
Electrochemistry is a branch of chemistry that deals with the study of the relationship between electricity and chemical reactions. It is a fundamental topic in chemistry and is widely used in various industries, including energy storage, corrosion prevention, and electroplating. To fully grasp the concepts of electrochemistry, it is essential to practice solving problems to apply the principles learned in theory.
This article provides a collection of electrochemistry practice problems with answers in PDF format. These practice problems cover various topics, including redox reactions, electrochemical cells, Faraday’s laws of electrolysis, and galvanic cells. Each problem is designed to test your understanding of the key concepts and principles in electrochemistry and to help you improve your problem-solving skills.
By practicing these problems and reviewing the provided answers, you can enhance your knowledge of electrochemistry and strengthen your ability to solve similar problems in exams or real-life situations. Whether you are a student studying chemistry or a professional working in a related field, these practice problems will serve as a valuable resource to reinforce your understanding of electrochemistry.
Electrochemistry Practice Problems with Answers PDF
Electrochemistry is a fundamental branch of chemistry that deals with the study of the interconversion of chemical and electrical energy. It involves the study of redox reactions, where one species undergoes oxidation and another species undergoes reduction. To master the concepts of electrochemistry, it is important to practice solving various electrochemistry problems.
If you are looking for electrochemistry practice problems with answers in a convenient PDF format, you’re in luck. There are several resources available online that provide electrochemistry practice problems with detailed solutions in PDF format. These resources usually contain a collection of multiple-choice questions, numerical problems, and conceptual questions to test your understanding of electrochemical principles.
The electrochemistry practice problems cover topics such as electrode potentials, electrolysis, Faraday’s laws, Nernst equation, and cell potential calculations. By solving these practice problems, you can enhance your problem-solving skills, improve your conceptual understanding, and ultimately excel in electrochemistry.
One popular resource for electrochemistry practice problems with answers in PDF format is the website ChemGuide. They offer a comprehensive set of electrochemistry practice questions that cover a wide range of topics. Another excellent resource is the Electrochemistry Practice Problems PDF provided by the University of Oregon’s Chemistry Department. This PDF contains a variety of practice problems with detailed explanations and answers.
Remember, practice is key to mastering any subject, and electrochemistry is no exception. By regularly practicing electrochemistry problems, you will become more proficient in solving these types of problems and gain a deeper understanding of the principles behind electrochemistry.
Basics of Electrochemistry

Electrochemistry is the branch of chemistry that deals with the relationship between electricity and chemical reactions. It studies the movement of electrons and ions in order to understand how oxidation-reduction reactions occur.
Electrochemical cells are devices that convert chemical energy into electrical energy through redox reactions. They consist of two electrodes – the anode and the cathode – that are placed in an electrolyte solution. The anode is where oxidation occurs, producing electrons, while the cathode is where reduction occurs, consuming electrons. These electrons flow through an external circuit, creating an electric current.
Electrolytes are substances that conduct electricity when dissolved in a solvent. They are often ionic compounds or solutions of acids and bases. Electrolytes provide ions that move towards the electrodes, completing the circuit. The movement of these ions allows the transfer of charge between the two electrodes, enabling the redox reactions to take place.
In electrochemical cells, electrons travel from the anode to the cathode, balancing the flow of ions. This flow of electrons can be harnessed to perform useful work, such as powering devices or storing energy. Different types of electrochemical cells, such as batteries, fuel cells, and electroplating cells, have specific designs and reactions that allow them to fulfill specific functions.
Redox reactions are at the core of electrochemistry. These reactions involve the transfer of electrons from one species to another. The species that loses electrons is oxidized, while the species that gains electrons is reduced. The transfer of electrons is accompanied by the movement of ions, resulting in a balanced overall reaction.
Understanding the basics of electrochemistry is crucial in many areas of science and technology. It is used in fields such as energy storage, corrosion prevention, electroplating, and even in biological processes like nerve impulses. By studying electrochemical reactions and their principles, scientists and engineers can develop more efficient and sustainable technologies.
Redox Reactions and Balancing Equations
A redox reaction is a type of chemical reaction in which the oxidation states of atoms are changed. It involves the transfer of electrons between species, where one species is oxidized (loses electrons) and another species is reduced (gains electrons). To balance redox reactions, one must ensure that the number of electrons lost in oxidation is equal to the number of electrons gained in reduction.
When balancing redox equations, it is helpful to follow a step-by-step process. First, identify the species that are oxidized and reduced in the reaction. Next, assign oxidation numbers to each element in the species. Then, write separate half-reactions for the oxidation and reduction processes. Balance the atoms in each half-reaction by adding coefficients as needed. Finally, balance the charges on both sides of the equation by adding electrons to one side or the other.
To illustrate this process, let’s consider the redox reaction between sodium (Na) and chlorine (Cl2) to form sodium chloride (NaCl). The oxidation state of sodium changes from 0 to +1, indicating oxidation, while the oxidation state of chlorine changes from 0 to -1, indicating reduction. The balanced equation for this reaction is:
| 2Na + Cl2 | → | 2NaCl |
In this equation, the sodium atoms are oxidized, and the chlorine molecules are reduced. The equation is balanced because the number of Na atoms, Cl atoms, and charges are equal on both sides.
By understanding the principles of redox reactions and how to balance equations, electrochemical problems involving redox reactions can be solved more effectively. Practice problems and exercises can further enhance understanding and proficiency in this area of electrochemistry.
Electrochemical Cells and Notation

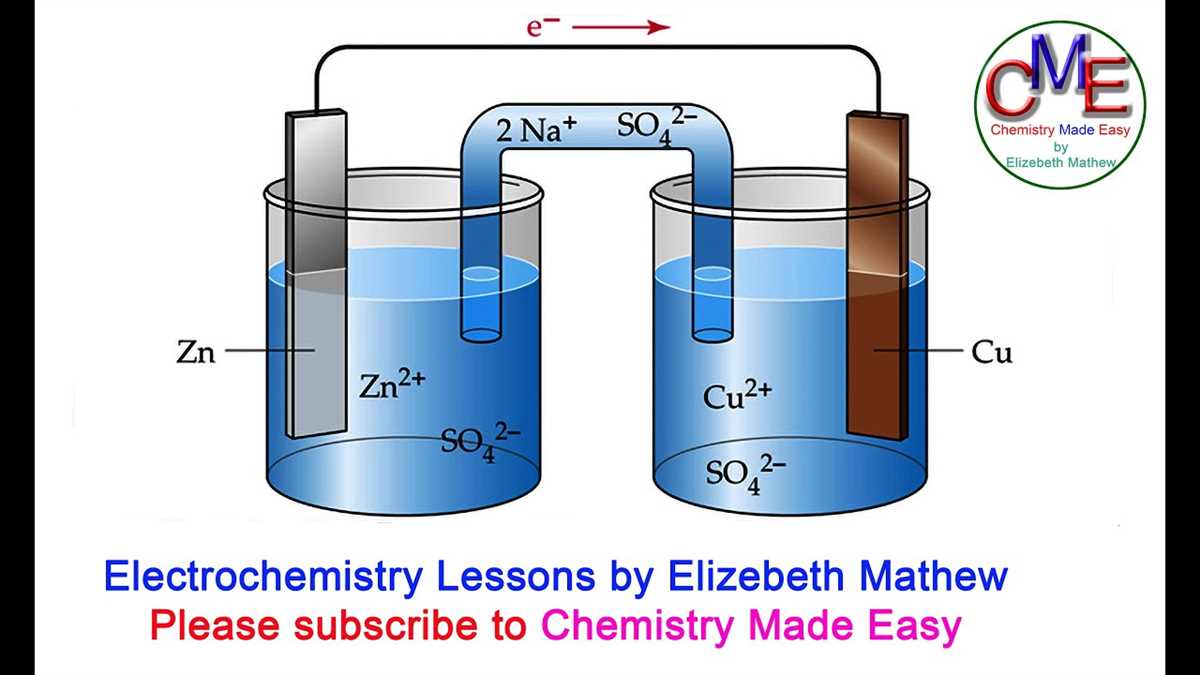
Electrochemical cells are devices that convert chemical energy into electrical energy through redox reactions. They consist of two half-cells, each containing an electrode immersed in an electrolyte solution. These half-cells are connected by a salt bridge or porous barrier, allowing for the movement of ions between them. The half-cell where oxidation occurs is called the anode, while the half-cell where reduction occurs is called the cathode.
Standard notation: The standard notation for representing an electrochemical cell consists of two electrodes separated by a double vertical line. The anode is written on the left side, followed by a single vertical line and the solution containing the anode. On the right side of the double vertical line, the cathode is written, followed by a single vertical line and the solution containing the cathode. For example, a cell with a zinc anode and a copper cathode in solutions of their respective ions would be represented as:
Zn(s) | Zn2+(aq) || Cu2+(aq) | Cu(s)
In this notation, the solid electrode is always written on the far left or far right side, indicating that it is not in direct contact with the electrolyte solution.
Cell potential: The cell potential, often denoted as Ecell or E°cell, is a measure of the ability of a cell to produce an electric current. It is determined by the difference in the reduction potentials of the two half-reactions occurring at the electrodes. The cell potential can be calculated using the Nernst equation:
Ecell = E°cell - (0.05916/n) log(Q)
Where E°cell is the standard cell potential, n is the number of moles of electrons transferred in the balanced equation, and Q is the reaction quotient.
- Electrolytic cells: Electrolytic cells are electrochemical cells in which electrical energy is used to drive a non-spontaneous redox reaction. They have a positive cell potential and require an external power source to operate. The anode is still the site of oxidation, and the cathode is still the site of reduction.
- Galvanic cells: Galvanic cells, on the other hand, are electrochemical cells in which a spontaneous redox reaction produces electrical energy. They have a negative cell potential and do not require an external power source. The anode is where oxidation occurs, and the cathode is where reduction occurs.
Calculating Cell Potential
In electrochemistry, the cell potential is a measure of the ability of a redox reaction to produce an electric current. It is a fundamental concept in understanding the behavior of electrochemical cells. The cell potential, also known as the electromotive force (EMF) or voltage, is calculated using the standard reduction potentials of the half-reactions involved in the cell.
To calculate the cell potential, you first need to identify the half-reactions occurring in the cell. Each half-reaction consists of a reduction reaction and an oxidation reaction. The half-reaction with the more positive standard reduction potential is the reduction half-reaction, while the other half-reaction is the oxidation half-reaction.
The cell potential is then calculated by subtracting the reduction potential of the oxidation half-reaction from the reduction potential of the reduction half-reaction. The resulting cell potential represents the potential difference between the two electrodes of the electrochemical cell.
It is important to note that the cell potential is dependent on the concentrations of the reactants and products involved in the redox reaction. In electrochemical cells, the concentrations are typically specified as 1 M for all species except for gases, which are typically specified at 1 atm pressure. Additionally, the cell potential is also affected by temperature, so it is important to take into account the appropriate temperature dependence when calculating the cell potential.
Overall, calculating the cell potential is an essential step in understanding the behavior of electrochemical cells. It allows scientists and engineers to determine the feasibility and efficiency of electrochemical reactions, and plays a crucial role in various applications such as batteries, fuel cells, and corrosion prevention.
Nernst Equation and Concentration Cells
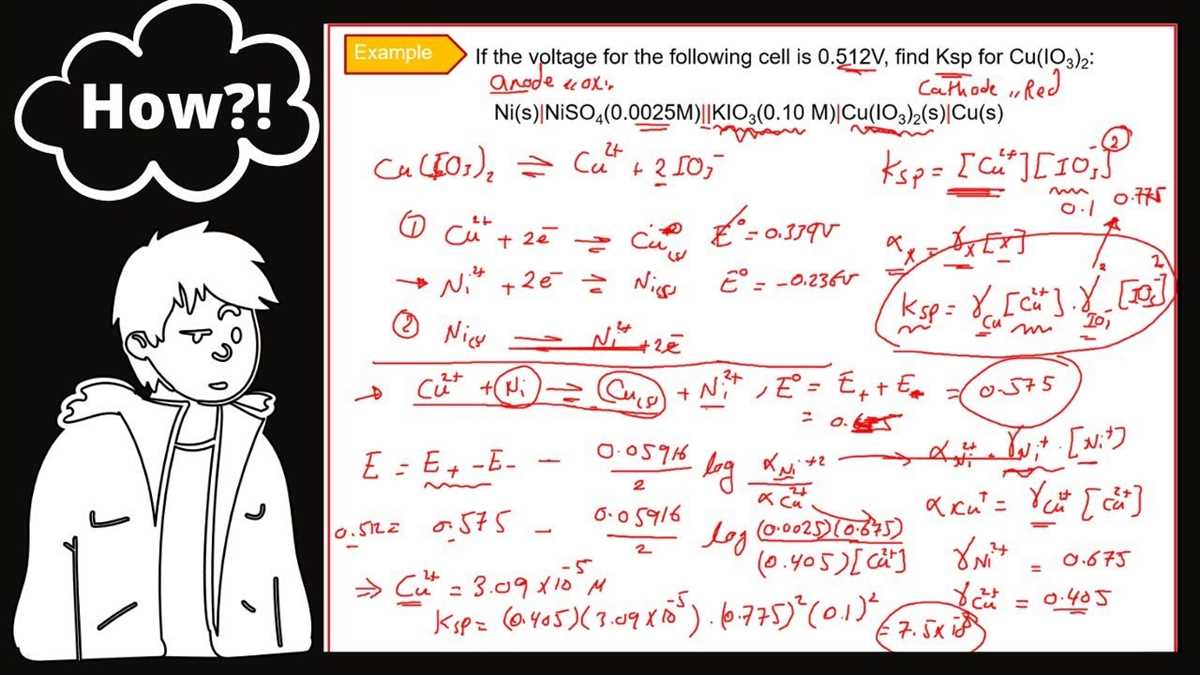
The Nernst equation is an important equation in electrochemistry that relates the potential difference between two electrodes (E) to the concentration of species involved in the redox reaction. It is named after the German scientist Walther Nernst, who formulated the equation.
The Nernst equation is represented as follows:
E = E° – (RT/nF)ln(Q)
Where:
- E is the electric potential difference between the two electrodes
- E° is the standard electrode potential
- R is the ideal gas constant (8.314 J·K^-1·mol^-1)
- T is the temperature in Kelvin
- n is the number of electrons transferred in the redox reaction
- F is Faraday’s constant (96485 C/mol)
- ln denotes the natural logarithm
- Q is the reaction quotient, which is the ratio of the concentrations of the products to the concentrations of the reactants
The Nernst equation can be used to calculate the potential difference (E) at non-standard conditions, where the concentrations of the species involved in the reaction are not at their standard state. It allows us to determine how changing the concentrations of the species will impact the potential difference between the electrodes.
Concentration cells are electrochemical cells that utilize the Nernst equation. They consist of two half-cells with different concentrations of the same ion. The potential difference between the two half-cells is generated based on the concentration gradient of the ion. This type of cell is commonly used in pH measurements, as it allows for accurate determination of the pH of a solution.
Overall, the Nernst equation and concentration cells play a crucial role in understanding and predicting the behavior of electrochemical reactions. They provide a mathematical framework for calculating the potential difference under non-standard conditions and offer insights into the relationship between concentrations and electrochemical processes.
Electrolysis and Faraday’s Laws
In electrochemistry, electrolysis is a process that involves the use of an electric current to drive a non-spontaneous chemical reaction. It is used in various industrial applications, such as metal plating, purification of metals, and production of chemicals.
Faraday’s Laws of Electrolysis, formulated by Michael Faraday in the 19th century, provide a mathematical relationship between the amount of substance reacted at an electrode and the quantity of electricity passed through the electrolyte. These laws are fundamental to understanding and predicting the outcome of electrolysis reactions.
Faraday’s First Law: The amount of substance converted or produced at an electrode during electrolysis is directly proportional to the quantity of electricity passed through the electrolyte.
Faraday’s Second Law: The mass of different substances produced at the same electrode by the passage of the same quantity of electricity is directly proportional to their chemical equivalents. The chemical equivalent is the mass of a substance that reacts with or is produced from one mole of electrons.
These laws can be expressed mathematically using Faraday’s constant, which is the charge of one mole of electrons. The charge Q (in coulombs) passed through the electrolyte can be related to the amount of substance converted or produced (mass or moles) using the equations:
Q = nF
m = ZFn
Where Q is the charge passed, n is the number of moles of electrons transferred, F is Faraday’s constant, m is the mass of substance produced or converted, Z is the electrochemical equivalent (mass of substance produced per mole of electrons), and Fn is the product of Faraday’s constant and the number of moles of electrons transferred.
By applying Faraday’s Laws, scientists and engineers can determine the efficiency of an electrolysis process and calculate the amount of reactants and products involved. These laws are crucial in optimizing industrial electrolysis processes and ensuring reliable and sustainable electrochemical reactions.
In conclusion, electrolysis and Faraday’s Laws play a vital role in electrochemical processes. They provide a fundamental framework for understanding and predicting the outcome of electrolysis reactions. By using these laws, scientists and engineers can optimize industrial processes and contribute to advancements in various fields, including metallurgy, chemical production, and environmental sustainability.
Q&A:
What is electrolysis?
Electrolysis is a chemical process that uses an electric current to drive a non-spontaneous chemical reaction. It involves the breaking down of a compound into its individual elements using electricity.
What are Faraday’s laws of electrolysis?
Faraday’s laws of electrolysis are two quantitative relationships that describe the amount of substances produced or consumed during electrolysis. The first law states that the amount of substance produced at an electrode is directly proportional to the amount of charge passed through the electrolyte. The second law states that the amounts of different substances produced or consumed at electrodes are in a simple integer ratio.
What is Faraday’s first law equation?
Faraday’s first law equation is Q = nF, where Q is the total charge passed through the electrolyte, n is the number of moles of the substance produced or consumed, and F is the Faraday constant. The value of the Faraday constant is approximately 96485 C/mol.
What is electrolytic cell?
An electrolytic cell is a type of electrochemical cell that uses an external electrical source to drive a non-spontaneous chemical reaction. It consists of two electrodes – an anode and a cathode – immersed in an electrolyte, which is a conductive solution or melt. The anode is the positive electrode, where oxidation occurs, and the cathode is the negative electrode, where reduction occurs.