PhET simulations provide an interactive way for students to explore scientific concepts and deepen their understanding. In this article, we will discuss the key findings and answers from the PhET investigation of acids and bases simulation.
The PhET investigation of acids and bases simulation allows students to delve into the properties and behaviors of these fundamental substances. Through the simulation, students have the opportunity to experiment with different concentrations of acids and bases, observe pH changes, and explore the concept of neutralization.
One of the main objectives of the investigation is to determine the effect of concentration on the pH of a solution. By manipulating the concentration sliders in the simulation, students can observe how increasing the concentration of an acid or a base affects the pH reading. Through their exploration, students will discover that as the concentration of an acid or a base increases, the pH becomes more acidic or basic, respectively.
Furthermore, the investigation allows students to explore the concept of neutralization and the role of acids and bases in this chemical reaction. Students can mix different combinations of acids and bases and observe how the pH changes as neutralization occurs. They will find that when an acid and a base react, they neutralize each other, resulting in a solution with a neutral pH of 7.
Understanding Acids and Bases: An In-depth Investigation with PhET
Acids and bases are fundamental concepts in chemistry that play a crucial role in various chemical reactions and everyday life. To gain a deeper understanding of these concepts, the PhET Interactive Simulations project offers an engaging and interactive tool for students to explore acids and bases in a virtual laboratory setting. This investigation aims to provide students with hands-on experience and enable them to comprehend the properties and behaviors of acids and bases.
The PhET simulation allows students to conduct experiments and manipulate different variables to observe the effects on the pH scale, color changes, and conductivity of various substances. With the help of this simulation, students can explore the behavior of acids and bases in different scenarios and understand the concept of neutralization reactions. Through this interactive learning experience, students can gain a concrete understanding of the properties of acids and bases.
During the investigation, students can explore the strength of acids and bases by varying their concentrations and observing the resulting pH changes. They can also investigate the impact of dilutions on pH values and understand the concept of pH as a measure of hydrogen ion concentration. Additionally, the simulation provides opportunities to explore the concept of acid-base indicators and their role in determining the pH of a solution.
Furthermore, the PhET simulation allows students to explore the behavior of acids and bases in different solvents, such as water and ammonia. By analyzing the conductivity of solutions and observing color changes, students can gain insights into the ionization of acids and bases and the role of solvents in their behavior. This investigation provides a comprehensive understanding of the behavior and properties of acids and bases, allowing students to make connections between theory and real-life examples.
In conclusion, the PhET investigation of acids and bases offers an interactive and engaging learning experience for students to deepen their understanding of these fundamental concepts. Through hands-on experimentation, manipulation of variables, and analysis of results, students can gain a comprehensive understanding of the properties and behaviors of acids and bases. This virtual laboratory experience enables students to make connections between theory and practical applications, enhancing their overall comprehension of the topic.
Exploring the PhET Acid-Base Solutions Simulation

The PhET Acid-Base Solutions Simulation allows users to explore the behavior of acids and bases in a virtual laboratory setting. The simulation provides an interactive experience wherein users can manipulate various factors such as concentration, strength, and pH levels to observe their effects on acid-base reactions. This simulation serves as a valuable educational tool for students and educators alike, providing a hands-on learning experience that enhances understanding of key concepts in chemistry.
One of the key features of the simulation is the ability to test different combinations of acids and bases and observe the resulting reactions. By adjusting the concentration and strength of the solutions, users can see how pH levels change and determine whether a solution is acidic, basic, or neutral. Additionally, the simulation allows users to measure the pH of a solution using a pH meter, further enhancing their understanding of pH as a measure of acidity or alkalinity.
The simulation also includes a variety of interactive tools that aid in the exploration of acid-base behavior:
- The color-coded pH scale: This visual tool allows users to easily determine the pH level of a solution by comparing its color to the pH scale. By observing the color change of acids and bases as they react, users can gain a deeper understanding of the concept of pH and the pH scale.
- The concentration slider: This slider allows users to adjust the concentration of an acid or base solution, providing insight into the relationship between concentration and pH. Users can observe how increasing or decreasing the concentration affects the pH and learn about the concept of dilution.
- The strength slider: This slider allows users to adjust the strength of an acid or base solution, exposing them to the concept of strong and weak acids and bases. By observing the behavior of strong and weak acids and bases in the simulation, users can learn about the dissociation of ions and the impact on pH.
In conclusion, the PhET Acid-Base Solutions Simulation offers an engaging and interactive way to explore the behavior of acids and bases. Through the simulation, users can manipulate various factors and observe the resulting changes in pH, providing a deeper understanding of key concepts in chemistry. Whether used in a classroom setting or for self-guided learning, this simulation is a valuable tool for enhancing understanding and knowledge in the field of acid-base chemistry.
Key Concepts in Acid-Base Chemistry
The concept of acidity and basicity is a fundamental topic in chemistry, as it plays a crucial role in various chemical reactions and processes. Acids and bases are substances that can donate or accept protons (H+ ions) during a chemical reaction. Understanding the key concepts in acid-base chemistry is essential for explaining and predicting the behavior of these substances.
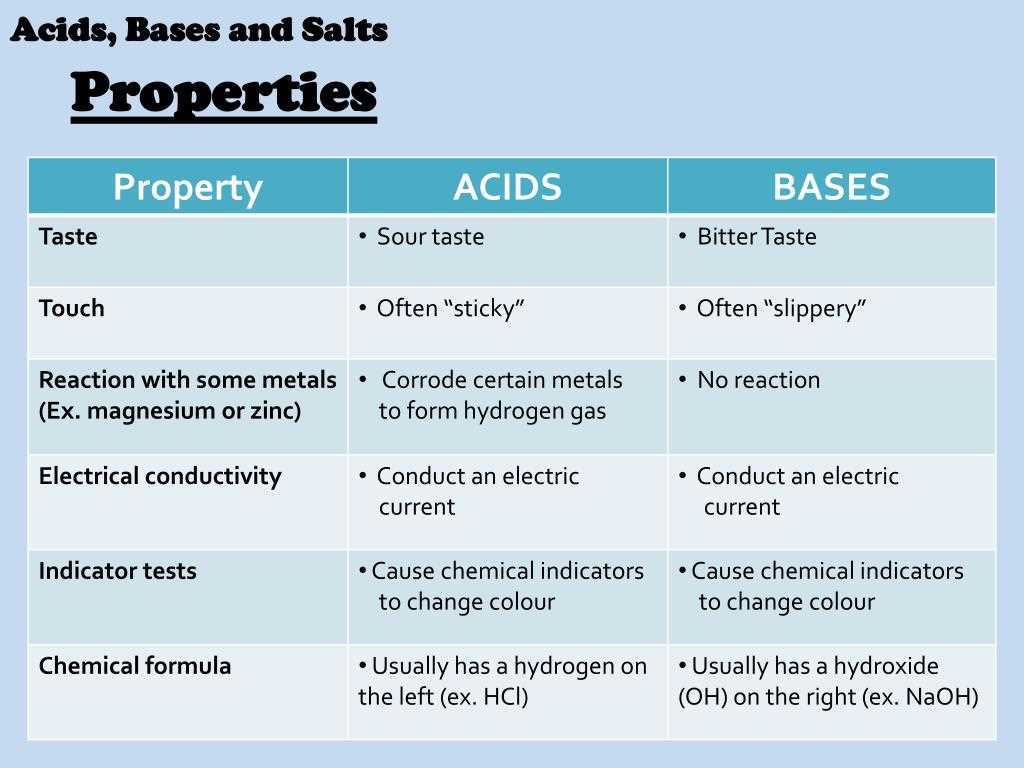
The first key concept is the definition of acids and bases. According to the Arrhenius definition, an acid is a substance that releases H+ ions when dissolved in water, while a base is a substance that releases OH- ions when dissolved in water. The Bronsted-Lowry definition expands on this, stating that an acid is a proton donor, and a base is a proton acceptor. This definition allows for the classification of substances as acids or bases in various solvents, not just water.
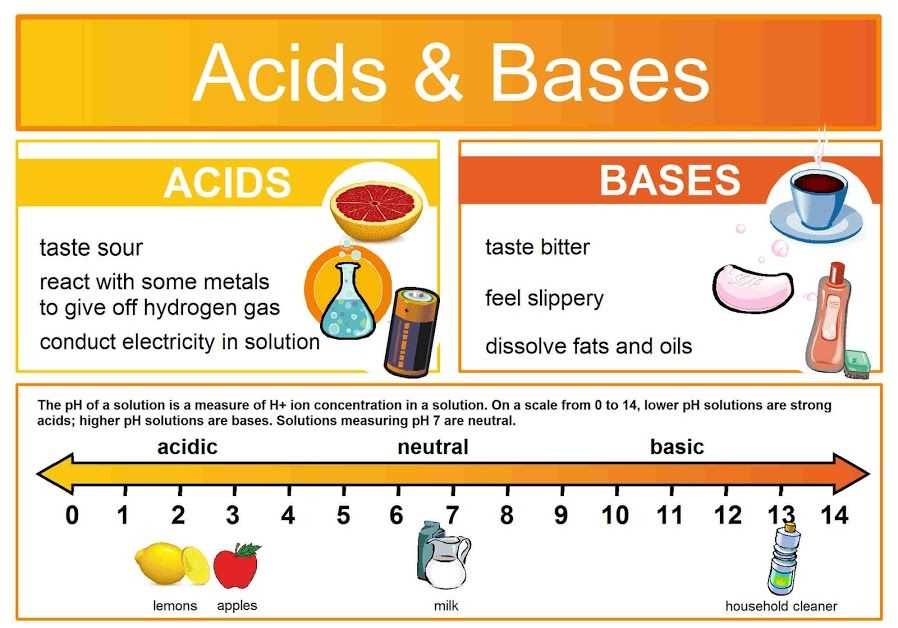
Another key concept is the pH scale, which is used to measure the acidity or basicity of a solution. The pH scale ranges from 0 to 14, with a pH of 7 considered neutral, less than 7 being acidic, and greater than 7 being basic. The pH is determined by the concentration of H+ ions in the solution. Acids have a higher concentration of H+ ions, which lowers the pH, while bases have a lower concentration of H+ ions, which raises the pH.
Furthermore, acid-base reactions involve the transfer of protons from the acid to the base. In a typical acid-base reaction, the acid donates a proton to the base, forming a conjugate base and a conjugate acid, respectively. The strength of an acid or base is determined by its ability to donate or accept protons. Strong acids and bases completely dissociate in water, while weak acids and bases only partially dissociate.
In summary, the key concepts in acid-base chemistry include the definition of acids and bases, the pH scale, and acid-base reactions. Understanding these concepts allows scientists to analyze and predict the behavior of acids and bases in various chemical reactions and environments.
Experimental Investigation: pH and Acid-Base Reactions
Investigating the pH and acid-base reactions is crucial to understanding the chemical properties of substances and their interactions. The pH scale, ranging from 0 to 14, measures the acidity or alkalinity of a solution. Acids, with pH values below 7, release hydrogen ions (H+) when dissolved in water, while bases, with pH values above 7, produce hydroxide ions (OH-). This experiment aims to study the effects of acids and bases on different substances and determine their pH levels.
The experiment begins by gathering various substances, including acids (such as vinegar or lemon juice) and bases (such as baking soda or soap), as well as neutral substances like water. A series of tests are then conducted using pH paper or a pH meter to measure the pH levels of each substance. The pH paper changes color when it comes into contact with acidic or basic solutions, allowing for a visual representation of their pH levels.
Through the investigation, students can observe the different reactions that occur when acids and bases are mixed with certain substances. For example, when an acid is added to baking soda, a chemical reaction takes place, resulting in the production of carbon dioxide gas. This reaction is commonly observed in baking, as the carbon dioxide gas helps dough rise. Similarly, when a base is mixed with an acid, a neutralization reaction occurs, resulting in the formation of a salt and water.
- Acids have a sour taste and can cause a burning sensation when in contact with the skin or eyes.
- Bases have a bitter taste and can feel slippery or soapy to the touch.
The experimental investigation of pH and acid-base reactions provides students with hands-on experience and a deeper understanding of these chemical concepts. It allows them to observe the characteristics of different substances, their pH levels, and the reactions they undergo when mixed with acids or bases. This knowledge is essential in various fields, including chemistry, biology, and environmental science, as it helps scientists and researchers understand the behavior of substances in different chemical reactions and their effects on the natural world.
Analysis of Experimental Results: Interpreting Data and Observations
During the Phet investigation of acids and bases, several experiments were conducted to study the properties and behavior of different solutions. By analyzing the data and observations collected, we can gain insights into the nature of acids and bases and how they interact with various substances.
Data Analysis:
The collected data included measurements of pH levels, color changes, and reaction rates. These measurements were essential in determining the acidity or basicity of each solution and understanding the reactions that took place during the experiments.
By analyzing the pH levels, we were able to identify whether a solution was acidic (pH < 7), neutral (pH = 7), or basic (pH > 7). This helped us classify each solution and compare their properties.
Color changes observed in certain solutions indicated a chemical reaction had occurred. These color changes were indicators of the presence or absence of specific ions or compounds, helping us understand the nature of the reactions that took place.
Reaction rates were also analyzed to determine the speed at which different solutions interacted. Faster reaction rates indicated stronger acidity or basicity, while slower rates suggested weaker acidity or basicity.
Interpretation of Observations:

Based on the data and observations collected, several conclusions can be drawn about the properties of acids and bases:
- Acids tend to have a lower pH level, below 7, indicating a higher concentration of hydrogen ions (H+).
- Bases, on the other hand, have a higher pH level, above 7, indicating a higher concentration of hydroxide ions (OH-).
- Acids have the ability to donate H+ ions, while bases can accept H+ ions or donate OH- ions.
- The neutralization reaction between an acid and a base results in the formation of water and a salt.
- Certain indicators, such as litmus paper or phenolphthalein, can be used to determine whether a solution is acidic or basic based on color changes.
Conclusion:
The analysis of the experimental results has provided valuable insights into the properties and behavior of acids and bases. Understanding the pH levels, color changes, and reaction rates has allowed us to classify and differentiate between different solutions. By interpreting the data and observations, we have gained a deeper understanding of the nature of acids and bases and their interactions. This knowledge can be applied in various fields, such as chemistry, biology, and environmental science, to better understand and control chemical reactions and their effects.