The development of atomic theory has been a fascinating journey that has spanned centuries and involved the contributions of numerous scientists. From the ancient Greeks to the modern day, our understanding of atoms and their structure has evolved significantly.
One of the earliest atomic theories can be attributed to the Greek philosopher Democritus, who proposed that all matter is made up of indivisible particles called atoms. However, it wasn’t until the 19th century that advancements in technology and experimentation allowed for further exploration of atomic theory.
John Dalton, an English chemist, is often credited as the founder of modern atomic theory. In the early 1800s, he proposed that atoms are indivisible and that all atoms of a particular element are identical. His theory also included the concept of chemical reactions as a rearrangement of atoms, and a law of multiple proportions.
Over time, new evidence and discoveries challenged Dalton’s original theories. For example, the development of the cathode ray tube and the discovery of the electron by J.J. Thomson in the late 19th century brought about a new understanding of atomic structure. Thomson proposed the “plum pudding” model, in which electrons were embedded in a positively charged sphere.
These are just a few of the key developments in atomic theory, and today our understanding of atoms continues to expand. The study of atomic theory answers the fundamental questions about the building blocks of matter and provides a foundation for many areas of science and technology.
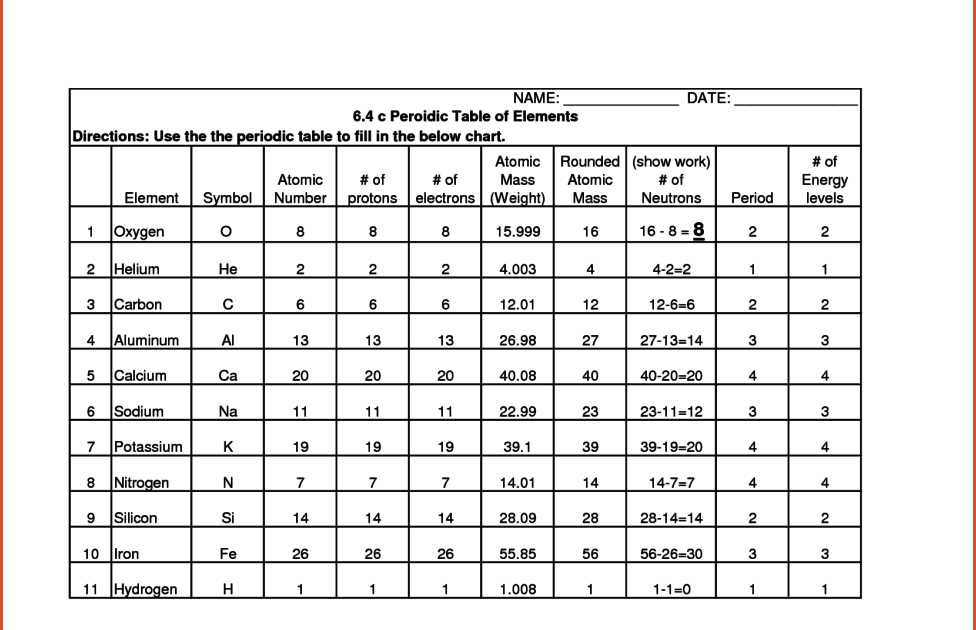
Worksheet Answers for the Development of Atomic Theory
The development of atomic theory is a fascinating journey that has been shaped by various scientists and their contributions. By examining the worksheet answers for the development of atomic theory, we can gain a deeper understanding of how our knowledge of atoms and their properties has evolved over time.
Dalton’s Atomic Theory: According to Dalton’s atomic theory, atoms are indivisible and indestructible particles that combine in simple whole number ratios to form compounds. This theory laid the foundation for our understanding of chemical reactions and the conservation of mass. Worksheet answers for this theory may include concepts such as the law of definite proportions and the law of multiple proportions.
Thomson’s Discovery of the Electron: One of the major breakthroughs in atomic theory came with Thomson’s discovery of the electron. His experiments with cathode rays led to the realization that atoms contain subatomic particles with negative charge. Worksheet answers for this discovery may discuss the plum pudding model of the atom, where electrons are dispersed throughout a positively charged sphere.
Rutherford’s Gold Foil Experiment: Rutherford’s gold foil experiment provided evidence for the existence of a small, dense nucleus within the atom. This experiment led to the development of the nuclear model of the atom, where electrons orbit around a central nucleus. Worksheet answers for this experiment may include descriptions of the unexpected results, such as the deflection of alpha particles and the conclusion that most of the atom’s mass is concentrated in the nucleus.
Bohr’s Planetary Model: Building upon Rutherford’s model, Bohr proposed the idea of electron energy levels or shells. According to this model, electrons orbit the nucleus in specific energy levels, and they can transition between these levels by absorbing or emitting energy. Worksheet answers for this model may discuss the concept of quantized energy, as well as the limitations of the model in explaining more complex atoms.
Quantum Mechanical Model: The quantum mechanical model, based on the principles of quantum mechanics, is the most accurate representation of the atom to date. It describes electrons as wave-particle duality, occupying regions known as orbitals around the nucleus. Worksheet answers for this model may touch upon concepts such as electron probability density, electron configurations, and the periodic table.
In conclusion, examining the worksheet answers for the development of atomic theory allows us to trace the progression of our understanding of atoms and their structure. From Dalton’s atomic theory to the quantum mechanical model, each stage has contributed to our current knowledge and continues to shape scientific advancements in the field of atomic theory.
What is Atomic Theory?
Atomic theory is a scientific explanation of the nature and behavior of matter. It is a fundamental concept in the field of chemistry that has evolved over time through the work of various scientists and experiments.
Dalton’s Atomic Theory: One of the earliest and most influential atomic theories was proposed by John Dalton in the early 19th century. His theory stated that atoms are indivisible particles that combine to form compounds in fixed ratios.
Thomson’s Electron Model: In the late 19th and early 20th centuries, J.J. Thomson discovered the existence of electrons, negatively charged particles that orbit around the atom’s nucleus. His model suggested that atoms are not indivisible, but composed of smaller subatomic particles.
Rutherford’s Nuclear Model: Ernest Rutherford’s famous gold foil experiment in the early 20th century revealed that atoms have a dense, positively charged nucleus at the center, surrounded by a cloud of negatively charged electrons. This model led to the understanding of atomic structure and the concept of atomic number.
Bohr’s Planetary Model: Niels Bohr’s model of the atom in the early 20th century proposed that electrons orbit the nucleus in specific energy levels or shells, similar to planets orbiting the sun. This model explained the stability of atoms and the emission and absorption of energy.
Modern Quantum Mechanical Model: The quantum mechanical model, developed in the early 20th century, describes the behavior of electrons as wave-particle duality and uses complex mathematical equations to determine the probability distribution of electrons in an atom. This model is the basis of our current understanding of atomic structure.
Key Scientists and Their Contributions
The development of atomic theory can be attributed to a number of key scientists who made significant contributions to our understanding of the atom. These scientists conducted experiments, formulated theories, and proposed models that laid the foundation for our current understanding of atomic structure and behavior.
1. John Dalton (1766-1844) – Dalton is often referred to as the father of modern atomic theory. He proposed that atoms are indivisible and indestructible particles, and that different elements are made up of different types of atoms. Dalton also introduced the concept of chemical reactions and the conservation of mass.
2. J.J. Thomson (1856-1940) – Thomson’s experiments with cathode rays led to the discovery of the electron. He proposed the “plum pudding” model, in which electrons were embedded in a positive sphere to form an atom. Thomson’s work laid the foundation for our understanding of subatomic particles and atomic structure.
3. Ernest Rutherford (1871-1937) – Rutherford’s famous gold foil experiment demonstrated the existence of a small, dense, positively charged nucleus at the center of an atom. He proposed the nuclear model of the atom, in which electrons orbit around the nucleus. Rutherford’s work revolutionized our understanding of the atom and led to the development of the nuclear model.
4. Niels Bohr (1885-1962) – Bohr’s work on atomic spectra and the behavior of electrons led to the development of the Bohr model of the atom. This model proposed that electrons exist in specific energy levels or orbitals, and that they can jump between these levels by absorbing or emitting energy. Bohr’s model provided a better understanding of the behavior of electrons and contributed to the development of quantum mechanics.
5. Erwin Schrödinger (1887-1961) – Schrödinger developed the mathematical model of the atom known as quantum mechanics. His equation, known as the Schrödinger equation, describes the behavior of electrons as wave-like entities within the atom. Schrödinger’s contribution to atomic theory provided a more accurate description of electron behavior and helped to shape our current understanding of the atom.
The Early Development of Atomic Theory

The development of atomic theory is a long and intricate process that spans several centuries. It began in ancient Greece, with the philosopher Democritus proposing that matter is made up of tiny indivisible particles called atoms. This idea, however, was largely ignored by other philosophers and scientists of the time.
It wasn’t until the 19th century that significant progress was made in understanding the nature of atoms. John Dalton, an English chemist, formulated the first modern atomic theory. According to Dalton, all elements are composed of tiny indivisible particles called atoms, and chemical reactions occur when atoms are rearranged to form new compounds. His theory gained widespread acceptance and laid the foundation for further research.
Building on Dalton’s work, J.J. Thomson discovered the electron in 1897. Using cathode ray experiments, he determined that atoms contain negatively charged particles called electrons. This discovery led to the development of the “plum pudding” model of the atom, where electrons were thought to be embedded in a positive sphere.
However, Thomson’s model was soon challenged by Ernest Rutherford’s famous gold foil experiment in 1911. Rutherford discovered that atoms have a dense, positively charged nucleus and that the majority of the atom is empty space. He proposed a new atomic model, known as the nuclear model, where electrons orbit the nucleus in distinct energy levels.
The early development of atomic theory was marked by groundbreaking discoveries and evolving models of the atom. Each new discovery paved the way for further research and a deeper understanding of the fundamental building blocks of matter. Today, atomic theory continues to be refined and expanded upon as scientists uncover more about the intricate structure and behavior of atoms.
The Discovery and Understanding of Atomic Structure
The development of our understanding of atomic structure has been a fascinating journey that has undergone numerous scientific breakthroughs over the centuries. Starting from the early theories proposed by ancient Greek philosophers, such as Democritus and Leucippus, who suggested that matter consists of indivisible particles called atoms, to the modern-day atomic models that explain the subatomic particles and their properties.
One significant milestone in the discovery of atomic structure was the experiments conducted by Ernest Rutherford in the early 20th century. Through his famous gold foil experiment, Rutherford demonstrated that atoms have a small, dense, positively charged nucleus, surrounded by a vast empty space where negatively charged electrons orbit. This experiment shattered the previously held “plum pudding” model proposed by J.J. Thomson and paved the way for the development of the nuclear model of the atom.
The nuclear model led to further advancements in our understanding of atomic structure, such as the discovery of subatomic particles. It was later discovered that the nucleus of an atom contains protons, which are positively charged, and neutrons, which have no charge. Electrons, on the other hand, are negatively charged particles that orbit the nucleus in specific energy levels or shells. The arrangement and behavior of these subatomic particles determine the chemical and physical properties of different elements.
In conclusion, the continuous exploration and experimentation in the field of atomic theory have enabled scientists to unravel the complex nature of atomic structure. From the early concepts of indivisible atoms to the discovery of subatomic particles, our understanding has progressed tremendously. This understanding of atomic structure serves as the foundation for numerous scientific disciplines, including chemistry, physics, and materials science.
Modern Atomic Theory and its Applications

In conclusion, the development of atomic theory has been a journey of discovery and refinement over centuries. From the earliest ideas of Democritus to the groundbreaking experiments of Rutherford and Bohr, our understanding of the atom has evolved significantly.
Modern atomic theory, based on quantum mechanics, provides a more accurate description of the behavior and structure of atoms. It explains the wave-particle duality of electrons, the concept of energy levels, and the phenomenon of electron clouds. This theory has not only deepened our understanding of fundamental particles but also revolutionized various fields of science and technology.
The applications of modern atomic theory are vast and diverse. In chemistry, it helps us understand how atoms bond to form molecules and predicts their properties. In physics, it forms the basis for studying the behavior of particles in accelerators and developing new materials with specific properties. In medicine, it enables the development of imaging techniques like MRI that rely on the behavior of atomic nuclei. In energy production, it guides the design and optimization of nuclear reactors and helps us harness the power of the atom.
Furthermore, modern atomic theory has led to the development of numerous technological advancements. It has paved the way for the miniaturization of electronic devices, allowing us to have powerful computers and smartphones. It has also facilitated advancements in materials science, leading to the creation of stronger and lighter materials. Additionally, it has revolutionized our ability to study and manipulate matter at the atomic and molecular levels.
In summary, modern atomic theory has not only enhanced our understanding of the fundamental building blocks of matter but has also had a profound impact on various scientific disciplines and technological advancements. It continues to drive innovation and exploration in fields ranging from chemistry and physics to medicine and engineering. As we delve deeper into the atomic realm, there is no doubt that our understanding will continue to evolve, uncovering new mysteries and possibilities for future generations.