Understanding the concept of pH is crucial in the fields of chemistry and biology. pH is a measure of the acidity or alkalinity of a substance, and it plays a significant role in determining the behavior and interactions of various chemicals and organisms. To calculate pH accurately, one needs to have a solid understanding of the principles and equations involved.
In this article, we will provide a comprehensive answer key for the “Calculating pH” POGIL activity. POGIL, which stands for Process Oriented Guided Inquiry Learning, is a popular instructional approach that emphasizes active learning and critical thinking.
The “Calculating pH” activity focuses on teaching students how to calculate the pH of a given substance based on its concentration of hydrogen ions (H+). By going through the answer key, students will gain a deeper understanding of the pH scale, logarithms, and the relationship between acid and base concentrations.
Throughout the answer key, we will provide detailed step-by-step explanations for each question in the activity. This will help students comprehend the thought process required to solve pH calculations and clarify any misconceptions they may have had. Additionally, we will offer valuable tips and shortcuts to simplify the calculations and improve accuracy.
Calculating pH POGIL Answer Key
The pH scale is used to measure the acidity or alkalinity of a solution. pH is calculated using the formula: pH = -log[H+], where [H+] represents the concentration of hydrogen ions in the solution. In order to calculate pH, it is necessary to know the concentration of hydrogen ions.
The POGIL activity on calculating pH provides a step-by-step approach to determine the pH of various solutions. The activity includes a series of questions and calculations to guide students through the process of calculating pH. The answer key for the POGIL activity contains the correct answers and explanations for each question.
- Question 1: Calculate the pH of a solution with a hydrogen ion concentration of 0.001 M.
- Answer: To calculate pH, we take the negative logarithm of the hydrogen ion concentration. pH = -log(0.001) = 3.
- Question 2: Calculate the pH of a solution with a hydrogen ion concentration of 1 x 10^-5 M.
- Answer: pH = -log(1 x 10^-5) = 5.
The answer key for the POGIL activity provides students with the correct answers and explanations for each question. This allows students to check their work and understand the process of calculating pH. It is important for students to practice calculating pH in order to develop a strong understanding of the concept and its applications in chemistry.
Understanding pH Calculations

The concept of pH is essential in understanding the chemical properties of solutions. pH is a measure of the acidity or basicity of a solution, and it is calculated using the concentration of hydrogen ions (H+) in the solution. The pH scale ranges from 0 to 14, with values below 7 indicating acidity, values above 7 indicating alkalinity, and a pH of 7 indicating neutrality. To calculate the pH of a solution, several steps need to be followed, including determining the concentration of hydrogen ions and taking the negative logarithm of this concentration.
To calculate the pH of a solution, the first step is to determine the concentration of hydrogen ions by using the formula pH = -log[H+]. This equation involves taking the negative logarithm of the hydrogen ion concentration. The hydrogen ion concentration is usually given in moles per liter (M) or molar concentration. By taking the negative logarithm of this value, the pH value is obtained.
To perform the calculation, it is crucial to have accurate and precise values for the hydrogen ion concentration. This can be obtained experimentally through various methods, such as titration or using a pH meter. Alternatively, if the concentration of a strong acid or a strong base is known, the concentration of hydrogen ions can be calculated using stoichiometry.
Understanding pH calculations is essential in various scientific disciplines, including chemistry, biology, and environmental science. It allows scientists to accurately describe and measure the acidity or alkalinity of solutions. Additionally, pH calculations are used in many practical applications, such as water treatment, food and beverage industry, and pharmaceuticals. Being able to calculate and interpret pH values is a fundamental skill for anyone working in these fields.
Factors Affecting pH

The pH of a solution is a measure of its acidity or alkalinity. It is determined by the concentration of hydrogen ions (H+) present in the solution. Several factors can affect the pH of a solution, including the presence of acids or bases, the concentration of these substances, temperature, and the presence of buffers.
Acids and bases are substances that can donate or accept hydrogen ions, respectively. When an acid is added to a solution, it increases the concentration of hydrogen ions and lowers the pH. On the other hand, when a base is added, it decreases the concentration of hydrogen ions and raises the pH. The strength and concentration of acids and bases play a role in determining the extent of the pH change.
Temperature can also affect pH. In general, as temperature increases, the pH of acidic solutions decreases, while the pH of basic solutions increases. This is because temperature influences the dissociation of acids and bases, affecting the concentration of hydrogen ions in the solution. Additionally, changes in temperature can affect the solubility of certain compounds, leading to changes in pH.
Buffers are substances that can resist changes in pH when small amounts of acids or bases are added. They are composed of weak acids or bases and their respective conjugate bases or acids. Buffers help maintain the pH of a solution within a narrow range by accepting or donating hydrogen ions to prevent significant changes in pH. They are commonly used in biological systems to maintain the pH necessary for enzymatic reactions and other cellular processes.
In conclusion, the pH of a solution is influenced by the presence and concentration of acids and bases, temperature, and the presence of buffers. Understanding these factors is important in various fields, including chemistry, biology, and environmental science, as pH plays a crucial role in many chemical and biological processes.
Step-by-Step Guide to Calculating pH

Calculating the pH of a solution is an important concept in chemistry. pH measures the acidity or alkalinity of a solution on a scale of 0 to 14, with 0 being the most acidic and 14 being the most alkaline. Here is a step-by-step guide on how to calculate pH:
- Find the concentration of hydrogen ions: pH is a measure of the concentration of hydrogen ions in a solution. To calculate the pH, you first need to determine the concentration of hydrogen ions in the solution. This can be done by using the formula: pH = -log[H+], where [H+] represents the concentration of hydrogen ions.
- Convert the concentration to scientific notation: If the concentration is not already in scientific notation, convert it to this format. Scientific notation is written as a number between 1 and 10 multiplied by a power of 10. For example, a concentration of 0.001 M would be written as 1 x 10^-3 M.
- Calculate the negative logarithm: Take the negative logarithm of the hydrogen ion concentration using a calculator or logarithm table. The negative sign in the formula accounts for the inverse relationship between hydrogen ion concentration and pH. For example, if the concentration is 1 x 10^-3 M, the negative logarithm would be -log(1 x 10^-3) = 3.
- Determine the pH: The negative logarithm calculated in the previous step represents the pH of the solution. In the example given, the pH would be 3.
Remember that pH is a logarithmic scale, so each unit change in pH represents a tenfold change in hydrogen ion concentration. A higher pH indicates a more alkaline solution, while a lower pH indicates a more acidic solution. By following these steps, you can accurately calculate the pH of a solution based on its hydrogen ion concentration.
Common Mistakes in pH Calculations
Calculating pH is an essential skill in chemistry, but it can be prone to errors if certain precautions are not taken. Here are some common mistakes to avoid when performing pH calculations.
1. Not accounting for the autoprotolysis of water: One of the most fundamental concepts in pH calculations is the autoprotolysis of water, which allows for the generation of both protons (H+) and hydroxide ions (OH-) in aqueous solutions. Failing to consider this equilibrium can lead to incorrect pH values. It is important to remember that the pH of pure water at 25°C is 7, as it represents a neutral solution.
2. Using the wrong logarithm base: In pH calculations, the negative logarithm of the hydrogen ion concentration (pH = -log[H+]) is used. However, it is crucial to use the correct base for the logarithm. The commonly used base for pH calculations is 10, resulting in a logarithm known as the common logarithm (logarithm base 10). Using a different base, such as natural logarithm (logarithm base e), can yield incorrect pH values.
3. Ignoring significant figures: pH calculations often involve several steps and multiple measurements. It is important to pay attention to significant figures throughout the calculations. Rounding errors and improper use of significant figures can result in inaccurate pH values. It is crucial to carry out all calculations with the appropriate number of significant figures and round the final pH value to the correct number of decimal places.
4. Forgetting to account for temperature: pH calculations are temperature-dependent, and the pH of a solution can change with temperature due to variations in the autoprotolysis constant of water. It is important to account for temperature changes in pH calculations, especially when comparing pH values obtained at different temperatures. The use of a temperature correction factor or the conversion to a standard temperature can help to obtain accurate pH values.
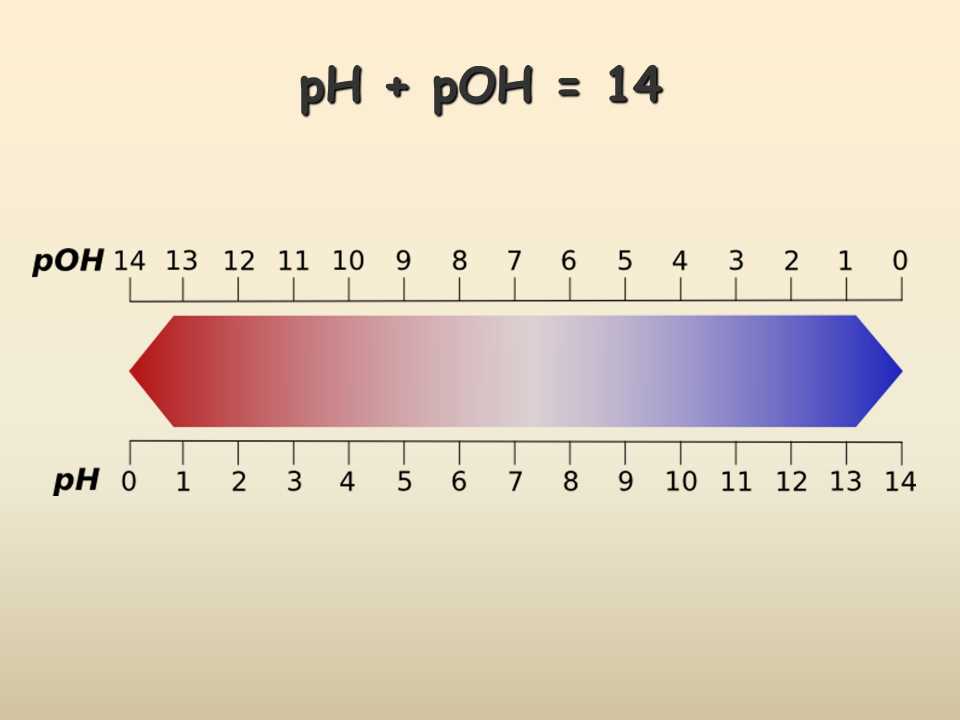
5. Confusing pH and pOH: pH and pOH are logarithmic scales that represent the acidity and alkalinity of a solution, respectively. It is essential to differentiate between these two scales and understand their relationship. pH and pOH are related by the equation pH + pOH = 14. Mistakenly using the wrong scale can lead to erroneous pH calculations.
In conclusion, avoiding these common mistakes can greatly improve the accuracy of pH calculations. Remembering to account for water autoprotolysis, using the correct logarithm base, paying attention to significant figures, considering temperature effects, and distinguishing between pH and pOH are essential for obtaining reliable pH values in chemical calculations.
Practice Problems and Examples

Now that we have learned the basics of calculating pH, let’s practice our skills with some examples and problems. These will help solidify our understanding of the concept and improve our problem-solving abilities.
Example 1:
Calculate the pH of a solution with a hydrogen ion concentration of 0.001 M.
Solution:
- Use the equation pH = -log[H+].
- -log(0.001) = 3.
Therefore, the pH of the solution is 3.
Example 2:
A solution has a pH of 6. What is the hydrogen ion concentration?
Solution:
- Use the equation [H+] = 10^(-pH).
- [H+] = 10^(-6).
- [H+] = 0.000001 M.
Therefore, the hydrogen ion concentration of the solution is 0.000001 M.
Practice Problem 1:
Calculate the pH of a solution with a hydrogen ion concentration of 0.00001 M.
Practice Problem 2:
A solution has a pH of 4. What is the hydrogen ion concentration?
Answer Key:
- pH = 5.
- [H+] = 0.0001 M.
By practicing these problems and examples, we are able to reinforce our knowledge of calculating pH. Remember to use the appropriate equations and formulas, and don’t forget to take the negative logarithm of the hydrogen ion concentration. Keep practicing, and soon you will become a master at calculating pH!