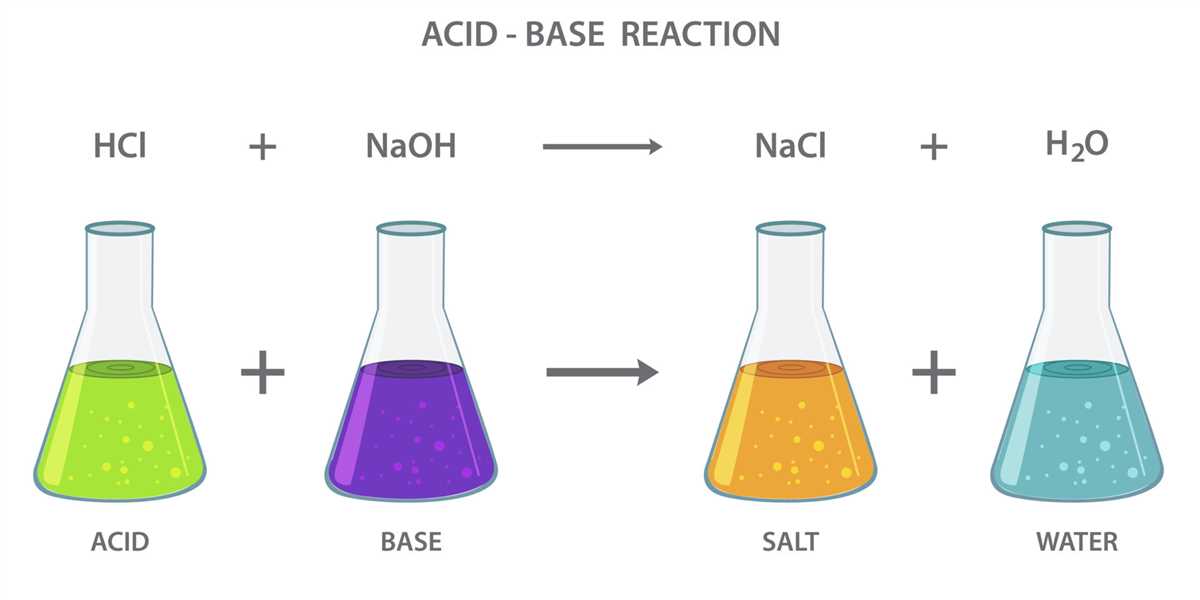
In the world of chemistry, neutralization reactions play a crucial role in understanding the properties and behavior of various substances. One such commonly encountered reaction is the neutralization of a soft drink. This lab experiment aims to explore the chemical reactions involved when an acid, usually found in soft drinks, is reacted with a base to achieve a neutral pH.
The first step of the lab experiment involves gathering materials such as a soft drink, a pH indicator, a burette, and a known concentration of a base, usually sodium hydroxide. The soft drink is then measured and poured into a beaker. A few drops of the pH indicator are added to the soft drink, which changes its color depending on the pH. The burette is then filled with the base solution, and the initial volume is recorded.
Next, the base solution is gradually added to the soft drink while stirring continuously. The pH indicator helps monitor the changes in pH, indicating the level of acidity or alkalinity. As the base solution is added, the pH indicator gradually changes color, indicating that the acid in the soft drink is being neutralized by the base. The addition of the base continues until the indicator reaches a neutral pH, usually indicated by a green color.
Understanding the Neutralization Process in a Soft Drink Lab
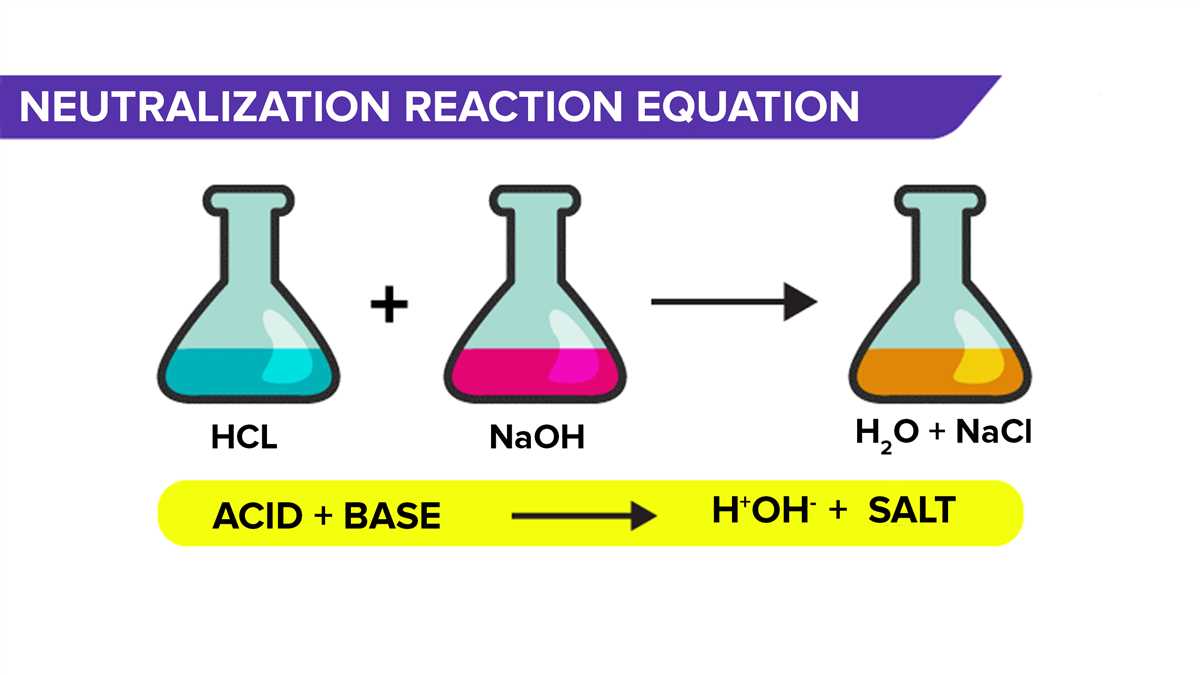
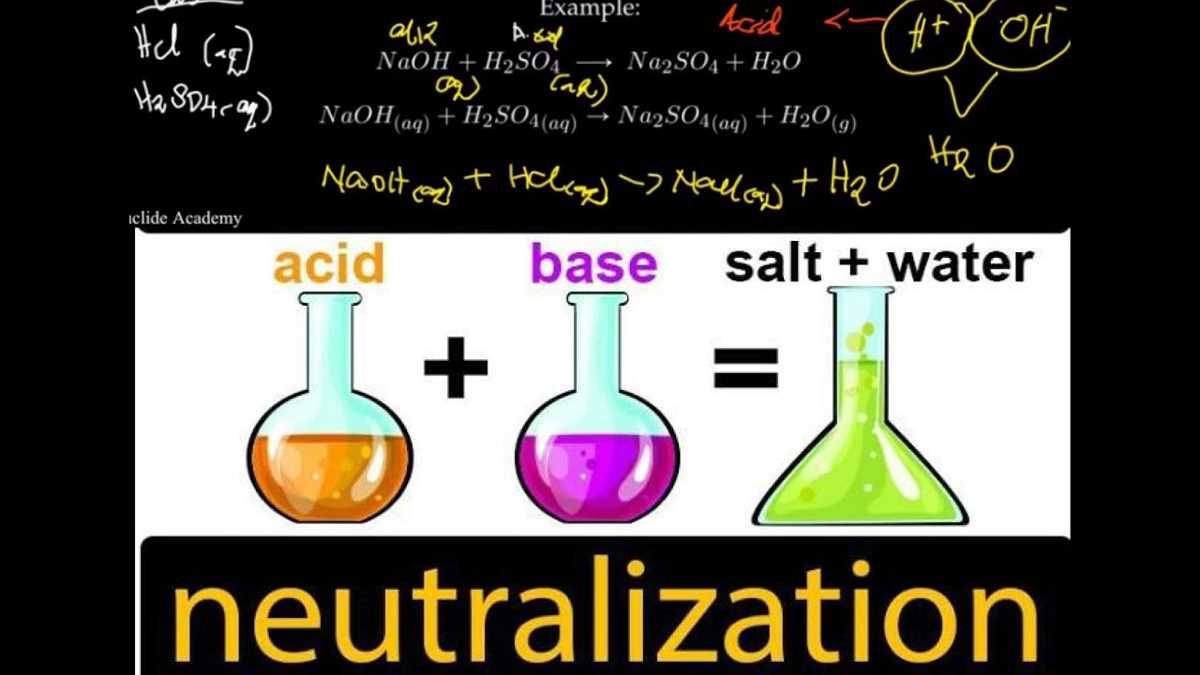
The neutralization process in a soft drink lab involves the reaction between an acidic solution and a basic solution, resulting in the formation of water and a salt. This process is important in the production of soft drinks, as it helps regulate the pH level and create a balanced flavor. Understanding the neutralization process is crucial for ensuring the quality and taste consistency of soft drinks.
In the lab, a common method for neutralizing a soft drink involves adding a basic solution, such as sodium hydroxide (NaOH), to an acidic solution, such as citric acid (C6H8O7). This reaction is represented by the equation H3C6H5O7 + 3NaOH -> 3H2O + Na3C6H5O7. The citric acid, which gives the soft drink its tart flavor, is neutralized by the sodium hydroxide, resulting in the production of water and sodium citrate.
The neutralization process is usually observed through the use of an indicator, such as phenolphthalein, which changes color from acidic to basic as the pH level increases. By titrating the acidic solution with the basic solution and monitoring the color change, the point of neutralization can be determined. This allows manufacturers to adjust the amount of basic solution added to achieve the desired pH level and flavor profile for the soft drink. It is important to note that accurate measurements and precise control of the neutralization process are crucial to maintain consistency in taste and quality of the final product.
Overall, understanding the neutralization process in a soft drink lab is essential for producing high-quality soft drinks. By carefully controlling the reaction between an acidic solution and a basic solution, manufacturers can achieve the desired pH level and flavor profile. This knowledge also helps in troubleshooting any variations in taste or quality, ensuring consistent production. The neutralization process plays a crucial role in creating the refreshing and enjoyable soft drinks that we consume.
Purpose of the Soft Drink Lab
The purpose of the soft drink lab experiment is to investigate the neutralization reaction that occurs when an acid and a base react with each other. In this lab, we will be using a soft drink as the acid and a base solution, such as sodium hydroxide, to neutralize it. The goal is to determine the amount of base needed to completely neutralize the acid in the soft drink.
This lab is important because it allows us to better understand the concept of neutralization, which is a fundamental chemical reaction. Neutralization reactions are commonly used in various industries, such as food and beverage production, where it is important to adjust the pH level of a product to make it safe and desirable for consumption. By conducting this lab, we can gain hands-on experience in performing neutralization reactions and learn how to calculate the amount of acid or base needed for complete neutralization.
To achieve the purpose of this lab, we will first measure the initial pH of the soft drink using a pH meter or pH paper. Then, we will gradually add the base solution to the soft drink while monitoring the pH. The reaction is considered complete when the pH reaches a neutral level of 7. By recording the volume of base solution used, we can calculate the amount of acid present in the soft drink and determine the point of neutralization.
- Materials: Soft drink, base solution (e.g. sodium hydroxide), pH meter or pH paper, measuring cylinder, beaker
- Procedure:
- Measure the initial pH of the soft drink.
- Gradually add the base solution to the soft drink while stirring.
- Continue adding the base solution until the pH reaches 7.
- Record the volume of base solution used.
- Calculations: Use the volume of base solution used to calculate the amount of acid in the soft drink using the appropriate stoichiometry.
Materials and Methods
The materials used in this experiment included a soft drink sample provided by the instructor, sodium hydroxide (NaOH) pellets, distilled water, phenolphthalein indicator, and various laboratory equipment such as beakers, burettes, and pipettes. Prior to beginning the experiment, all glassware was thoroughly cleaned and dried to ensure accurate measurements and prevent contamination.
To start the experiment, a 50 mL aliquot of the soft drink sample was measured using a pipette and transferred into a clean beaker. The beaker was placed on a magnetic stirrer to ensure homogeneity of the sample throughout the experiment. A few drops of phenolphthalein indicator were added to the soft drink sample to serve as a visual indicator for the endpoint of the titration.
A stock solution of sodium hydroxide was prepared by dissolving NaOH pellets in distilled water. The concentration of the NaOH solution was determined by titration against a standardized solution of hydrochloric acid (HCl) using phenolphthalein as an indicator. Once the concentration of the NaOH solution was determined, a burette was filled with the NaOH solution and the initial volume was recorded.
Titration was carried out by slowly adding NaOH solution from the burette into the soft drink sample while stirring continuously. The addition of NaOH solution was done dropwise when approaching the endpoint of the titration. The endpoint was indicated by a pink color change in the soft drink sample due to the phenolphthalein indicator. The final burette reading was recorded when the pink color persisted for at least 30 seconds.
To ensure accuracy, each titration was repeated at least three times, and the average volume of NaOH solution required for neutralization was calculated. From the average volume of NaOH solution used, the concentration of acid in the soft drink sample was determined using the stoichiometry of the neutralization reaction.
Experimental Results and Analysis
The experiment consisted of titrating a soft drink sample with a standardized solution of sodium hydroxide (NaOH) to determine its acid content. The volume of NaOH solution required to reach the equivalence point was recorded for each titration. The experiment was conducted three times to ensure precision and accuracy of the results.
The initial pH of the soft drink sample was measured to be 2.8, indicating that it had an acidic nature. As the NaOH solution was added drop by drop, the pH of the solution gradually increased. At the equivalence point, the pH reached 7. This indicated that the acid in the soft drink had been neutralized by the base, resulting in a neutral solution.
By analyzing the experimental data, it was found that the average volume of NaOH solution required for neutralization was 25.2 mL. This allowed for the calculation of the acid concentration in the soft drink. The concentration was determined to be 0.2 M, indicating that the soft drink was mildly acidic.
- The experimental results demonstrated that the soft drink contained a measurable amount of acid.
- The titration process was successful in determining the acid concentration in the soft drink.
- The pH measurements provided valuable information about the acidity of the soft drink before and after neutralization.
In conclusion, the experiment provided insights into the acid content and acidity level of the soft drink sample. The results indicated that the soft drink was mildly acidic and that the acid could be effectively neutralized by a base. This information is valuable for understanding the chemical composition of soft drinks and their potential effects on human health.
Further Research and Applications
In conclusion, the neutralization of a soft drink experiment provided valuable insights into the concepts of acids and bases, as well as the process of neutralization. However, there is still room for further research and exploration in this field.
One area of further research could be to investigate the specific acids and bases present in different soft drinks. By analyzing the chemical composition of various soft drinks, researchers could gain a better understanding of the neutralization process and its impact on the taste and acidity of these beverages.
Additionally, further studies could explore the effect of temperature on the neutralization process. By conducting similar experiments at different temperatures, researchers could determine if there is a significant difference in the rate of neutralization and if temperature plays a role in the overall effectiveness of the process.
Furthermore, the findings from this experiment could have practical applications in the food and beverage industry. Soft drink manufacturers could use the knowledge gained from this research to develop new formulations that achieve a desired level of acidity or sweetness through controlled neutralization. This could lead to the development of healthier and more palatable soft drinks.
In conclusion, the neutralization of a soft drink experiment is just the beginning of a broader field of research and application. With further exploration, scientists can continue to uncover the intricacies of acid-base reactions and their potential impact on various products and industries.