Understanding intermolecular forces is crucial in explaining the properties and behavior of different substances. These forces are the attractive forces that exist between molecules, and they play a significant role in determining the physical properties of substances such as boiling point, melting point, and solubility. To better understand these intermolecular forces, practice worksheets with answer keys can provide valuable insights.
Intermolecular forces worksheets typically consist of various types of questions that require students to analyze and apply their knowledge of these forces. These questions may involve identifying the type of intermolecular force present in a given substance, comparing the strength of different forces, explaining the effect of intermolecular forces on physical properties, or predicting the behavior of substances based on their intermolecular forces.
An answer key for an intermolecular forces worksheet provides students with the correct solutions and explanations. This key allows students to check their answers and understand any mistakes they may have made. It also serves as a useful tool for teachers, assisting them in grading and providing feedback on students’ work.
When using an intermolecular forces worksheet with an answer key, it is important for students to carefully review their answers and understand the reasoning behind each solution. This process helps solidify their understanding of intermolecular forces and their effects on substances. Additionally, practicing with these worksheets and answer keys enhances students’ problem-solving skills and prepares them for more challenging questions or real-life situations that involve intermolecular forces.
Intermolecular Forces Worksheet Answer Key: Exploring the World of Chemistry
Chemistry is a fascinating subject that allows us to understand the world at a molecular level. It helps explain why certain substances behave the way they do and how different molecules interact with each other. One important concept in chemistry is intermolecular forces, which are the forces between molecules that determine their physical properties.
The Intermolecular Forces Worksheet provides students with an opportunity to explore and understand these forces in depth. The answer key for this worksheet is an invaluable resource that allows students to check their understanding and ensure they are on the right track. It provides step-by-step solutions and explanations for each question, helping students grasp the underlying concepts effectively.
The answer key includes explanations for various types of intermolecular forces, such as hydrogen bonding, dipole-dipole interactions, and London dispersion forces. Each force is discussed in detail, with examples provided to illustrate their effects on different substances. This comprehensive approach enables students to see the relevance of these forces in real-life situations and how they contribute to the properties of substances.
In addition to the explanations, the answer key also includes practice problems that allow students to apply their knowledge and test their understanding. These problems cover a range of topics, from identifying the type of intermolecular forces present in a substance to predicting the boiling points of different compounds. The detailed solutions provided in the answer key help students grasp the concepts more effectively and build their problem-solving skills.
The Intermolecular Forces Worksheet Answer Key is not just a tool for students; it is also a valuable resource for teachers. It allows teachers to assess their students’ understanding, identify areas that need further clarification, and provide targeted support. With this answer key, teachers can ensure that their students have a strong foundation in understanding intermolecular forces, setting them up for success in their future studies in chemistry and related fields.
Understanding the Types of Intermolecular Forces
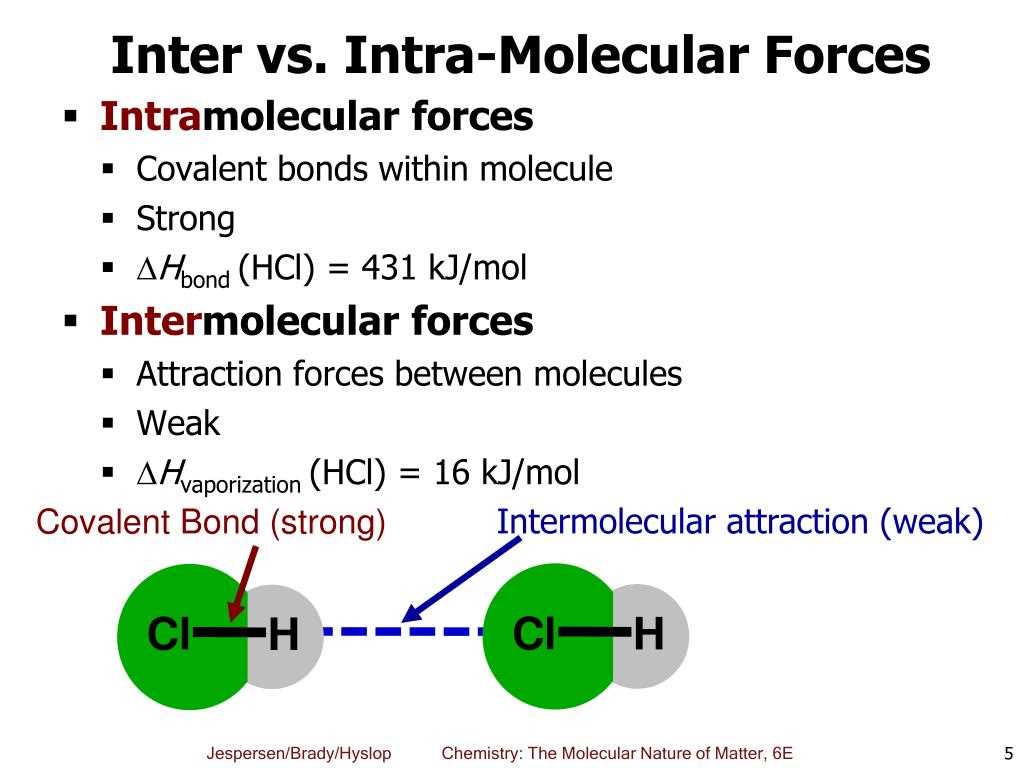
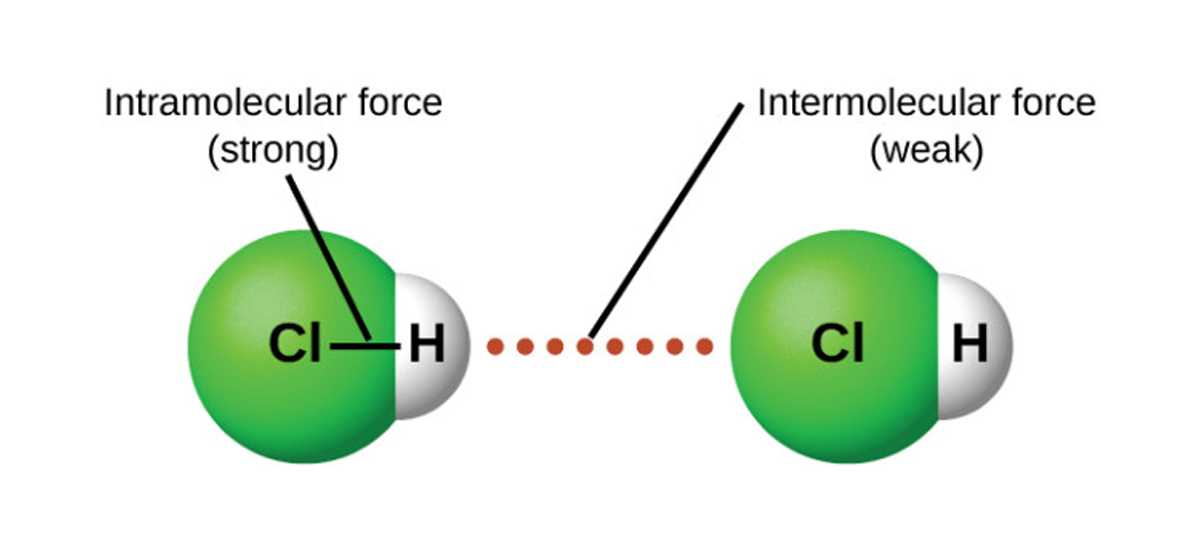
The behavior of molecules in different states of matter is influenced by intermolecular forces. Intermolecular forces are attractions between molecules that determine their physical properties, such as boiling point, melting point, and solubility. It is crucial to understand the different types of intermolecular forces to comprehend how substances interact at the molecular level.
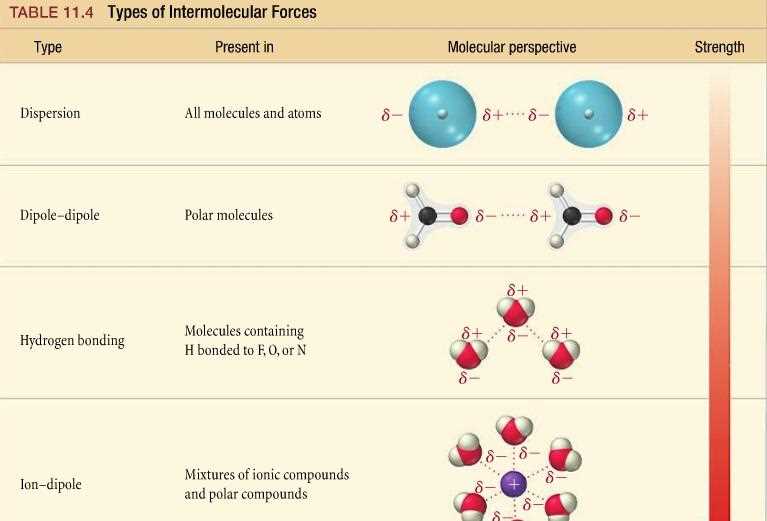
There are three main types of intermolecular forces: dispersion forces, dipole-dipole forces, and hydrogen bonding. Dispersion forces, also known as London forces, occur in all molecules. They arise from temporary fluctuations in electron distribution that create an instantaneous dipole. These temporary dipoles can induce dipoles in neighboring molecules, leading to attractive forces. Dispersion forces increase with the size of the molecule and its shape, as more electrons are available to create temporary dipoles.
Dipole-dipole forces exist in polar molecules due to the unequal distribution of electrons. The positive end of one polar molecule attracts the negative end of another polar molecule, creating an electrostatic attraction. These forces are stronger than dispersion forces and can significantly impact the physical properties of substances. The strength of dipole-dipole forces depends on the magnitude of the dipole moment and the distance between the molecules.
Hydrogen bonding is a special type of dipole-dipole force that occurs when a hydrogen atom is covalently bonded to an electronegative element, such as oxygen, nitrogen, or fluorine. The hydrogen atom carries a partial positive charge, while the electronegative element carries a partial negative charge. This configuration allows for a strong electrostatic attraction between the hydrogen atom of one molecule and the electronegative element of another molecule. Hydrogen bonding is responsible for the unique properties of substances like water, which has a higher boiling point due to the presence of numerous hydrogen bonds.
In summary, understanding the types of intermolecular forces is essential for comprehending the behavior of substances in different states of matter. Dispersion forces, dipole-dipole forces, and hydrogen bonding all play significant roles in determining physical properties and interactions between molecules.
Key Concepts for Interpreting Intermolecular Forces Worksheet

When interpreting the intermolecular forces worksheet, it is important to understand several key concepts related to intermolecular forces.
1. Intermolecular forces: These are the forces of attraction between molecules. They are responsible for determining the physical properties of substances such as boiling points, melting points, and solubility.
2. Types of intermolecular forces: The worksheet may cover different types of intermolecular forces, including London dispersion forces, dipole-dipole interactions, and hydrogen bonding. Each type of force has specific characteristics and strength based on the polarity and size of the molecules involved.
3. Molecular structure and intermolecular forces: The structure of a molecule plays a crucial role in determining the type and strength of intermolecular forces. Molecules with polar bonds or asymmetrical shapes are more likely to exhibit stronger intermolecular forces.
4. Relationship with physical properties: Understanding intermolecular forces provides insight into various physical properties of substances. For example, molecules with strong intermolecular forces tend to have higher boiling points, while those with weak forces have lower boiling points.
5. Comparing intermolecular forces: The worksheet may require comparisons between different substances and their intermolecular forces. It is important to analyze the molecular structure, polarity, and size of molecules to determine the relative strength of the intermolecular forces present.
6. Real-world applications: Intermolecular forces have practical implications in various fields, such as pharmaceuticals, materials science, and environmental science. Understanding these forces allows scientists to design and optimize substances for specific applications.
In summary, interpreting the intermolecular forces worksheet involves grasping the fundamental concepts related to intermolecular forces, understanding the different types of forces, analyzing molecular structures, relating forces to physical properties, making comparisons, and recognizing the real-world applications of these forces.
London Dispersion Forces in Detail
London Dispersion Forces are a type of intermolecular force that occurs between non-polar molecules. These forces are also known as dispersion forces or instantaneously induced dipoles. They are the weakest intermolecular force but can still have a significant impact on the physical properties of substances.
The London Dispersion Forces arise due to temporary fluctuations in the electron cloud around atoms or molecules. At any given moment, there may be an uneven distribution of electrons, creating a temporary dipole moment. The temporary dipole induces neighboring molecules to develop a complementary temporary dipole, resulting in a weak attractive force between the molecules.
This type of intermolecular force is present in all molecules, but its strength depends on the size of the electron cloud and the shape of the molecules. Larger molecules with more electrons have a greater chance of experiencing temporary fluctuations in their electron distribution, leading to stronger London Dispersion Forces. Additionally, molecules with a larger surface area or a more elongated shape can have stronger London Dispersion Forces due to the increased likelihood of electron fluctuations.
London Dispersion Forces play a significant role in determining the boiling points, melting points, and viscosity of substances. The greater the strength of the London Dispersion Forces, the higher the boiling point and melting point of the substance. This is because more energy is required to break the intermolecular forces between molecules. In substances with similar molecular sizes and shapes, the strength of the London Dispersion Forces can be used to predict their relative boiling points.
In summary, London Dispersion Forces are weak attractive forces that arise due to temporary fluctuations in the electron cloud around atoms or molecules. These forces are present in all molecules but are stronger in larger molecules or molecules with a greater surface area. They play a significant role in determining the physical properties of substances, such as boiling points and melting points.
Definition and Properties of London Dispersion Forces

London dispersion forces, also known as dispersion forces or induced dipole-induced dipole interactions, are a type of intermolecular force that exists between all types of molecules. These forces are the weakest of the intermolecular forces and occur due to temporary fluctuations in electron density within atoms or molecules.
London dispersion forces arise from the instantaneous polarization of electron clouds. Even though molecules may not have a permanent dipole moment, at any given moment, there could be an uneven distribution of electrons, resulting in a temporary dipole moment. This temporary dipole then induces a dipole in a neighboring molecule, creating an attractive force between the two molecules.
London dispersion forces are responsible for many physical properties of molecules, including boiling points, melting points, and solubility in nonpolar solvents. These forces increase with increasing molecular size and shape and are more significant for molecules with larger electron clouds. This is because larger electron clouds are more easily polarized and can induce larger temporary dipoles in nearby molecules.
Furthermore, London dispersion forces can also occur between nonpolar molecules and polar molecules. In these cases, the temporary dipole in the nonpolar molecule can induce a dipole in the polar molecule, resulting in an attractive force. However, these forces are usually weaker than the forces between polar molecules, such as dipole-dipole interactions or hydrogen bonds.
In summary, London dispersion forces are weak intermolecular forces that arise due to temporary fluctuations in electron density. They play a crucial role in determining physical properties of molecules and can occur between all types of molecules, including both polar and nonpolar molecules.
Examples of London Dispersion Forces in Action
The London dispersion force is a type of intermolecular force that occurs between nonpolar molecules. It is caused by temporary fluctuations in the electron distribution within a molecule, which can lead to the formation of temporary dipoles. These temporary dipoles induce similar dipoles in neighboring molecules, resulting in attractive forces between them.
London dispersion forces can be observed in various everyday examples. One example is the condensation of gases into liquids. When a gas is cooled, the molecules slow down and come closer together, increasing the opportunities for London dispersion forces to act. As a result, the gas molecules can attract each other, leading to the formation of a liquid.
- In the case of hydrocarbons, such as methane (CH4), London dispersion forces play a significant role. Hydrocarbons are nonpolar molecules with only C-H and C-C bonds. The fluctuations in the electron distribution within these molecules can induce temporary dipoles, causing attractive forces between adjacent hydrocarbon molecules.
- Another example is the interaction between noble gases, such as helium (He) and neon (Ne). Noble gases are known for their low reactivity due to their stable electron configurations. However, even though noble gases are technically nonpolar, they can still exhibit London dispersion forces. The temporary fluctuations in their electron distribution can induce temporary dipoles, allowing them to attract each other.
In summary, London dispersion forces are present in various substances and play an essential role in intermolecular interactions. They are particularly noticeable in nonpolar molecules, such as hydrocarbons and noble gases. These forces contribute to the condensation of gases into liquids and can influence the physical properties of substances.
Dipole-Dipole Interactions: The Power of Polarity
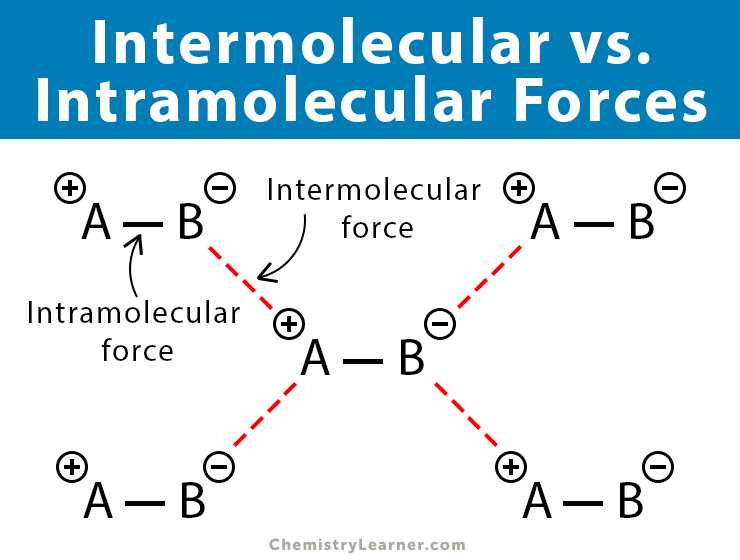
In the realm of intermolecular forces, dipole-dipole interactions play a significant role in determining the physical properties of substances. These interactions occur between molecules that possess a permanent dipole moment, meaning they have a separation of positive and negative charges within the molecule. This polarity arises due to differences in electronegativity between the atoms within the molecule.
As the name suggests, dipole-dipole interactions involve the attraction between the positive end of one dipole and the negative end of another. This attraction is relatively strong, resulting in higher boiling points, melting points, and surface tensions compared to substances without dipole-dipole interactions. These intermolecular forces also contribute to the unique properties of compounds such as water, which has high surface tension and boiling point due to its strong dipole-dipole interactions.
The strength of dipole-dipole interactions depends on the magnitude of the dipole moment and the proximity of the molecules. When the dipole moment is larger, the forces are stronger, resulting in higher melting and boiling points. Additionally, the proximity of the molecules plays a role in these interactions. Molecules that are closer together will experience stronger dipole-dipole interactions compared to those that are farther apart.
Understanding dipole-dipole interactions is crucial in predicting and explaining the behavior of various substances. By recognizing the presence and strength of these interactions, scientists can better comprehend the physical and chemical properties of compounds and develop applications in fields such as materials science, pharmaceuticals, and environmental research.