Acids and bases are fundamental concepts in chemistry that play a crucial role in our everyday lives. Understanding the properties and behavior of acids and bases is essential for students studying this subject. To reinforce their knowledge and test their understanding, teachers often provide worksheets that explore various aspects of acids and bases. Having access to the answers for these worksheets is incredibly helpful for students as it allows them to check their work and learn from their mistakes.
Fortunately, there are resources available that provide acid and bases worksheet answers in PDF format. These PDFs contain comprehensive solutions to the worksheet questions, giving students a valuable tool to enhance their learning. By referring to the answers, students can verify if they have solved the problems correctly and gain insight into the thought process behind each solution.
The PDF format is particularly advantageous as it allows students to easily access and save the answers on their devices. They can refer to the answers whenever they need to review or revise the concepts related to acids and bases. Additionally, the availability of PDF answer sheets means that students can work on the worksheets at their own pace, independently studying and solving the problems without the need for continuous teacher supervision.
Overall, having access to acids and bases worksheet answers in PDF format provides students with a valuable resource to enhance their understanding of this important chemistry topic. It promotes independent learning and allows students to check their work, identify areas for improvement, and gain a deeper comprehension of the subject. These answers are an invaluable tool for students striving to excel in their chemistry studies.
Acids and Bases Worksheet Answers PDF
Acids and bases are important concepts in chemistry. They are substances that can donate or accept protons (H+) respectively. Understanding the properties and reactions of acids and bases is crucial in various applications, from everyday household cleaning products to industrial processes. To aid in the learning and practice of these concepts, worksheets are often provided to students. These worksheets usually include questions and exercises that test the understanding of key concepts and principles.
Acids and bases worksheet answers PDFs are a valuable resource for students and teachers alike. They provide a comprehensive guide to the correct answers for each question or exercise on the worksheet. By having access to the answers, students can easily check their work and understand where they may have made mistakes. Teachers can also use the answers as a reference to assess student performance and provide feedback. This ensures that students have a clear understanding of the topic and can apply their knowledge effectively.
The format of the acids and bases worksheet answers PDFs typically includes a numbered list of questions or exercises followed by the corresponding answers. The answers are often accompanied by explanations or step-by-step solutions to help students comprehend the reasoning behind each answer. This approach not only provides immediate feedback but also aids in the learning process by reinforcing key concepts and principles.
In conclusion, acids and bases worksheet answers PDFs serve as a valuable tool for both students and teachers in the learning and teaching of the topic. They provide a comprehensive guide to the correct answers and explanations for each question or exercise. By using these resources, students can check their work, identify areas of improvement, and enhance their understanding of acids and bases. Teachers can also use them to assess student performance and provide targeted feedback. Overall, acids and bases worksheet answers PDFs contribute to a more effective learning experience.
The Importance of Acids and Bases in Chemistry
Acids and bases are fundamental concepts in chemistry that play a crucial role in various chemical reactions and everyday life. These substances have unique properties and are involved in countless processes, making them essential to study and understand in the field of chemistry.
Acids are substances that release hydrogen ions (H+) when dissolved in water, while bases are substances that release hydroxide ions (OH-) when dissolved in water. These ions are responsible for the characteristic properties of acids and bases, such as the ability to react with other substances and change their pH.
One of the primary importance of acids and bases is their role in neutralization reactions. When an acid reacts with a base, they undergo a chemical reaction called neutralization, where the acidic and basic properties are canceled out, resulting in the formation of salt and water. This process is utilized in various applications, such as the production of fertilizers, pharmaceuticals, and food processing.
Acids and bases are also essential in maintaining the pH balance in biological systems. Many biological processes, such as enzyme activity and cellular functions, are highly sensitive to pH levels. Acids and bases help regulate these pH levels and maintain the optimal conditions for these processes to occur.
Furthermore, acids and bases are widely used in industries and technology. They are crucial in metal etching, electroplating, and the production of batteries. They also play a vital role in wastewater treatment, as acids and bases are used to adjust pH levels and remove contaminants.
In summary, acids and bases are fundamental concepts in chemistry that have significant importance in various aspects of life, from chemical reactions to biological processes and industrial applications. Understanding the properties and behavior of acids and bases is essential for further advancements in chemistry and other scientific fields.
Understanding the Properties of Acids
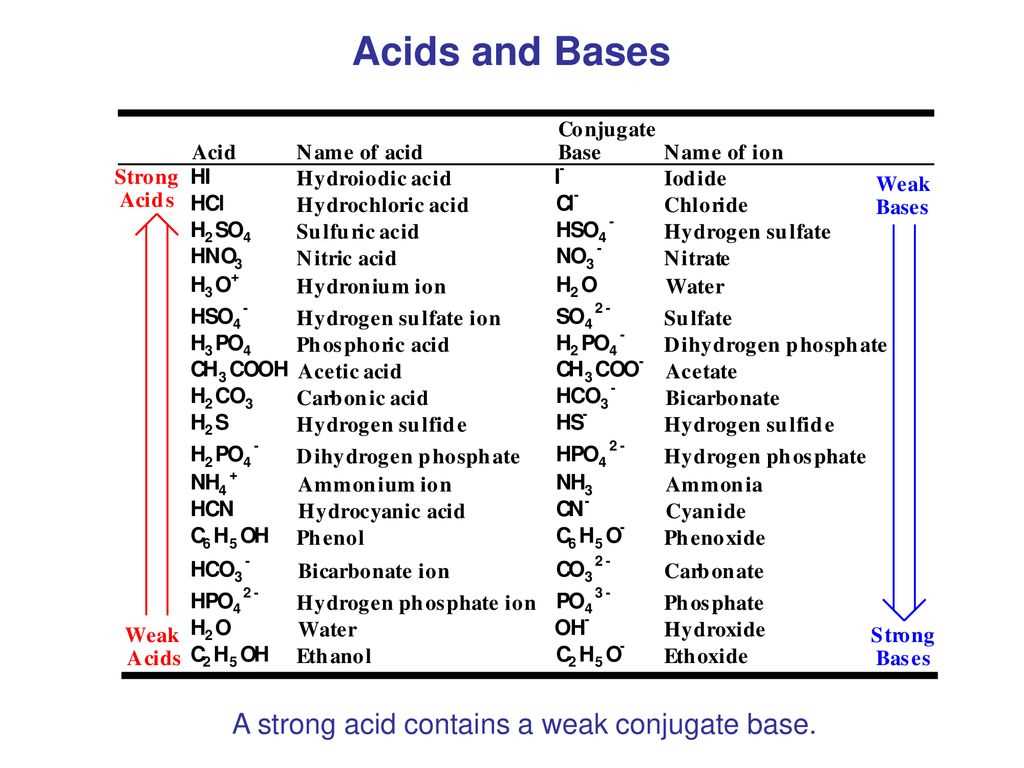
Acids are a group of chemical substances that have distinct properties. By understanding these properties, we can better comprehend the behavior and characteristics of acids.
Acidic taste: One of the most recognizable properties of acids is their sour taste. This characteristic is often used as a way to identify whether a substance is an acid or not. Acids such as lemon juice and vinegar have a distinct sour and tangy flavor.
Conductivity: Acids are also known for their ability to conduct electricity. When an acid dissolves in water, it forms ions that allow electric current to flow. This property is important in various industrial applications, such as in batteries and electroplating processes.
Corrosive nature: Acids have the ability to corrode or eat away certain materials. This is due to their high reactivity with metals and other substances. For example, hydrochloric acid is commonly used in industries to clean and etch metals.
pH level: Acids have a pH value less than 7. The pH scale is a measure of the acidity or alkalinity of a solution. Acids have a higher concentration of hydrogen ions (H+) compared to hydroxide ions (OH-), leading to a lower pH value.
Reaction with bases: Another important property of acids is their reaction with bases. Acids and bases neutralize each other, forming water and a salt. This reaction, known as neutralization, is often used in various chemical processes and in common household cleaning products.
In conclusion, understanding the properties of acids allows us to identify and distinguish these chemical substances. It also helps us to comprehend their behavior and their interactions with other substances, such as bases. Through this knowledge, we can better utilize and manage acids in various fields, from industry to everyday applications.
Exploring the Characteristics of Bases
Bases are a class of substances that possess specific characteristics and play a crucial role in various chemical reactions. Understanding the properties and behavior of bases is essential for comprehending their role in chemistry and everyday life.
One of the defining characteristics of bases is their ability to accept protons or donate pairs of electrons. They are considered the opposite of acids, which donate protons or accept pairs of electrons. Bases have a pH value greater than 7 and can neutralize acids to form salts and water.
Bases display certain physical and chemical properties that distinguish them from other substances. Physically, bases can range from being solid, like sodium hydroxide (NaOH), to liquid, like ammonia (NH3), or even gaseous, like ammonia water (NH4OH). Bases also have a characteristic taste and feel slippery to the touch.
Chemically, bases are known to react with acids to form neutral substances. This process is called neutralization and is a fundamental concept in acid-base chemistry. Bases can also react with certain metals, such as zinc or aluminum, to produce hydrogen gas. Additionally, bases can conduct electricity when dissolved in water due to the presence of ions.
Bases have applications in various fields, such as medicine, industry, and agriculture. Some common examples of bases include household cleaning products like bleach, antacids used to neutralize excess stomach acid, and fertilizers used to improve soil pH. Bases are also essential in laboratory settings for carrying out chemical reactions and maintaining specific conditions.
In conclusion, exploring the characteristics of bases is crucial to understanding their role and applications in chemistry. Bases possess unique properties that allow them to react with acids, conduct electricity, and have various physical forms. The study of bases contributes to a deeper understanding of chemical reactions and their impact on our daily lives.
Common Reactions Involving Acids and Bases

Acids and bases are two important types of substances that can undergo various reactions. These reactions are essential in many chemical processes, ranging from everyday household activities to industrial production.
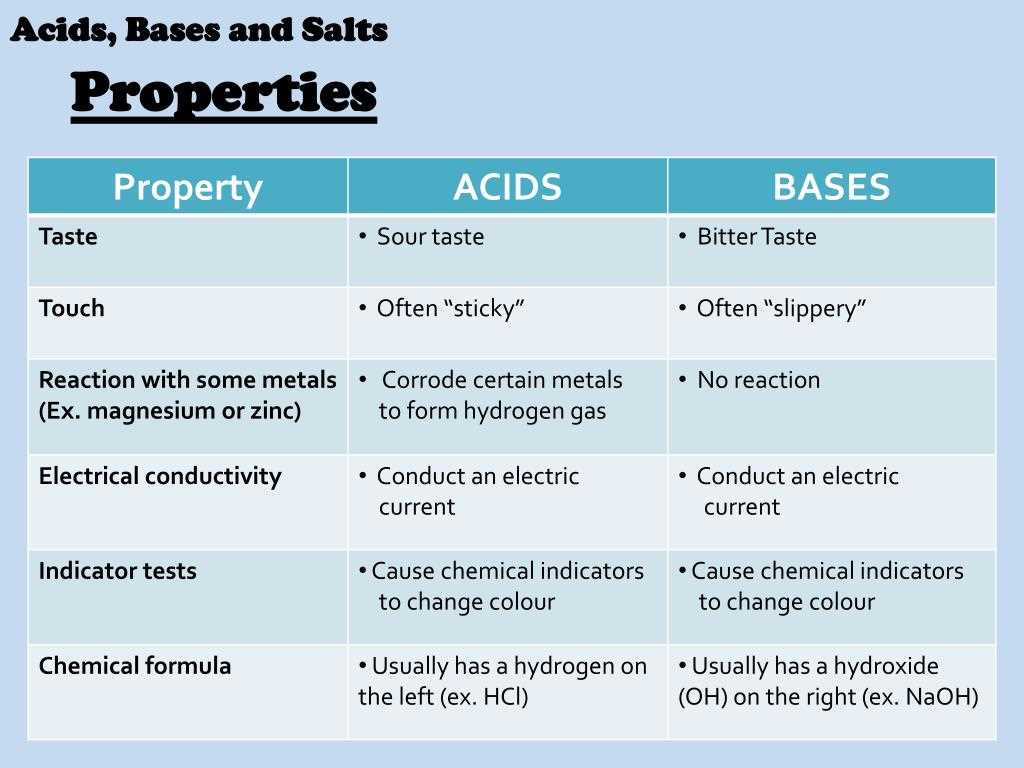
Neutralization Reactions: One of the most common reactions involving acids and bases is neutralization. In a neutralization reaction, an acid and a base react to form a salt and water. The acid donates a proton (H+) to the base, resulting in the formation of water. For example, when hydrochloric acid (HCl) reacts with sodium hydroxide (NaOH), the products formed are sodium chloride (NaCl) and water (H2O). Neutralization reactions are often used to neutralize the acidic or basic properties of a solution.
Corrosion Reactions: Acids and bases can cause corrosion on different materials. For example, strong acids like sulfuric acid can react with metals, such as iron, to produce metal oxides and hydrogen gas. This reaction is known as a metal corrosion reaction. On the other hand, bases like sodium hydroxide can react with fats and oils, leading to saponification, a process used in soap-making. These corrosion reactions have important implications in industries like construction, manufacturing, and cleaning.
Acid-Base Titration: Another important reaction involving acids and bases is acid-base titration. This is a method used to determine the concentration of an acid or a base in a solution. In a titration, a known volume of a solution with a known concentration of an acid or base (often called the titrant) is added to a solution with an unknown concentration of the other (often called the analyte). The reaction between the acid and base is monitored using an indicator or pH meter until it reaches the equivalence point, where the moles of acid and base are stoichiometrically equivalent. From the volume and concentration of the titrant used, the concentration of the analyte can be calculated.
In summary, acids and bases can participate in different reactions, including neutralization, corrosion, and acid-base titration. These reactions have practical applications in various fields and are important in understanding the chemistry behind many everyday phenomena.
How to Solve Acid-Base Equations
Solving acid-base equations involves understanding the different components of the equation and balancing them to achieve a neutral outcome. The following steps can help guide you through the process:
- Identify the reactants and products: Begin by identifying the acid and base in the equation. Reactants are the substances that are present before the reaction takes place, while products are the substances that are formed after the reaction.
- Write the balanced chemical equation: Write the chemical equation by representing the reactants and products using their respective formulas and symbols. Make sure the equation is balanced in terms of both atoms and charge.
- Determine the type of reaction: Determine whether the reaction is a neutralization reaction, where an acid and a base combine to form a salt and water, or another type of acid-base reaction.
- Balance the equation: Balance the equation by adjusting the coefficients in front of the formulas to ensure an equal number of atoms on both sides. This step may require trial and error.
- Check the balance: Double-check that the equation is balanced by counting the total number of atoms of each element on both sides of the equation. The number of each type of atom should be the same.
- Calculate the concentration or volume: If the problem provides additional information, such as the concentration or volume of one of the solutions, use this information to calculate the concentration or volume of the other solution using stoichiometry.
- Verify the solution: Finally, verify your solution by substituting the values you have calculated back into the equation. Ensure that the mass, charge, and other properties are properly balanced on both sides.
By following these steps, you can effectively solve acid-base equations and determine the necessary quantities and concentrations of reactants and products.
Accessing Acid and Bases Worksheet Answers in PDF Format
As you work through your acids and bases worksheet, you may find it helpful to have access to the answers in a PDF format. This can make it easier to check your work and ensure that you’re on the right track. Luckily, obtaining acid and bases worksheet answers in PDF format is a simple process.
One way to access acid and bases worksheet answers in PDF format is to check with your teacher or instructor. They may have a copy of the answers or know where to find them. Additionally, they may be able to provide you with a PDF version of the worksheet and answers directly.
If your teacher does not have the answers readily available, another option is to search online. Many educational websites and forums offer resources for students studying acids and bases, including worksheets and answer keys. Simply inputting “acids and bases worksheet answers PDF” into a search engine should yield multiple results to choose from.
It’s important to note that not all websites or sources may have accurate or reliable answers. Therefore, it’s always a good idea to cross-check your answers with multiple sources to ensure their correctness. Additionally, discussing any uncertainties or questions with your teacher or classmates can provide further clarification and understanding.
In conclusion, accessing acid and bases worksheet answers in PDF format can be done by checking with your teacher or searching online. Remember to use reliable sources and cross-check your answers to ensure accuracy. Happy learning!