. By utilizing the answer key as a tool for reflection and self-assessment, students can enhance their understanding of the topic and improve their problem-solving skills.
Common Mistakes and Troubleshooting in the Pogil Activity

In the Pogil activity on intermolecular forces, students might make several common mistakes. One mistake is confusing the concepts of intermolecular forces and intramolecular forces. It is important for students to understand that intermolecular forces are between molecules, while intramolecular forces are within a molecule.
Another common mistake is not considering the different types of intermolecular forces. There are three main types of intermolecular forces: London dispersion forces, dipole-dipole forces, and hydrogen bonding. Students should be careful not to mix up these different types when identifying the intermolecular forces present in a substance.
When troubleshooting this Pogil activity, it is helpful to provide additional examples and practice problems for students to reinforce their understanding of intermolecular forces. This can help them recognize patterns and apply their knowledge to different scenarios.
Furthermore, it is important to encourage students to carefully read and analyze the information provided in the Pogil activity. Sometimes, students may overlook key details or misinterpret the given information, leading to incorrect answers. Emphasizing the importance of reading comprehension can help address this issue.
Additionally, providing explanations and clarifications for any ambiguous or confusing sections of the activity can also help students overcome challenges. This can be done through class discussions, small group discussions, or one-on-one interactions with the teacher.
Overall, by being aware of these common mistakes and implementing appropriate troubleshooting strategies, educators can help students successfully navigate the Pogil activity on intermolecular forces and enhance their understanding of the topic.