Chemistry is a fascinating branch of science that explores the properties and interactions of matter. It is a key subject for understanding the world around us, from the composition of the air we breathe to the materials that make up our bodies. In the study of chemistry, one often encounters challenging problems and questions that require careful analysis and problem-solving skills. This is where the Chemistry a study of matter 10.6 answer key comes in. It provides a comprehensive guide to understanding and solving the questions posed in Chapter 10.6 of the Chemistry a study of matter textbook.
Chapter 10.6 of the textbook focuses on a specific topic or concept in chemistry, providing students with a deeper understanding of the subject matter. However, it is not uncommon for students to encounter difficulties while working through the exercises and problems in the chapter. This is where the answer key becomes an invaluable resource. It serves as a tool to check one’s work, verify solutions, and gain a better grasp of the material. The Chemistry a study of matter 10.6 answer key offers step-by-step solutions and explanations to the questions posed in the chapter, guiding students towards a better understanding of the topic at hand.
The Chemistry a study of matter 10.6 answer key is designed to be a companion to the textbook, enabling students to reinforce the concepts covered in the chapter. It allows for independent learning and self-assessment, as students can compare their solutions to the provided answers and identify any areas where further study may be needed. By utilizing the answer key, students can gain confidence in their understanding of the material and develop essential analytical and problem-solving skills that will serve them well in future chemistry courses and applications.
In conclusion, the Chemistry a study of matter 10.6 answer key is an essential resource for students studying chemistry. It provides a comprehensive guide to understanding and solving the questions posed in Chapter 10.6 of the textbook. By using the answer key, students can enhance their learning experience, reinforce their understanding, and develop crucial analytical and problem-solving skills. Whether used as a tool for checking solutions or as a study aid, the answer key serves as a valuable companion to the Chemistry a study of matter textbook.
What is Chemistry?
Chemistry is the scientific study of matter and the transformations that occur within it. It is a branch of science that explores the composition, properties, structure, and behavior of substances, as well as the changes they undergo during chemical reactions. The study of chemistry allows us to understand the world around us at a molecular level and provides insights into the fundamental processes that govern nature.
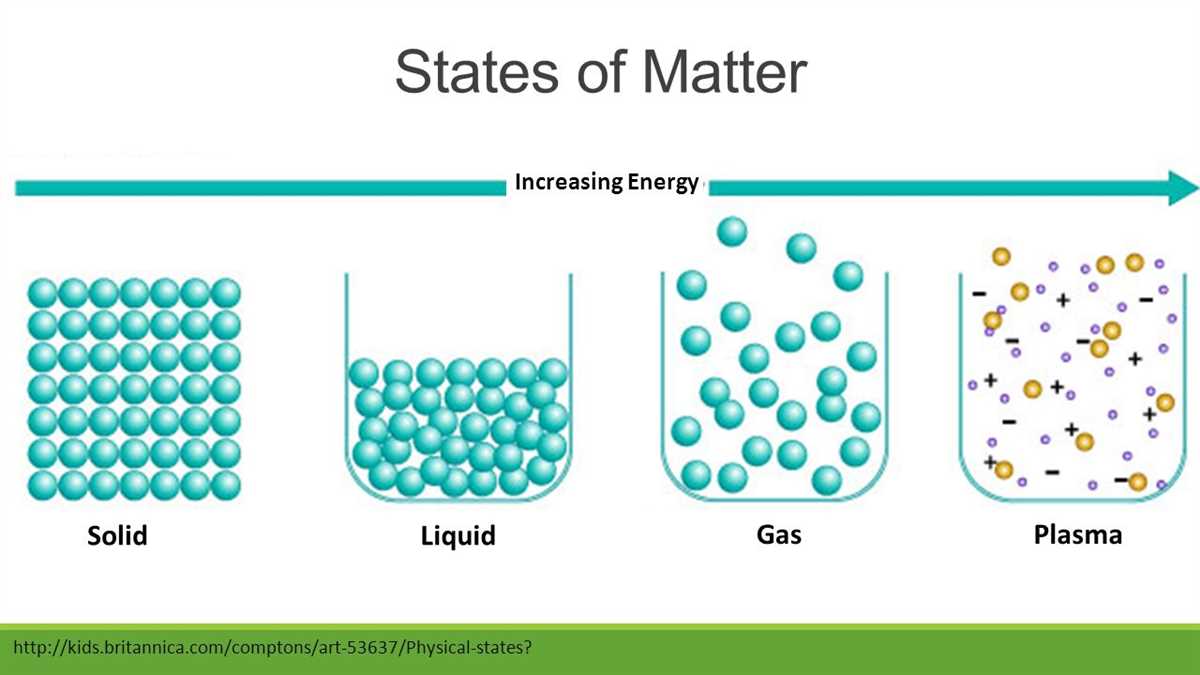
In chemistry, matter refers to anything that occupies space and has mass. It includes all substances that make up the physical world, such as solids, liquids, gases, and even plasma. Chemists investigate the properties of these substances, such as their physical appearance, density, melting point, boiling point, and solubility. They also examine the arrangement of atoms and molecules within a substance, known as its molecular structure, and study how this structure affects its properties and behavior.
Chemical reactions are at the core of chemistry. They involve the breaking and formation of chemical bonds, leading to the rearrangement of atoms and the conversion of substances into new ones. Chemists study the factors that influence the rate and extent of these reactions, as well as the energy changes that occur during the process. Understanding chemical reactions is crucial for developing new materials, discovering new drugs, and solving environmental problems.
Overall, chemistry plays a vital role in many scientific disciplines and industries, including medicine, pharmacology, materials science, environmental science, agriculture, and energy production. It provides the foundation for understanding how the universe works at its most fundamental level and allows us to develop new technologies and solutions to complex problems.
Overview of Matter
Matter is anything that has mass and takes up space. It is the physical substance of the universe and exists in various forms such as solid, liquid, and gas. Matter can be composed of individual atoms, molecules, or ions, which are the building blocks of matter.
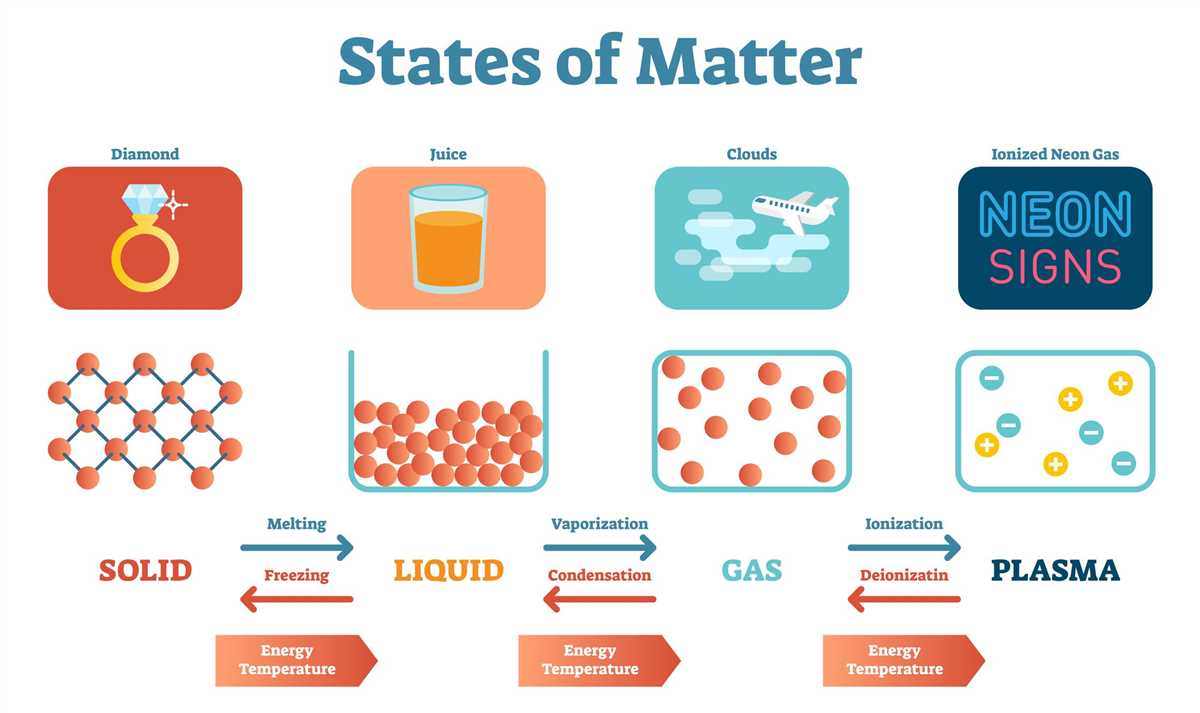
Matter can undergo physical and chemical changes. Physical changes do not result in the formation of new substances and can include changes in state, shape, or size. For example, water boiling and ice melting are physical changes as the substance remains the same. On the other hand, chemical changes involve the formation of new substances with different chemical properties. Examples of chemical changes include rusting of iron and burning of paper.
Classification of Matter:
Matter is classified into two main categories: pure substances and mixtures. Pure substances have a uniform composition and can be further classified as elements or compounds. Elements are substances that cannot be broken down into simpler substances by chemical means. They are composed of only one type of atom and have unique physical and chemical properties. Examples of elements include hydrogen, oxygen, and carbon. Compounds, on the other hand, are substances that are composed of two or more elements chemically combined in a fixed ratio. They have different physical and chemical properties compared to the elements they are composed of. Examples of compounds include water (H2O) and carbon dioxide (CO2).
Mixtures, on the other hand, are combinations of two or more substances that are physically blended together but not chemically combined. Mixtures can be further classified as homogeneous or heterogeneous. Homogeneous mixtures have a uniform composition throughout, and the individual substances are not easily distinguishable. Examples of homogeneous mixtures include saltwater and air. Heterogeneous mixtures, on the other hand, have a non-uniform composition and the individual substances are visibly distinguishable. Examples of heterogeneous mixtures include salad, soil, and granite.
Understanding the Study of Matter
The study of matter is a fundamental aspect of chemistry. Matter is everything that has mass and occupies space, and understanding its properties and behavior is crucial in various scientific and technological applications.
Matter can exist in different forms, such as solid, liquid, or gas, and can undergo physical and chemical changes. Physical properties of matter include characteristics like color, density, and melting point, while chemical properties refer to its ability to undergo chemical reactions and form new substances.
Physical changes involve alterations in the form or state of matter without changing its chemical composition. For example, melting ice into water or boiling water into vapor are physical changes. These changes can be reversible, meaning the original substance can be obtained again, or irreversible, where the new substance cannot be easily recovered. Understanding physical changes is essential in fields like materials science, where manipulating the properties of materials is crucial.
Chemical changes, on the other hand, involve the transformation of matter into new substances with different chemical compositions. These changes are often accompanied by the release or absorption of energy. For example, combustion, where a substance reacts with oxygen to produce heat and light, is a chemical change. Understanding chemical changes is vital in areas such as pharmaceuticals, where the synthesis of new compounds is necessary.
Chemists study matter by conducting experiments and using various techniques and instruments to analyze its properties. Through these investigations, they can identify the composition, structure, and behavior of different substances, enabling the development of new materials, medicines, and technologies.
In conclusion, the study of matter is essential in chemistry as it provides insight into the properties and behavior of substances. Understanding physical and chemical changes allows scientists to manipulate matter for practical applications, contributing to advancements in various fields.
Significance of Answer Keys

Answer keys play a crucial role in the learning process, especially in subjects like chemistry. They provide students with a reference to check their answers, ensuring that they are on the right track and understanding the concepts correctly. Answer keys serve as a tool for self-assessment, allowing students to identify their strengths and weaknesses in the subject matter.
Having access to an answer key can enhance the learning experience by promoting active learning. Students can compare their own answers to the provided key and analyze the differences. This process encourages critical thinking and problem-solving skills. It also enables students to identify any misconceptions they may have and seek further clarification from their teachers or peers.
The significance of answer keys can be summarized in the following points:
- Accuracy: Answer keys provide accurate solutions, eliminating any doubts or confusion about the correct answers.
- Self-assessment: Students can assess their understanding of the material and identify areas that need improvement.
- Feedback: Answer keys act as a source of immediate feedback, allowing students to gauge their progress and make necessary adjustments in their learning strategies.
- Learning from mistakes: Through the comparison of their answers with the answer key, students can learn from their mistakes and avoid repeating them in future assessments.
- Independence: Answer keys promote independent learning by providing students with the resources they need to check their own work and find solutions to problems.
Overall, answer keys are valuable tools in the learning process as they promote self-assessment, critical thinking, and independent learning. They empower students to take control of their own learning journey and encourage them to actively engage with the subject matter.
Key Concepts in Chemistry: A Study of Matter 10.6

The key concepts covered in Chemistry: A Study of Matter 10.6 include the following:
- Colligative Properties: This concept involves the study of how the physical properties of a solution are affected by the number of solute particles present. The colligative properties discussed in this chapter include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.
- Vapor Pressure Lowering: When a nonvolatile solute is added to a solvent, the vapor pressure of the solvent decreases. This is due to the fact that the solute particles occupy space at the surface of the solvent, reducing the number of solvent particles that can escape into the gas phase.
- Boiling Point Elevation: When a solute is dissolved in a solvent, the boiling point of the solvent increases. This is because the presence of the solute particles lowers the vapor pressure of the solvent, requiring a higher temperature to reach the boiling point.
- Freezing Point Depression: When a solute is dissolved in a solvent, the freezing point of the solvent decreases. This is because the presence of the solute particles disrupts the regular arrangement of the solvent molecules, making it more difficult for them to form a solid lattice.
- Osmotic Pressure: When a semipermeable membrane separates a solution from its pure solvent, a pressure difference can be observed across the membrane. This pressure difference is known as osmotic pressure and is due to the movement of solvent particles from the area of higher solvent concentration to the area of lower solvent concentration.
Understanding these key concepts is essential for studying the behavior of solutions and their physical properties. Knowledge of colligative properties allows chemists to predict and explain phenomena such as boiling point elevation and freezing point depression, which are important in various practical applications such as cooking, cryopreservation, and industrial processes.
Chemical Reactions and Balancing Equations

Chemical reactions are fundamental processes in chemistry that involve the transformation of one or more substances into new substances. These reactions occur when atoms rearrange their bonding patterns to form different molecules or compounds. Understanding chemical reactions is crucial for understanding how different substances interact and how new substances are formed.
In order to fully describe a chemical reaction, it is necessary to write a balanced chemical equation. A balanced chemical equation shows the reactants on the left side of the equation and the products on the right side, with an arrow indicating the direction of the reaction. The coefficients in front of the reactants and products indicate the stoichiometry of the reaction, or the ratio of the number of moles of each substance involved.
When balancing a chemical equation, it is important to ensure that the law of conservation of mass is obeyed. This law states that matter cannot be created or destroyed in a chemical reaction, only rearranged. Therefore, the number of atoms of each element must be the same on both sides of the equation. Balancing chemical equations often involves adjusting the coefficients to achieve this balance.
In addition to balancing the number of atoms, it is also important to balance the charge in a chemical equation. This is especially relevant for reactions involving ions, where the overall charge must be conserved. This can be done by adding coefficients or adjusting the charges of the individual species involved.
Example:
A common chemical reaction is the combustion of methane (CH4) in oxygen (O2) to produce carbon dioxide (CO2) and water (H2O). The balanced chemical equation for this reaction is:
CH4 + 2O2 → CO2 + 2H2O
In this equation, there is one carbon atom, four hydrogen atoms, and four oxygen atoms on both sides, satisfying the law of conservation of mass. The coefficients in front of the reactants and products indicate that one molecule of methane reacts with two molecules of oxygen to produce one molecule of carbon dioxide and two molecules of water.