
As the end of the semester approaches, students are eagerly preparing to showcase their knowledge in the final exam for physical science. This exam is designed to test their understanding of the fundamental principles and concepts in the field, covering a wide range of topics such as physics, chemistry, and astronomy.
One of the key aspects of this exam is the ability to apply theoretical concepts to real-life situations. Students are expected to demonstrate their problem-solving skills by analyzing and interpreting data, conducting experiments, and making accurate predictions. The exam also assesses their understanding of scientific models, laws, and theories, and their ability to communicate their findings effectively.
To succeed in the physical science final exam, students must have a strong grasp of the basic principles and formulas. They need to be able to comprehend scientific literature, solve complex equations, and manipulate variables. Additionally, they should be familiar with different laboratory techniques and be able to perform experiments accurately and safely.
Overall, the physical science final exam is a comprehensive evaluation of the students’ knowledge, analytical thinking, and problem-solving abilities. It tests their ability to think critically and apply scientific principles in various scenarios. By preparing thoroughly and understanding the key concepts, students can confidently approach the exam and showcase their proficiency in the subject.
Key concepts and topics covered in a physical science final exam
Physical science is a broad field that encompasses the study of various natural phenomena and the fundamental principles that govern them. A final exam in physical science is designed to assess a student’s understanding of these key concepts and topics. Here are some of the main areas that may be covered in a physical science final exam:
- Chemistry: This topic explores the properties and behavior of matter. It can include concepts such as atomic structure, chemical reactions, stoichiometry, and the periodic table. Students may be asked to identify elements, balance chemical equations, or calculate molar masses.
- Physics: Physics focuses on the study of energy, motion, and forces. Key topics may include kinematics, Newton’s laws of motion, work and energy, and simple machines. Students may need to solve problems involving motion, calculate forces, or analyze the effects of different variables.
- Energy: This concept covers various forms of energy, including potential and kinetic energy, thermal energy, and electrical energy. Students may be asked to determine the amount of energy transferred in different systems or calculate efficiency.
- Waves and Sound: This topic delves into the properties and behaviors of waves, as well as the nature of sound. Students may need to understand concepts such as wavelength, frequency, amplitude, and the Doppler effect. They may also be asked to analyze wave patterns or solve problems related to sound waves.
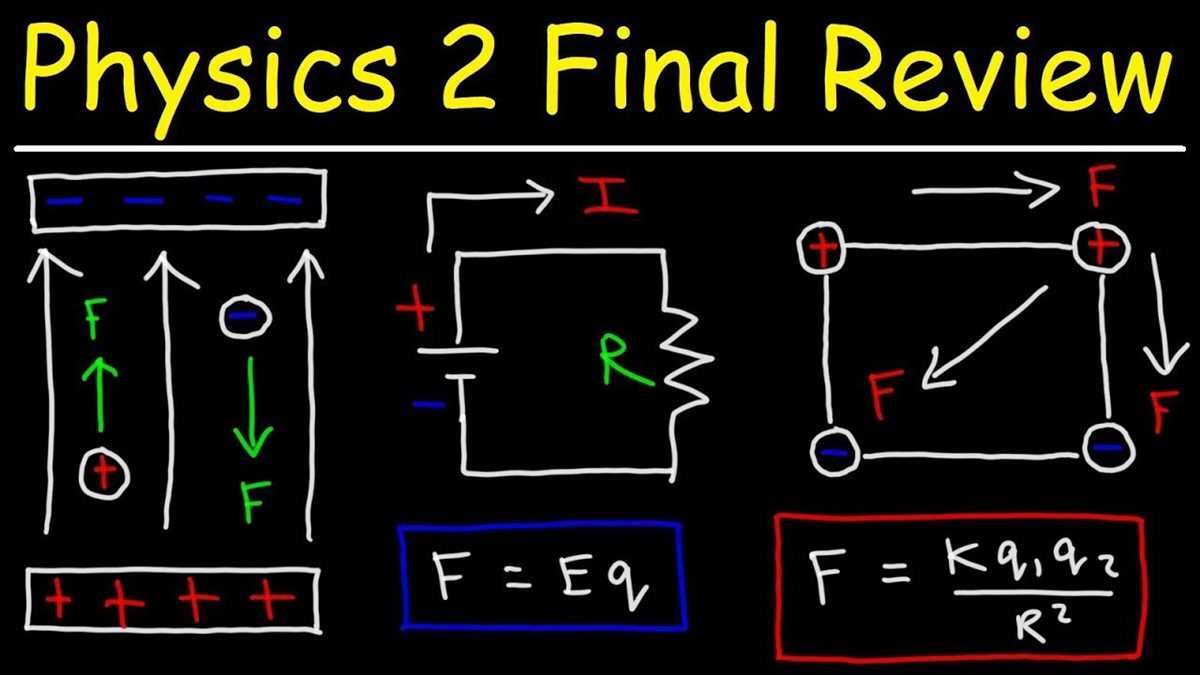
- Electricity and Magnetism: This area of study explores the principles of electricity and magnetism, including circuits, electromagnetism, and electromagnetic waves. Students may need to analyze circuits, calculate currents, or understand the relationship between magnetic fields and electric currents.
A physical science final exam can cover a wide range of topics within these overarching themes. It requires students to apply their knowledge and problem-solving skills to real-world scenarios. By mastering these key concepts, students will have a solid foundation in physical science and be prepared for further studies in related fields.
Properties of Matter

Matter is anything that has mass and occupies space. It can exist in various forms including solid, liquid, and gas. The properties of matter can be observed and measured to determine its characteristics and behavior. These properties can be classified into two categories: physical properties and chemical properties.
Physical properties of matter are those that can be observed or measured without changing the substance’s chemical composition. Examples of physical properties include mass, volume, density, color, and temperature. These properties can be used to identify and classify different types of matter.
Chemical properties of matter describe how it behaves in the presence of other substances or during a chemical reaction. These properties can only be observed when the substance undergoes a chemical change. Examples of chemical properties include reactivity, flammability, and toxicity. Understanding the chemical properties of a substance is important for predicting its behavior in various situations.
Physical Properties of Matter
- Mass: the amount of matter in an object
- Volume: the amount of space occupied by an object
- Density: the ratio of an object’s mass to its volume
- Color: the visual appearance of an object
- Temperature: a measure of the average kinetic energy of the particles in a substance
Chemical Properties of Matter
- Reactivity: the ability of a substance to undergo a chemical reaction with other substances
- Flammability: the ability of a substance to burn
- Toxicity: the degree to which a substance can cause harm to living organisms
Understanding the properties of matter is essential in fields such as chemistry, physics, and materials science. By studying these properties, scientists can gain a deeper understanding of the nature of matter and its interactions with other substances. This knowledge can be utilized in various applications, from designing new materials to developing medications and exploring the mysteries of the universe.
Atomic Structure and the Periodic Table
The study of atomic structure and the periodic table is fundamental to understanding the behavior of matter at its most basic level. Atoms are the building blocks of matter, and their structure determines their properties and behavior. The periodic table organizes all known elements based on their atomic number, which is the number of protons in the nucleus of an atom.
An atom consists of a nucleus, which contains protons and neutrons, surrounded by electrons in energy levels or shells. Protons have a positive charge, neutrons have no charge, and electrons have a negative charge. The balance between the protons and electrons determines the overall charge of the atom. In a neutral atom, the number of protons and electrons is equal.
The periodic table is a tabular arrangement of elements, where elements with similar properties are grouped together in vertical columns called groups. Each element is represented by a symbol and is designated by its atomic number. Elements in the same group have similar outer electron configurations, which gives them similar chemical properties. The periods in the periodic table represent the energy levels or shells that electrons occupy.
- The elements in the periodic table are arranged in increasing order of atomic number.
- Elements in the same group have similar chemical properties.
- Elements in the same period have the same number of energy levels.
- The periodic table can be divided into blocks based on the electron configurations of elements.
Understanding atomic structure and the periodic table is essential for predicting and explaining chemical reactions, understanding bonding, and analyzing the properties of different elements. It provides a framework for understanding the behavior of matter and the composition of substances in the physical world.
Forces and Motion
In the field of physics, forces play a crucial role in understanding and predicting the motion of objects. A force can be defined as a push or pull that can change the state of motion of an object. Forces can act in different directions and have different magnitudes, resulting in various types of motion.
One of the fundamental principles in the study of forces and motion is Newton’s laws of motion. According to Newton’s first law, an object at rest tends to stay at rest, and an object in motion tends to stay in motion with the same speed and in the same direction unless acted upon by an external force.
There are different types of forces, including:
- Gravity: The force that attracts objects towards each other, such as the force that pulls objects towards the Earth.
- Friction: The force that opposes the motion of objects when they are in contact with each other.
- Tension: The force that is transmitted through a string, rope, or cable when it is pulled tight.
- Normal force: The force exerted by a surface to support the weight of an object resting on it.
Understanding the forces acting on an object and their interactions is essential in predicting the object’s motion. It allows scientists to explain phenomena such as the motion of planets, the behavior of falling objects, and the mechanics of various machines. By studying forces and motion, scientists can make advancements in fields such as engineering, transportation, and even sports.
Energy and its forms
Energy is a fundamental concept in physical science that is defined as the ability to do work. It is a property possessed by objects and substances, and it can take different forms. These forms of energy can be categorized into various types based on their characteristics and modes of transfer. Understanding the different forms of energy is crucial in comprehending the various processes and phenomena that occur in the physical world.
One of the most common forms of energy is mechanical energy, which encompasses both kinetic energy and potential energy. Kinetic energy is the energy possessed by an object in motion, while potential energy is the energy stored in an object due to its position or condition. Other forms of energy include thermal energy, which is the energy associated with the motion of particles in an object, and electromagnetic energy, which is the energy carried by electromagnetic waves such as light.
- Chemical energy: Energy stored in chemical bonds between atoms or molecules.
- Nuclear energy: Energy released from the nucleus of an atom through processes such as fission and fusion.
- Electrical energy: Energy associated with the movement of electric charges.
- Sound energy: Energy produced by the vibration of objects, transmitted through a medium such as air or water.
- Gravitational energy: Energy possessed by an object due to its height or position in a gravitational field.
- Elastic energy: Energy stored in a compressed or stretched object, such as a spring.
Energy can be converted from one form to another, and it is subject to the law of conservation of energy, which states that energy cannot be created or destroyed, but only transformed from one form to another. This principle is fundamental to understanding various natural processes and the functioning of systems in the physical world.
Waves and Electromagnetic Radiation

In the field of physical science, waves play a crucial role in understanding and explaining the behavior of various phenomena. Waves are disturbances that transfer energy from one point to another without the physical transfer of matter. They can be categorized into two main types: mechanical waves and electromagnetic waves.
Mechanical waves require a physical medium, such as air, water, or solids, to propagate. These waves travel through the vibration or oscillation of particles in the medium. Examples of mechanical waves include sound waves, water waves, and seismic waves. The behavior of mechanical waves can be described by their amplitude, wavelength, frequency, and speed.
On the other hand, electromagnetic waves do not rely on a medium for their propagation. They can travel through empty space, such as the vacuum of outer space. Electromagnetic waves are generated by the vibrations of electric and magnetic fields. This type of wave includes radio waves, microwaves, infrared radiation, visible light, ultraviolet radiation, X-rays, and gamma rays. Electromagnetic waves are characterized by their wavelength and frequency, which determine their energy and the type of radiation they represent.
- Radio waves have the longest wavelength and lowest frequency among electromagnetic waves.
- Visible light, which is crucial for human vision, falls within a specific range of wavelengths and frequencies.
- X-rays and gamma rays have the shortest wavelength and highest frequency among electromagnetic waves.
The study of waves and electromagnetic radiation is essential in many areas of science and technology. It helps us understand the behavior of light, the transmission of energy, and the interaction between matter and radiation. Scientists and engineers apply this knowledge in various fields, including telecommunications, medical imaging, astronomy, and environmental monitoring.
Chemical reactions and equations
A chemical reaction is a process in which one or more substances, known as reactants, interact and undergo a chemical change to form new substances, called products. These reactions involve the breaking and forming of chemical bonds, resulting in the rearrangement of atoms and the conversion of one set of substances into another.
Chemical equations are used to represent chemical reactions. They provide a concise and standardized way to show the reactants, products, and stoichiometric ratios involved in the reaction. In a chemical equation, the reactants are written on the left side of the arrow, indicating the starting materials, while the products are written on the right side, indicating the substances formed as a result of the reaction.
A balanced chemical equation ensures that the number of atoms of each element is the same on both sides of the equation. This is achieved by adjusting the coefficients in front of each reactant and product. Balancing equations is important in order to accurately represent the conservation of matter, as atoms are neither created nor destroyed during a chemical reaction.
Chemical reactions can be classified into different types, including synthesis (combination) reactions, decomposition reactions, displacement reactions, and combustion reactions. Synthesis reactions involve the combination of two or more substances to form a single compound, while decomposition reactions involve the breakdown of a compound into its constituent elements or simpler compounds. Displacement reactions involve the transfer of an element from one compound to another, while combustion reactions involve the reaction of a substance with oxygen, typically resulting in the release of energy in the form of heat and light.
In conclusion, chemical reactions and equations are fundamental concepts in physical science. They help us understand and predict how different substances interact and transform into new substances. By balancing chemical equations and understanding the different types of reactions, we can analyze and interpret the chemical processes that occur in the natural world and in various industries, such as pharmaceuticals, materials science, and environmental science.