
When performing chemical reactions, it is important to determine the limiting reagent. The limiting reagent is the reactant that will be completely consumed in a reaction, thus limiting the amount of product that can be formed. To determine the limiting reagent, one must compare the mole ratios of the reactants in a balanced chemical equation.
In this worksheet, students are given a balanced chemical equation and the amounts of each reactant. They must calculate the amount of product that can be formed and identify the limiting reagent. The answer key provides the correct calculations and identifies the limiting reagent, helping students understand and practice this important concept in chemistry.
Understanding the concept of limiting reagents is crucial in chemistry, as it allows scientists and students to accurately predict the yield of a reaction and optimize the use of reactants. By using this worksheet and answer key, students can reinforce their understanding of stoichiometry and gain confidence in solving limiting reagent problems.
Understanding Limiting Reagent Worksheet Answer Key
When studying chemistry, it is essential to understand the concept of a limiting reagent. A limiting reagent is the reactant that will be completely consumed in a chemical reaction, thereby limiting the amount of product that can be formed. To determine the limiting reagent, stoichiometric calculations are often performed, and a worksheet answer key can help students practice these calculations.
The limiting reagent worksheet answer key provides the correct answers for the given problems, allowing students to check their work and understand the correct approach. The key typically includes step-by-step explanations of the calculations involved, helping students understand the underlying concepts. It also helps students identify any mistakes they may have made, allowing them to learn from their errors and improve their problem-solving skills.
Example Problem:
To illustrate how the limiting reagent worksheet answer key can be used, let’s consider the following problem: In a chemical reaction between hydrogen gas (H2) and oxygen gas (O2) to form water (H2O), calculate the limiting reagent and the amount of water produced when 3 moles of H2 reacts with 5 moles of O2. The balanced chemical equation for this reaction is:
- 2H2 + O2 → 2H2O
By using the limiting reagent worksheet answer key, students can follow the step-by-step calculations to determine the limiting reagent and calculate the amount of water produced. This allows them to practice their stoichiometry skills and gain a deeper understanding of this important concept in chemistry.
In conclusion, the limiting reagent worksheet answer key is a valuable tool for students studying chemistry. It helps them practice stoichiometry calculations, understand the concept of a limiting reagent, and learn from their mistakes. By using the answer key, students can gain confidence in their problem-solving abilities and improve their overall understanding of chemical reactions.
What is a Limiting Reagent?
A limiting reagent, also known as a limiting reactant, is a substance that is completely consumed in a chemical reaction, thereby preventing further reaction. In simple terms, it is the reactant that is present in the smallest stoichiometric amount compared to the other reactants involved. The concept of limiting reagents is crucial in determining the theoretical yield of a reaction and understanding the efficiency of the reaction.
During a chemical reaction, reactants combine to form products in specific ratios based on the balanced equation. However, the actual amounts of reactants used may not always be in those specific ratios, which leads to one reactant being used up completely before others. This reactant becomes the limiting reagent as it restricts the amount of product that can be formed. The other reactants present in excess are called excess reagents.
The identification of the limiting reagent is essential for calculating the theoretical yield of a reaction. Once the limiting reagent is determined, stoichiometric calculations can be done to find the amount of product that can be formed based on the limiting reagent. This helps in predicting the maximum amount of product that can be obtained and evaluating the efficiency of the reaction.
The concept of limiting reagents is commonly encountered in various fields such as chemistry, biology, and industrial processes. Understanding and identifying the limiting reagent is crucial for accurate calculations and optimizing reactions for desired outcomes. It allows scientists and engineers to control the reaction conditions and reagent amounts to obtain the highest possible yield of the desired product.
Importance of Limiting Reagent in Chemistry

The concept of limiting reagent is crucial in chemistry as it helps determine the maximum amount of product that can be formed in a chemical reaction. A limiting reagent is the reactant that gets completely consumed during the reaction, limiting the amount of product that can be produced. It plays a key role in determining the efficiency and yield of a reaction, as well as in understanding the stoichiometry of the reaction.
Identifying the limiting reagent allows chemists to calculate the theoretical yield of a reaction, which is the maximum amount of product that can be obtained under ideal conditions. This information is important in practical applications, such as drug synthesis, where the yield of the desired product needs to be maximized in order to minimize costs and waste. By knowing the limiting reagent, chemists can adjust the reactant ratios in order to optimize the yield of the desired product.
The concept of limiting reagent is also important in understanding the stoichiometry of a chemical reaction. It allows chemists to determine the molar ratios between reactants and products, which is essential for balancing chemical equations and predicting the outcomes of reactions. Without the concept of limiting reagent, it would be difficult to accurately determine the amounts of reactants and products involved in a chemical reaction, making it challenging to perform calculations and analyze the reaction.
In summary, the concept of limiting reagent is crucial in chemistry as it helps determine the maximum amount of product that can be formed in a reaction, allows for the calculation of theoretical yield, and aids in understanding the stoichiometry of the reaction. It is an essential tool for chemists in optimizing yields, predicting reaction outcomes, and performing accurate calculations in various chemical processes.
Limiting Reagent Worksheet Questions
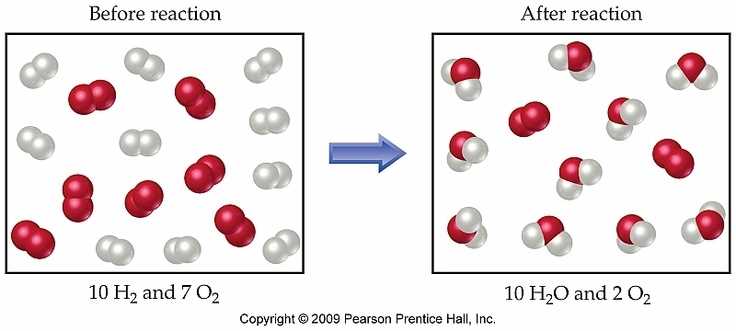
In chemistry, the concept of limiting reagents is essential to understand the outcome of a reaction. A limiting reagent is a reactant that limits the amount of product that can be formed. It is the reactant that is completely consumed in the reaction, thus determining the amount of product that can be obtained.
In a limiting reagent worksheet, students are provided with a reaction equation and given the amounts of reactants involved. They are then asked to determine which reactant is the limiting reagent and calculate the amount of product that can be formed. This concept is crucial for understanding stoichiometry and predicting yield in chemical reactions.
The worksheet questions usually involve multiple steps and require students to use their knowledge of molar ratios, balanced equations, and stoichiometry. They may also require conversions between different units, such as grams to moles or moles to liters. Students need to carefully analyze the given information and use it to calculate the theoretical yield of the product.
By working on limiting reagent worksheet questions, students develop their problem-solving skills and gain a deeper understanding of chemical reactions. They learn to identify the limiting reagent by comparing the molar amounts of the reactants and use stoichiometry to calculate the theoretical yield. This practice helps them become more proficient in performing calculations and making predictions in chemistry.
How to Solve Limiting Reagent Problems
When dealing with a chemical reaction, it is important to determine the limiting reagent, which is the reactant that will be completely consumed first and limits the amount of product that can be formed. To solve limiting reagent problems, follow these steps:
- Write out the balanced chemical equation: Start by writing out the balanced chemical equation for the reaction. This equation shows the reactants and products involved in the reaction.
- Calculate the moles of each reactant: Convert the given quantities of each reactant into moles. This can be done by dividing the mass of each reactant by its molar mass.
- Determine the mole ratio: Use the coefficients in the balanced equation to determine the mole ratio of the reactants. This ratio gives the number of moles of one reactant that is required to react with a certain number of moles of the other reactant.
- Identify the limiting reagent: Compare the moles of each reactant to the mole ratio. The reactant with the smaller number of moles is the limiting reagent, as it will be completely consumed first in the reaction.
- Calculate the amount of product: Once the limiting reagent is determined, use its moles to calculate the moles of product that can be formed. This can be done using the mole ratio from the balanced equation.
Solving limiting reagent problems requires a good understanding of stoichiometry and the ability to balance chemical equations. It is important to carefully follow these steps to ensure accurate results. By identifying the limiting reagent, one can determine the maximum amount of product that can be obtained in a reaction, which is crucial for calculating reaction yields and determining the efficiency of a chemical process.
Step-by-Step Guide to Using the Limiting Reagent Worksheet Answer Key

When working with limiting reagents in chemistry, it can be helpful to use a worksheet and an answer key to ensure accuracy and understanding. Here is a step-by-step guide to using the limiting reagent worksheet answer key effectively:
1. Read the problem carefully: Start by reading the problem and identifying the reactants and products involved. Take note of any information given about the quantities of reactants and the desired quantities of products.
2. Identify the limiting reagent: Determine which reactant is the limiting reagent, meaning it will be completely consumed in the reaction and limit the amount of product formed. This can be done by comparing the mole ratios of the reactants and the desired mole ratio of products.
3. Use the limiting reagent worksheet: Open the limiting reagent worksheet and locate the problem you are working on. Each problem on the worksheet will have a corresponding answer key.
4. Plug in the given values: Fill in the given values from the problem into the worksheet. This may include the initial quantities of reactants, the molar mass of each reactant, and the desired quantities of products.
5. Calculate the mole ratios: Use the coefficients from the balanced chemical equation to calculate the mole ratios of the reactants and products. This step is crucial in determining the limiting reagent.
6. Compare the mole ratios: Compare the calculated mole ratios to determine which reactant is the limiting reagent. The limiting reagent will be the one with the lowest mole ratio.
7. Calculate the quantities of products: Use the mole ratios and the initial quantities of the limiting reagent to calculate the quantities of products that can be formed.
8. Check the answer key: Once you have completed the calculations on the worksheet, refer to the answer key to verify your answers. Compare your results to the answers provided to ensure accuracy.
9. Review and understand: Take the time to review the answer key and understand the steps and calculations used to arrive at the correct answers. This will help reinforce your understanding of limiting reagents and their role in chemical reactions.
By following this step-by-step guide and using the limiting reagent worksheet answer key, you can effectively practice and master the concept of limiting reagents in chemistry.
Examples and Practice Problems
Now that we have discussed the concept of limiting reagents and how to calculate them, let’s look at some examples and practice problems to reinforce our understanding.
Example 1:
Consider the reaction:
2 H2 + O2 -> 2 H2O
If we have 4 moles of H2 and 3 moles of O2, how many moles of water can we produce?
To solve this problem, we first need to determine the limiting reagent. We can do this by comparing the mole ratios of the reactants and the coefficients in the balanced equation:
- For H2: 4 moles / 2 = 2 moles of water
- For O2: 3 moles / 1 = 3 moles of water
Since we can only produce a maximum of 2 moles of water from 4 moles of H2, H2 is the limiting reagent. Therefore, we will form 2 moles of water.
Practice Problem 1:
Consider the reaction:
2 H2 + O2 -> 2 H2O
If we have 6 moles of H2 and 4 moles of O2, how many moles of water can we produce?
Using the same process as before, we find:
- For H2: 6 moles / 2 = 3 moles of water
- For O2: 4 moles / 1 = 4 moles of water
Since we can only produce a maximum of 3 moles of water from 6 moles of H2, H2 is the limiting reagent. Therefore, we will form 3 moles of water.
Through these examples and practice problems, we can see that identifying the limiting reagent allows us to determine the maximum amount of product that can be formed in a chemical reaction. This information is crucial for understanding reaction stoichiometry and calculating chemical yields.