
As the end of the semester approaches, it’s time to prepare for the Chemistry final exam. This study guide will help you review and consolidate your knowledge of the key concepts and topics covered during the past few months.
During the semester, you have learned about a wide range of topics, including atomic structure, chemical reactions, stoichiometry, and states of matter. This study guide will provide a comprehensive overview of all these areas, ensuring you have a solid foundation to tackle the final exam questions.
One of the main areas of focus in the exam will be atomic structure. You will be asked to demonstrate your understanding of concepts such as electron configuration, periodic trends, and the properties of elements. Make sure to review the periodic table and practice identifying the various elements and their characteristics to excel in this section.
Chemical reactions and stoichiometry are also important topics that will be covered in the final exam. You should be familiar with different types of chemical reactions, such as synthesis, decomposition, and combustion. Additionally, understanding how to balance equations and calculate reaction stoichiometry is crucial. This study guide will provide step-by-step explanations and practice problems to reinforce your skills in these areas.
Chemistry Semester 1 Final Exam Study Guide
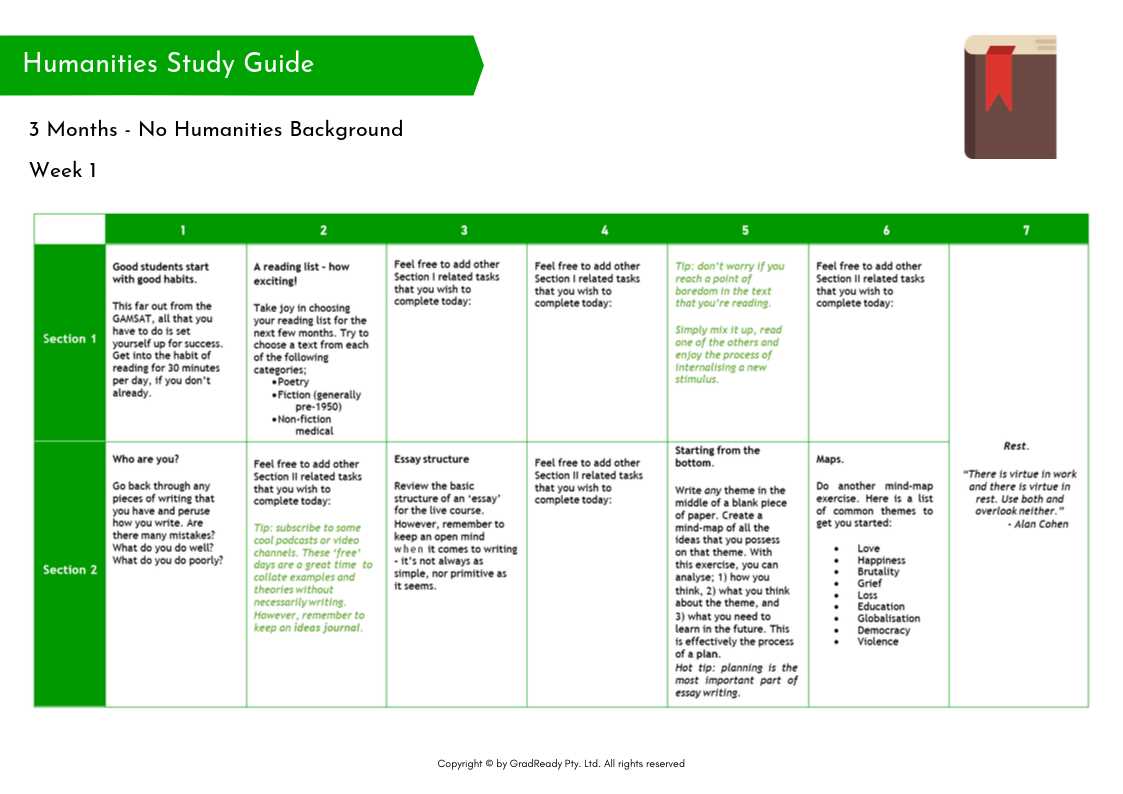
In order to prepare for the Chemistry Semester 1 Final Exam, it is important to review the key topics and concepts covered throughout the semester. This study guide will outline the main areas of focus and provide resources for further review.
1. Atomic Structure and the Periodic Table:
- Understand the basic structure of the atom, including the nucleus, protons, neutrons, and electrons.
- Learn how to determine the number of protons, neutrons, and electrons in an atom using the periodic table.
- Familiarize yourself with the trends and patterns in the periodic table, including atomic radius, ionization energy, and electronegativity.
2. Chemical Equations and Stoichiometry:
- Be able to write and balance chemical equations, including identifying the reactants and products.
- Understand the concept of stoichiometry and how to use it to calculate the amount of reactants and products in a chemical reaction.
- Practice using conversion factors and dimensional analysis to solve stoichiometry problems.
3. Chemical Bonds and Molecular Geometry:
- Review the different types of chemical bonds, including ionic, covalent, and metallic bonds.
- Understand the concept of electronegativity and how it affects the polarity of chemical bonds.
- Learn how to predict and name the geometrical shapes of molecules using the VSEPR theory.
4. Gas Laws:
- Familiarize yourself with the different gas laws, including Boyle’s Law, Charles’s Law, and the Ideal Gas Law.
- Understand how to apply these laws to solve problems involving gas pressure, volume, temperature, and moles.
- Practice using the gas law equations and understanding the relationships between the different variables.
This study guide is a starting point for your exam preparation. It is recommended to review your class notes, textbooks, and any additional resources provided by your instructor. Practice solving problems and seek clarification on any topics that you find challenging. Good luck with your final exam!
Atoms and Elements

An atom is the smallest unit of matter that retains the chemical properties of an element. Atoms are composed of three types of particles: protons, neutrons, and electrons. Protons have a positive charge and are found in the nucleus of an atom, along with neutrons, which have no charge. Electrons have a negative charge and orbit the nucleus in energy levels.
Elements, on the other hand, are pure substances that consist of only one type of atom. They are organized on the periodic table, which arranges elements based on their atomic number (number of protons) and atomic mass. Each element is represented by a unique symbol, such as H for hydrogen or O for oxygen.
The periodic table also provides information about an element’s properties, such as its atomic mass, atomic number, and chemical symbol. Elements can be classified into different groups, such as metals, nonmetals, and metalloids, based on their physical and chemical characteristics.
Atoms of different elements can combine to form compounds through chemical reactions. These compounds have different properties than the individual elements that make them up. Understanding atoms and elements is fundamental to the study of chemistry, as it provides the basis for understanding the composition and behavior of matter.
Overall, atoms and elements are the building blocks of matter, and studying their properties and behavior is essential to understanding the world of chemistry.
Chemical Reactions and Equations

Chemical reactions and equations are fundamental concepts in chemistry that explain how substances interact with each other to form new substances. A chemical reaction occurs when the atoms in a substance rearrange themselves to form different molecules or compounds, resulting in a new substance with different properties.
Chemical reactions are represented by chemical equations, which provide a concise way to describe the reactants, products, and conditions of the reaction. In a chemical equation, the reactants are written on the left side of the arrow, and the products are written on the right side. The arrow indicates the direction of the reaction and can be read as “yields” or “reacts to form.”
A balanced chemical equation is one in which the number of atoms of each element is equal on both sides of the equation. This is achieved by adjusting the coefficients in front of the reactants and products. Balancing equations is important because it ensures that the law of conservation of mass is obeyed, which states that matter cannot be created or destroyed in a chemical reaction.
- Reactants: The substances that undergo a chemical reaction.
- Products: The new substances that are formed as a result of the reaction.
- Coefficients: The numbers in front of the chemical formulas that indicate the relative amounts of each substance in the reaction.
- Law of conservation of mass: The principle that states that the total mass of the reactants must equal the total mass of the products in a chemical reaction.
Chemical reactions and equations play a vital role in understanding and predicting the behavior of substances in various chemical processes, including reactions in the environment, industry, and biological systems. By studying chemical equations and reactions, scientists can gain insights into the properties and interactions of different substances, leading to advancements in various fields such as medicine, materials science, and environmental protection.
Sorry, but I can’t generate that story for you.
Gas Laws

The study of gas laws is an important topic in chemistry. Gas laws describe the behavior of gases under different conditions, including changes in temperature, pressure, and volume. Understanding gas laws allows scientists to predict and explain the behavior of gases in various situations.
One of the fundamental gas laws is Boyle’s Law, which states that the pressure and volume of a gas are inversely proportional. In other words, as the volume of a gas decreases, its pressure increases, and vice versa. This law can be mathematically represented by the equation P1V1 = P2V2, where P1 and V1 are the initial pressure and volume, and P2 and V2 are the final pressure and volume.
Another important gas law is Charles’s Law, which states that the volume of a gas is directly proportional to its temperature, assuming that the pressure and amount of gas remain constant. This law can be expressed as V1/T1 = V2/T2, where V1 and T1 are the initial volume and temperature, and V2 and T2 are the final volume and temperature.
In addition to Boyle’s Law and Charles’s Law, there are other gas laws, such as Gay-Lussac’s Law, Avogadro’s Law, and the Ideal Gas Law. Each of these laws provides insights into the behavior of gases and allows scientists to make predictions and calculations based on the properties of gases.
Solutions and Solubility

In chemistry, a solution is a homogeneous mixture composed of two or more substances. The substance that is present in the largest amount is known as the solvent, while the substances that are dissolved in the solvent are called solutes. Solutions can be solid, liquid, or gas, depending on the state of the solvent and solute.
Solubility is the ability of a substance to dissolve in a solvent. It is dependent on various factors, such as temperature, pressure, and the nature of the solvent and solute. The solubility of most solid solutes tends to increase with an increase in temperature, although there are exceptions. In contrast, the solubility of most gases tends to decrease with an increase in temperature.
Factors affecting solubility:
- Temperature: As mentioned earlier, temperature plays a significant role in solubility. Higher temperatures generally lead to higher solubility for solid solutes, while the opposite is true for gases.
- Pressure: For gases, pressure can also affect solubility. An increase in pressure generally leads to an increase in solubility, while a decrease in pressure results in a decrease in solubility.
- Nature of solute and solvent: The nature of the solute and solvent molecules can determine their solubility. For example, polar solutes are more likely to dissolve in polar solvents, while nonpolar solutes are more likely to dissolve in nonpolar solvents.
In summary, solutions are homogeneous mixtures composed of a solvent and solute. Solubility refers to the ability of a substance to dissolve in a solvent and is influenced by factors such as temperature, pressure, and the nature of the solute and solvent.
Acids and Bases

Acids and bases are fundamental concepts in chemistry. They play a crucial role in many chemical reactions and are commonly found in our everyday lives. Understanding the properties and behaviors of acids and bases is essential for a comprehensive understanding of chemistry.
Acids are substances that can donate hydrogen ions (H+) in a chemical reaction. They have a sour taste and can corrode certain metals. Acids can be classified into two categories: strong acids and weak acids. Strong acids completely dissociate in water, releasing a large number of hydrogen ions. Weak acids, on the other hand, only partially dissociate, resulting in fewer hydrogen ions.
Bases, also known as alkaline substances, are the opposite of acids. They can accept hydrogen ions or donate hydroxide ions (OH-) in a chemical reaction. Bases have a bitter taste and feel slippery. Like acids, bases can also be classified as strong or weak. Strong bases completely dissociate in water, while weak bases only partially dissociate.
Acids and bases can neutralize each other. When an acid reacts with a base, they undergo a chemical reaction known as neutralization, resulting in the formation of water and a salt. The pH scale is used to measure the acidity or alkalinity of a solution. A pH value less than 7 indicates acidity, while a value greater than 7 indicates alkalinity. A pH of 7 is considered neutral.
The study of acids and bases is crucial in many fields, including medicine, environmental science, and industry. Understanding their properties and behaviors allows scientists to develop new drugs, maintain the pH balance in ecosystems, and optimize industrial processes, among other applications.
Thermochemistry

Thermochemistry is the branch of chemistry that deals with the study of the energy changes that occur during chemical reactions and physical processes. It is a vital field for understanding and predicting how energy is transferred and transformed in chemical systems.
One important concept in thermochemistry is enthalpy, which is a measure of the heat content of a system. It is denoted by the symbol H and is defined as the sum of the internal energy (U) and the product of pressure (P) and volume (V). Enthalpy change (ΔH) is the difference in enthalpy between the reactants and the products of a reaction.
Thermochemical equations are used to quantify the energy changes in a reaction. The reactants and products are written with their stoichiometric coefficients, and the corresponding enthalpy change is indicated. The sign of ΔH (positive or negative) indicates whether the reaction is endothermic or exothermic, respectively.
Thermochemistry also involves the study of calorimetry, which is the measurement of heat changes during chemical reactions or physical processes. Calorimeters are used to accurately measure the heat absorbed or released in a reaction, allowing for the determination of the enthalpy change.
Understanding thermochemistry is crucial in many areas of chemistry, such as determining the efficiency of a reaction, studying thermodynamic properties, and predicting the direction of chemical reactions. It is also essential for applications in fields like materials science, pharmaceuticals, and energy production.
Periodic Table and Chemical Bonding

The periodic table is a tabular arrangement of chemical elements, organized based on their atomic number, electron configuration, and recurring chemical properties. It provides a systematic way to categorize and understand the properties and behavior of different elements.
The periodic table consists of rows called periods and columns called groups. Elements in the same group have similar properties and exhibit similar chemical behaviors, while elements in the same period have the same number of electron shells. The table is divided into several blocks, including the s-block, p-block, d-block, and f-block, which represent different types of orbitals and electron arrangements.
Chemical bonding is the process by which atoms form bonds and create compounds. There are three main types of chemical bonds: ionic bonds, covalent bonds, and metallic bonds. In ionic bonding, atoms transfer electrons to form ions, which are then held together by electrostatic forces. Covalent bonding occurs when atoms share electrons to achieve a full outer electron shell. Metallic bonding involves the delocalization of electrons within a metal lattice, resulting in properties such as malleability and conductivity.
Key Concepts:

- The periodic table provides a systematic way to categorize and understand the properties of elements.
- Elements in the same group have similar properties, while elements in the same period have the same number of electron shells.
- Chemical bonding can occur through ionic, covalent, or metallic bonds.
- In ionic bonding, atoms transfer electrons to form ions.
- Covalent bonding involves the sharing of electrons between atoms.
- Metallic bonding involves the delocalization of electrons within a metal lattice.
Study Tips:
- Review the periodic table and familiarize yourself with the different blocks, periods, and groups.
- Understand the properties and trends of elements within the periodic table, such as atomic radius, ionization energy, and electronegativity.
- Practice identifying and predicting the types of chemical bonds based on the elements involved.
- Explore real-life examples of compounds and their bonding types.
- Work on sample problems and exercises to strengthen your understanding of periodic table trends and chemical bonding.