
Dalton’s law of partial pressures is a fundamental concept in the field of gas laws. It states that the total pressure exerted by a mixture of non-reacting gases is equal to the sum of the partial pressures of each individual gas in the mixture. This law is named after the English chemist and physicist, John Dalton, who first proposed it in the early 19th century.
The key principle behind Dalton’s law is that gases behave independently of each other, regardless of their interaction or presence with other gases. Each gas in a mixture exerts a pressure that is proportional to its concentration or partial mole fraction. This means that the total pressure of a gas mixture can be calculated by adding together the partial pressures of each gas component.
The practical applications of Dalton’s law are significant and wide-ranging. It is used in various fields, such as chemistry, physics, and engineering, to understand and predict the behavior of gases in different conditions. For example, it is commonly applied in the measurement of gas concentrations in the atmosphere, as well as in industrial processes where gas mixtures need to be controlled or analyzed.
Dalton’s Law of Partial Pressures Answer Key
Dalton’s Law of Partial Pressures states that in a mixture of gases, the total pressure exerted is equal to the sum of the individual pressures exerted by each gas. This law is based on the idea that gases behave independently of one another and do not interact with each other.
The key concept behind Dalton’s Law is that the total pressure is determined by the number of gas particles present and their individual velocities. Each gas contributes to the total pressure in proportion to its partial pressure, which is the pressure it would exert if it were the only gas present in the container.
To calculate the total pressure in a mixture of gases, the following steps can be followed:
- Determine the partial pressure of each gas in the mixture. This can be done by multiplying the mole fraction of each gas by the total pressure of the mixture. The mole fraction of a gas is the ratio of the number of moles of that gas to the total number of moles in the mixture.
- Add up the partial pressures of all the gases to get the total pressure of the mixture.
For example, let’s consider a mixture of nitrogen gas (N2) and oxygen gas (O2) in a container with a total pressure of 2 atm. If the mole fraction of nitrogen gas is 0.8 and the mole fraction of oxygen gas is 0.2, we can calculate the partial pressures as follows:
- Partial pressure of nitrogen gas = 0.8 x 2 atm = 1.6 atm
- Partial pressure of oxygen gas = 0.2 x 2 atm = 0.4 atm
Therefore, the total pressure of the mixture is 1.6 atm + 0.4 atm = 2 atm, which matches the given total pressure of the container.
Understanding Dalton’s Law of Partial Pressures is crucial in many areas of chemistry, such as analyzing gas mixtures and understanding the behavior of gases in various conditions. It allows scientists to accurately predict and control the behavior of gases in a wide range of applications.
What is Dalton’s Law of Partial Pressures?
Dalton’s Law of Partial Pressures, also known as Dalton’s Law of Partial Pressures, states that the total pressure exerted by a mixture of non-reacting gases is equal to the sum of the partial pressures that each gas would exert if it were present alone in the same volume and temperature. In other words, the total pressure of a gas mixture is the sum of the pressures of each individual gas in the mixture.
This law is based on the idea that each gas in a mixture behaves independently of the others and exerts its own pressure. The partial pressure of a gas is a measure of the pressure that gas would exert if it occupied the entire volume alone. It is directly proportional to the mole fraction of the gas in the mixture, which is the ratio of the number of moles of that gas to the total number of moles of gas in the mixture.
Dalton’s Law of Partial Pressures is useful in many areas of science and industry, particularly in the study of gas mixtures and their behavior. It is used, for example, in the analysis of air quality, the determination of the composition of gases in a sample, and the calculation of gas solubilities. By understanding the principles behind Dalton’s Law of Partial Pressures, scientists and engineers are able to accurately predict and control the behavior of gas mixtures in various applications.
Understanding Dalton’s Law of Partial Pressures
Dalton’s Law of Partial Pressures states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures of each individual gas. This law is based on the concept that gases behave independently of each other and that their individual pressures can be combined to determine the total pressure of the mixture.
In simple terms, Dalton’s Law of Partial Pressures can be understood as follows: when multiple gases are present in a container, each gas exerts its own pressure independently of the others. The total pressure exerted by the mixture is the sum of these individual pressures. This concept can be visualized by imagining a container divided into compartments, with each compartment containing a different gas. The pressure exerted by each gas in its respective compartment is its partial pressure, and the sum of these partial pressures gives the total pressure of the mixture.
For example, if a container contains equal amounts of oxygen and nitrogen gas, the total pressure exerted by the mixture will be the sum of the partial pressures of oxygen and nitrogen. If the partial pressure of oxygen is 200 mmHg and the partial pressure of nitrogen is 300 mmHg, then the total pressure of the mixture will be 500 mmHg.
Dalton’s Law of Partial Pressures is particularly useful in understanding the behavior of gases in various applications, such as in industrial processes, atmospheric science, and medical gas administration. By knowing the partial pressures of each gas in a mixture, it becomes possible to calculate the concentrations or amounts of each gas, as well as predict changes in pressure when conditions are altered.
In conclusion, Dalton’s Law of Partial Pressures provides a fundamental understanding of how gases behave in mixtures and allows for the determination of total pressure based on the partial pressures of individual gases. This concept is essential in various fields and applications where gases play a significant role.
Key Principles of Dalton’s Law of Partial Pressures
Dalton’s law of partial pressures states that the total pressure exerted by a mixture of gases is equal to the sum of the pressures exerted by each individual gas in the mixture. This law is based on the concept that gases behave independently of each other, and the total pressure is the result of the combined effect of each gas’s individual pressure.
One key principle of Dalton’s law is that the partial pressure of a gas is proportional to its mole fraction in the mixture. The mole fraction represents the ratio of the number of moles of a specific gas to the total number of moles in the mixture. The partial pressure of a gas can be calculated by multiplying the mole fraction of the gas by the total pressure of the mixture.
Another important principle of Dalton’s law is that gases do not affect each other’s behavior or properties when they mix. This means that each gas in a mixture behaves as if it is the only gas present, and its pressure is determined solely by its own properties. The total pressure of the mixture is simply the sum of the individual pressures of each gas.
Dalton’s law of partial pressures is an essential concept in understanding the behavior of mixtures of gases. It allows scientists to predict and calculate the pressure of each gas in a mixture, and is commonly used in various fields such as chemistry, physics, and environmental science. By understanding and applying these key principles, scientists can accurately analyze and study the properties and behavior of gas mixtures.
Application of Dalton’s Law of Partial Pressures

Dalton’s Law of Partial Pressures states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures of each individual gas. This law has important applications in various scientific and industrial processes.
One application of Dalton’s Law is in the field of gas chemistry and analysis. It is used to calculate the partial pressures of gases in a mixture, which is essential for determining the composition of the mixture. For example, in the analysis of air, Dalton’s Law can be used to determine the partial pressures of oxygen, nitrogen, and other gases present in the atmosphere.
Dalton’s Law is also applied in the field of gas collection and storage. It is used to calculate the partial pressure of a specific gas in a mixture, which is important for storing and transporting gases. For instance, in the storage of compressed gases, the partial pressure of each gas helps to determine the appropriate container and storage conditions to ensure safety.
Another practical application of Dalton’s Law is in the field of respiratory physiology. It explains the exchange of gases in the lungs and the blood, as well as the effects of altitude on gas exchange. Dal
Examples of Dalton’s Law of Partial Pressures in Real Life

Dalton’s Law of Partial Pressures states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures of each individual gas. This concept has many practical applications in various fields. Here are some examples of how Dalton’s Law can be observed in real life:
1. Scuba Diving
Scuba divers rely on Dalton’s Law when diving at different depths. As they descend deeper into the water, the pressure increases, and this affects the partial pressures of the gases they breathe. The air tanks they carry contain a mixture of gases, typically oxygen and nitrogen. According to Dalton’s Law, as the divers go deeper, the partial pressure of each gas increases, which can lead to nitrogen narcosis or oxygen toxicity if not managed properly.
2. Gasoline in Vehicles
In internal combustion engines, the fuel-air mixture is ignited to generate power. The air that is drawn into the engine contains a mixture of gases, including oxygen, nitrogen, carbon dioxide, and others. Dalton’s Law allows us to calculate the partial pressure of oxygen, which is essential for the combustion process. By adjusting the air-fuel ratio, the partial pressure of oxygen can be optimized for efficient combustion and power output in vehicles.
3. Atmospheric Pressure
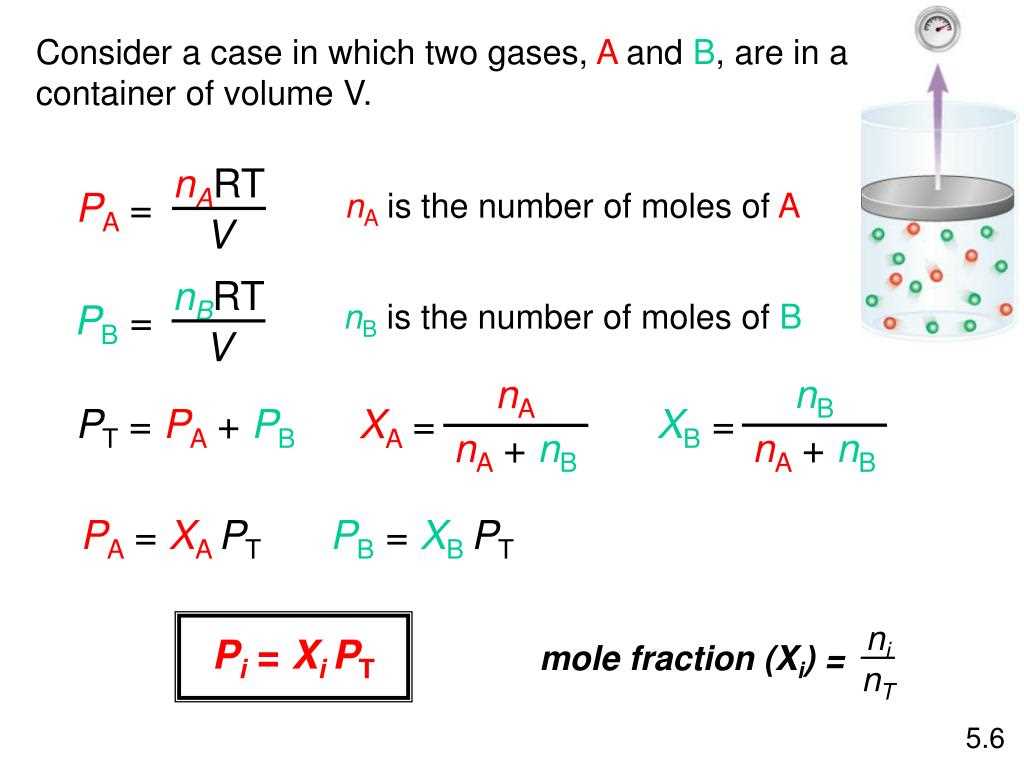
Dalton’s Law explains why atmospheric pressure decreases with altitude. The atmosphere is composed of different gases such as nitrogen, oxygen, and trace amounts of other gases. As we ascend to higher altitudes, the air becomes less dense, and the partial pressures of each gas decrease. This decrease in pressure is important for aviation and the functioning of high-altitude equipment, as it affects the performance of engines and the behavior of gases.
In summary, Dalton’s Law of Partial Pressures is a fundamental principle in understanding the behavior of gas mixtures. Its applications can be seen in various fields, including scuba diving, combustion engines, and the study of atmospheric pressure. By considering the partial pressures of each gas, we can better understand the properties and interactions of gas mixtures in real-life scenarios.
Tips for Solving Dalton’s Law of Partial Pressures Problems
When working with Dalton’s Law of Partial Pressures, it’s important to keep in mind some key tips that can help you solve problems more effectively. Here are some tips to consider:
- Identify the gases involved: Start by identifying all the gases present in the system. Make a list of the different gases and their respective partial pressures.
- Convert units if necessary: Make sure all the partial pressures are in the same units. If necessary, convert the units to make the calculations easier.
- Calculate the total pressure: Add up all the partial pressures to get the total pressure of the system. This should be equal to the atmospheric pressure if the system is open to the atmosphere.
- Use the mole fraction: Calculate the mole fraction of each gas by dividing its moles by the total moles of all the gases. This will allow you to determine the proportion of each gas in the mixture.
- Apply Dalton’s Law of Partial Pressures: Use the mole fractions to calculate the partial pressure of each gas. Multiply the mole fraction of each gas by the total pressure to obtain the partial pressure of that gas.
By following these tips, you should be able to solve Dalton’s Law of Partial Pressures problems with greater ease. Remember to pay attention to units, accurately identify the gases involved, and use the appropriate formulas to find the partial pressures. Practice and repetition will also help improve your problem-solving skills in this area. Happy problem-solving!