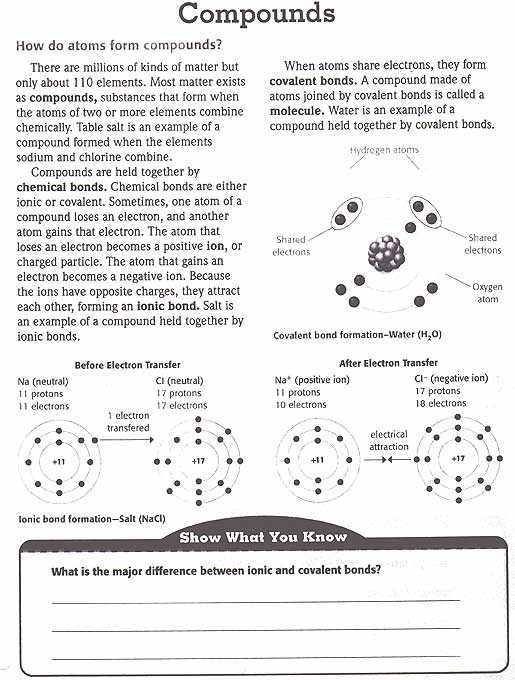
Chapter 4 of the textbook covers the topic of electrons in atoms. This is an important concept in understanding the behavior of atoms and their properties. Electrons play a crucial role in determining the chemical and physical properties of elements and compounds. In this chapter, students will learn about the arrangement of electrons in atoms, as well as how to determine the electron configurations of different elements.
One of the main objectives of this chapter is to introduce students to the concept of energy levels and sublevels in atoms. The electron configuration of an atom is determined by the distribution of electrons among these energy levels and sublevels. Understanding this distribution is essential in predicting the chemical reactivity and bonding behavior of elements.
Throughout the chapter, students will also learn about the periodic table and its relation to electron configurations. The periodic table provides a systematic way of organizing elements based on their electron configurations, as well as their chemical and physical properties. By the end of the chapter, students should be able to identify patterns in electron configurations and predict the properties of elements based on their positions in the periodic table.
What are electrons and how are they related to atoms?
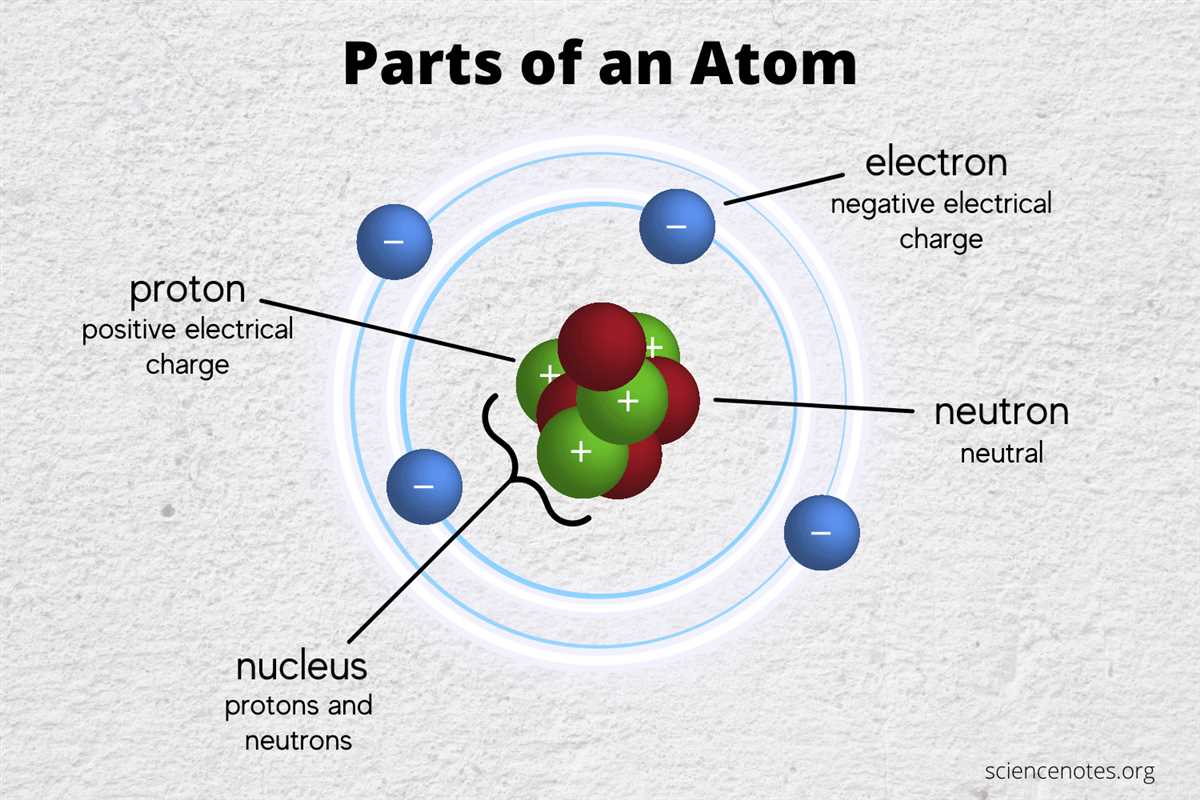
Electrons are subatomic particles that are found orbiting around the nucleus of an atom. They have a negative charge and are extremely small in size compared to the nucleus. Electrons play a crucial role in determining the chemical and physical properties of an atom.
The relationship between electrons and atoms is vital for understanding the nature of matter. Electrons are arranged in specific energy levels or shells around the nucleus, and each shell can only hold a certain number of electrons. The arrangement of electrons in an atom determines its electronic configuration, which in turn affects its behavior and reactivity.
The distribution of electrons in an atom is governed by the rules of quantum mechanics. Electrons occupy different orbitals within each energy level, and these orbitals have different shapes and orientations. The arrangement of electrons in orbitals is represented by electron configurations, which provide a framework for understanding the behavior and properties of elements.
The interaction between electrons and atoms is responsible for various chemical phenomena. The exchange or sharing of electrons between atoms leads to the formation of chemical bonds, which hold atoms together in molecules and compounds. Furthermore, the movement of electrons within an atom contributes to the emission and absorption of light, giving rise to the field of spectroscopy.
In summary, electrons are fundamental particles that orbit around the nucleus of an atom. Their arrangement within an atom determines its electronic configuration and plays a crucial role in determining its properties and behavior. The study of electrons and their relationship to atoms is essential for understanding the nature of matter and chemical reactions.
The significance of Chapter 4 on the study of electrons in atoms
In Chapter 4 of the textbook, the focus is on the study of electrons in atoms. This chapter is of great significance in understanding the behavior and properties of atoms, as electrons play a fundamental role in determining the chemical properties and reactivity of elements.
The chapter starts by introducing the concept of electromagnetic radiation and the characteristics of waves. This understanding is crucial in grasping how electrons move around the nucleus of an atom and how they interact with each other and with external forces. By studying the behavior of electrons, scientists can gain insights into the structure and stability of atoms.
One of the key topics covered in this chapter is the quantization of energy levels in atoms. This concept, known as the Bohr model, revolutionized our understanding of the arrangement of electrons in atoms. The chapter explores the different energy levels and sublevels that electrons occupy, and how these arrangements give rise to the distinct electronic configurations observed in the periodic table.
Furthermore, the chapter delves into the phenomena of electron spin and electron orbitals. Electron spin is an intrinsic property of electrons, and it influences their behavior and magnetic properties. Understanding electron spin is crucial in comprehending the principles behind electron pairing and the filling of electron orbitals.
Overall, Chapter 4 provides the foundation for understanding the complex behavior of electrons in atoms. By studying this chapter, students can gain a deeper understanding of atomic structure and the relationship between electron configuration and the chemical properties of elements. This knowledge is essential for the study of chemistry and is applicable to various fields such as materials science, biochemistry, and quantum physics.
Understanding the properties of electrons in atoms
Electrons are subatomic particles that orbit around the nucleus of an atom. They possess negative charge and are essential for determining the chemical behavior and properties of an element. Understanding the properties of electrons in atoms is crucial for grasping the fundamentals of atomic structure and the principles of chemical bonding.
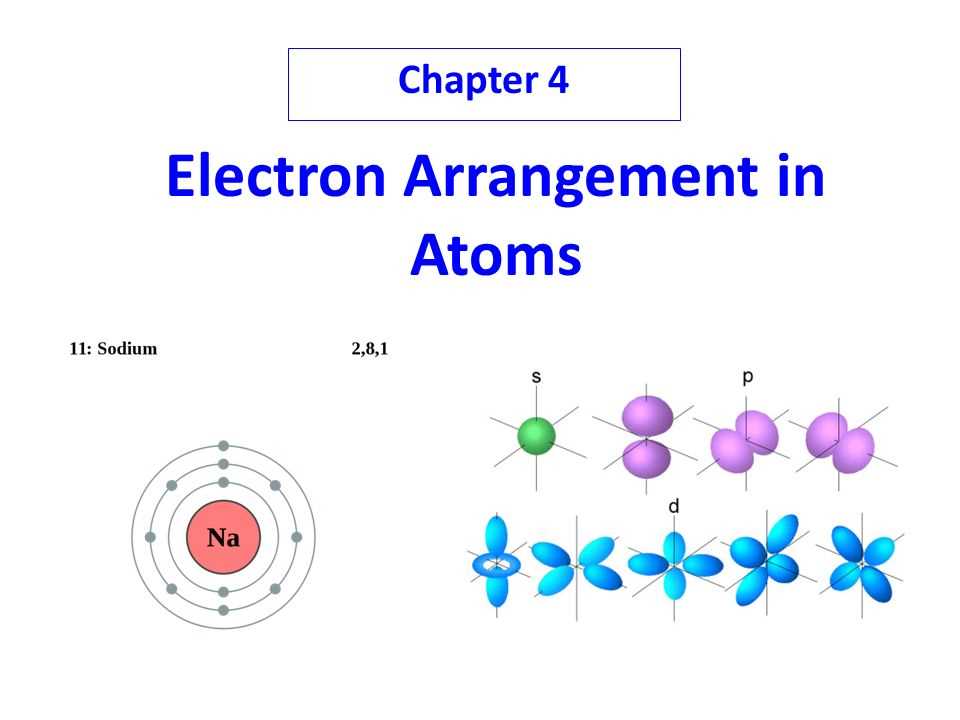
One key property of electrons is their energy levels, or shells, which determine their distance from the nucleus. Electrons occupy specific energy levels, with the lowest energy level being closest to the nucleus. Each energy level can hold a certain number of electrons, with the first level holding a maximum of 2 electrons, the second level holding up to 8 electrons, and so on. This arrangement is known as the electron configuration of an atom.
The arrangement of electrons within the energy levels follows the Aufbau principle, which states that electrons fill the lowest available energy levels first before filling higher energy levels. The Pauli exclusion principle states that each energy level can hold a maximum of two electrons, and they must have opposite spins. This means that if one electron in a specific energy level has a spin in one direction, the other electron in the same energy level must have a spin in the opposite direction.
Another crucial property of electrons is their ability to occupy orbitals within the energy levels. Orbitals can be thought of as regions in space where electrons are most likely to be found. Each energy level has a specific number of orbitals, and each orbital can hold a maximum of two electrons. The shapes of the orbitals vary, ranging from spherical (s-orbitals) to dumbbell-shaped (p-orbitals) and more complex shapes for higher energy levels.
In summary, understanding the properties of electrons in atoms involves grasping concepts related to energy levels, electron configuration, and orbital arrangement. These properties provide insights into the behavior and interaction of atoms, laying the foundation for studying chemical reactions, bonding, and various other aspects of chemistry.
Key concepts and principles in Chapter 4: electrons in atoms
The fourth chapter of the chemistry textbook focuses on electrons in atoms. This chapter explores the behavior and properties of electrons within the atomic structure. Understanding these key concepts and principles is crucial in comprehending the structure of matter and the interactions between atoms.
Electron Configuration
One of the central concepts covered in this chapter is electron configuration. Electrons occupy specific energy levels and sublevels within an atom. The electron configuration provides a systematic way of representing the arrangement of electrons, using a series of notations such as the noble gas configuration and orbital diagrams.
Quantum Mechanical Model
The quantum mechanical model is another fundamental concept discussed in this chapter. It is a mathematical representation that describes the behavior of electrons as both particles and waves. The model helps explain various phenomena, such as the discrete energy levels and the uncertainty principle. It also introduces the concept of orbitals, which are regions of space where electrons are likely to be found.
Electron Spin and Electron Pairing
The concept of electron spin and electron pairing is crucial to understanding electron configurations. Electrons possess a property called spin, which can be either up or down. According to the Pauli exclusion principle, no two electrons in an atom can have the same four quantum numbers. Therefore, when filling orbitals, electrons will pair up in the same orbital only when all the orbitals in that sublevel are half-filled.
Periodic Trends
Understanding the behavior of electrons in atoms allows for the exploration of periodic trends. The periodic table is organized based on these trends, such as atomic size, ionization energy, and electronegativity. Electron configuration plays a significant role in explaining these trends, as elements within the same group often have similar electron configurations.
Overall, Chapter 4 provides a foundation for understanding the fundamental principles and concepts related to electrons in atoms. Mastering these key ideas is essential for further exploration of chemical bonding, reactions, and the properties of different elements on the periodic table.
Exploring electron configurations and their importance in chemistry
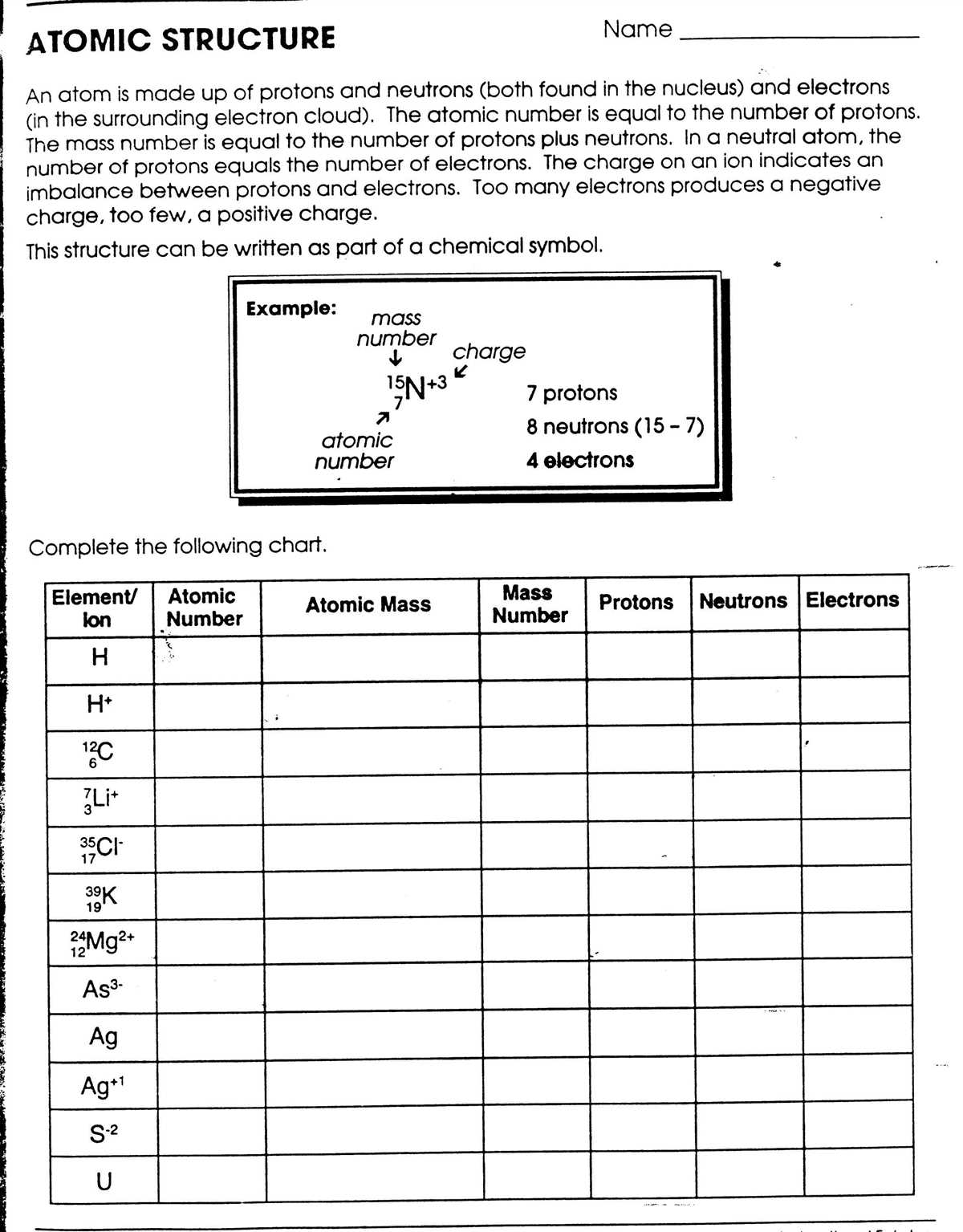
The study of electron configurations is a fundamental aspect of chemistry, as it provides insight into the behavior and properties of atoms. Electron configurations describe the arrangement of electrons within an atom’s energy levels and sublevels. Understanding electron configurations enables chemists to predict an element’s chemical reactivity, bonding behavior, and overall stability.
Electron configurations follow a specific set of rules based on the principles of quantum mechanics. The Aufbau principle states that electrons occupy the lowest available energy levels before filling higher energy levels. This concept is crucial in determining the order in which electrons fill the sublevels and shells.
The electron configuration of an atom is represented using a series of numbers, letters, and superscripts. The numbers represent the principal energy level (n), while the letters represent the sublevel (s, p, d, f). The superscripts indicate the number of electrons present in each sublevel. For example, the electron configuration of carbon is 1s² 2s² 2p², indicating that there are two electrons in the 1s sublevel, two electrons in the 2s sublevel, and two electrons in the 2p sublevel.
Electron configurations are important in understanding the periodic trends of the elements. Elements within the same group (vertical column) of the periodic table often have similar electron configurations, leading to similar chemical behavior. Additionally, electron configurations help determine an element’s valence electrons, which are crucial in predicting bonding patterns and the formation of chemical compounds.
In summary, electron configurations play a vital role in understanding the behavior, properties, and reactivity of atoms. They provide a framework for predicting chemical behavior, bonding patterns, and the arrangement of electrons within an atom. By studying electron configurations, chemists can gain valuable insight into the fascinating world of chemistry.
The Role of Electron Shells and Energy Levels in Atoms
The arrangement of electrons in an atom is governed by the concept of electron shells and energy levels. These shells and energy levels play a crucial role in determining the properties and behavior of an atom.
Electron Shells: Electron shells are regions surrounding the nucleus of an atom where electrons are most likely to be found. These shells are represented by the principal quantum numbers (n=1, 2, 3, etc.). The first shell, closest to the nucleus, has the lowest energy level (n=1), followed by the second shell (n=2), and so on. Each shell can hold a maximum number of electrons based on its energy level.
Energy Levels: Energy levels represent the amount of energy an electron possesses when it occupies a particular shell. The energy of an electron increases as it moves to higher energy levels. Electrons tend to occupy the lowest available energy levels before filling the higher ones, according to the Aufbau principle.
Electron shells and energy levels together determine the electronic configuration and chemical behavior of atoms. The outermost shell, known as the valence shell, plays a crucial role in the formation of chemical bonds and interactions with other atoms. Elements with similar valence shell configurations exhibit similar chemical properties.
In summary, the concept of electron shells and energy levels provides a framework for understanding the arrangement and behavior of electrons in atoms. These shells and energy levels determine which orbitals are occupied and how electrons are distributed around the nucleus. The understanding of electron shells and energy levels has been fundamental in explaining various phenomena in chemistry and physics.