
Preparing for your organic chemistry lab final exam can be challenging, but with the right study guide, you can approach it with confidence. The final exam is designed to test your understanding of various organic chemistry lab techniques, concepts, and principles. It will require you to demonstrate your ability to perform experiments, analyze data, and interpret results.
One key aspect of preparing for the final exam is understanding the different types of organic chemistry lab techniques and their applications. This includes knowledge of extraction techniques, distillation, recrystallization, and chromatography methods. Familiarizing yourself with these techniques and their underlying principles will help you approach lab-related questions on the exam more effectively.
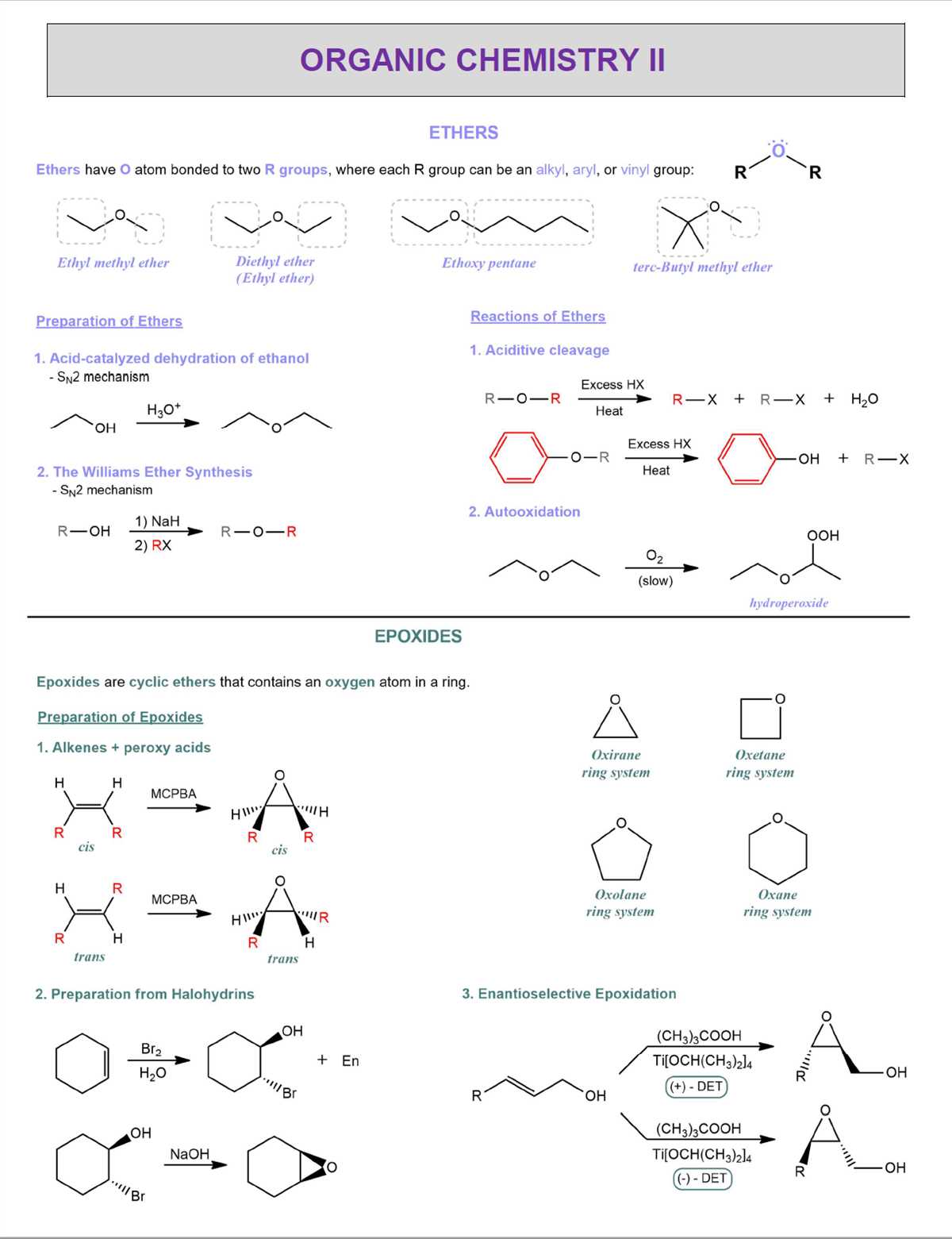
It is also important to review the different types of organic compounds you have encountered throughout the lab course and understand their properties, reactions, and synthesis methods. This includes alkanes, alkenes, alkynes, alcohols, ethers, and more. By studying the structures, naming conventions, and reactions associated with these compounds, you will be better equipped to analyze and solve problems related to them on the final exam.
Understanding the basics of organic chemistry lab

Organic chemistry lab is a key component of any organic chemistry course and plays a crucial role in developing practical skills and understanding of chemical reactions. In the lab, students have the opportunity to apply the concepts they have learned in lectures and gain hands-on experience with various laboratory techniques.
One of the first things students learn in organic chemistry lab is how to handle and work with chemicals safely. This includes understanding the properties of chemicals, proper handling and storage, and following safety protocols. Safety goggles, lab coats, and gloves are commonly used to minimize the risk of accidents and exposure to hazardous substances.
Another important aspect of organic chemistry lab is learning how to properly set up and perform experiments. This involves setting up apparatus such as glassware, heating devices, and measuring devices, and following experimental procedures precisely. Students are taught how to measure and mix chemicals accurately, as well as how to observe and record data effectively.
Key techniques that students learn include:
- Extraction: the process of separating one or more compounds from a mixture using solvents.
- Distillation: a technique used to separate mixtures based on differences in boiling points.
- Crystallization: the process of purifying a solid compound by dissolving it in a solvent and allowing it to slowly crystallize.
- Chromatography: a method used to separate and analyze mixtures based on differences in their affinity for a stationary phase and a mobile phase.
Understanding the basics of organic chemistry lab is essential for students to be able to perform experiments effectively and safely. By mastering laboratory techniques and procedures, students can gain a better understanding of the principles and concepts of organic chemistry, as well as develop important skills that are valuable in future scientific research or careers in the field.
Key Concepts and Techniques in Organic Chemistry Lab
In the organic chemistry lab, students learn and apply various concepts and techniques to conduct experiments and analyze organic compounds. These experiments are designed to reinforce theoretical understanding and develop practical skills necessary for working with organic substances.
1. Synthesis and purification: One of the key concepts in organic chemistry lab is the ability to synthesize organic compounds and purify them. Students are taught different methods of synthesizing compounds, such as using Grignard reagents or condensation reactions. They also learn purification techniques like recrystallization, distillation, and extraction, which help to obtain pure compounds for further analysis.
2. Spectroscopic analysis: Another important aspect of organic chemistry lab is the use of spectroscopic techniques for compound identification. Students learn to interpret and analyze data from techniques such as infrared spectroscopy (IR), proton nuclear magnetic resonance (NMR), and mass spectrometry. These techniques provide valuable information about the functional groups and connectivity of atoms within the organic compounds.
3. Safety and handling: Safety is a paramount consideration in the organic chemistry lab. Students are taught proper handling and storage of chemicals, as well as the use of safety equipment such as goggles, gloves, and lab coats. They learn to follow safety protocols and handle hazardous materials with caution, ensuring the well-being of themselves and their colleagues.
4. Experimental design and data analysis: In the lab, students are encouraged to develop their experimental design skills. They learn to plan and execute experiments, considering factors such as reaction conditions, reagent concentrations, and reaction times. After conducting experiments, students analyze data and draw appropriate conclusions, helping them to understand the reaction mechanisms and their implications.
Common Organic Reactions and Mechanisms
The study of organic chemistry involves understanding various reactions that can occur between organic compounds. These reactions can be classified into different categories based on the type of bond formation or breakage that takes place. A few common organic reactions and their mechanisms are discussed below:
1. Substitution Reactions:
Substitution reactions involve the replacement of one atom or group of atoms in a molecule with another atom or group of atoms. These reactions typically occur in the presence of a nucleophile, which attacks the electrophilic carbon center and displaces the leaving group. This results in the formation of a new bond and the removal of the leaving group. The most common types of substitution reactions include nucleophilic substitution and electrophilic substitution.
2. Addition Reactions:
Addition reactions involve the addition of atoms or groups of atoms to a molecule, resulting in the formation of a new bond. These reactions typically occur in the presence of an electrophile, which reacts with a nucleophile to form a new bond. Addition reactions can be classified into different types based on the nature of the electrophile and the nucleophile involved. Some common examples include electrophilic addition, nucleophilic addition, and radical addition.
3. Elimination Reactions:
Elimination reactions involve the removal of atoms or groups of atoms from a molecule, resulting in the formation of a double or triple bond. These reactions typically occur in the presence of a base or an acid, which abstracts a proton from the molecule, leading to the formation of a new bond. Elimination reactions can be classified into different types based on the mechanism involved, such as E1, E2, and E1cb.
4. Oxidation and Reduction Reactions:

Oxidation and reduction reactions involve the transfer of electrons between molecules or atoms. Oxidation reactions involve the loss of electrons, while reduction reactions involve the gain of electrons. These reactions are typically carried out using oxidizing or reducing agents, such as potassium permanganate for oxidation and sodium borohydride for reduction. The mechanisms of oxidation and reduction reactions can vary depending on the specific reaction and the functional groups involved.
5. Rearrangement Reactions:
Rearrangement reactions involve the rearrangement of atoms or groups of atoms within a molecule, resulting in the formation of a different isomer or structure. These reactions typically occur in the presence of acid or heat and involve the migration of a group, such as a hydrogen atom or a carbocation. Rearrangement reactions play a crucial role in organic synthesis, as they allow for the conversion of one functional group into another.
Overall, understanding the mechanisms of these common organic reactions is essential for predicting the behavior of organic compounds and designing synthetic pathways in organic chemistry.
Mastering Organic Synthesis: Strategies and Tips
Organic synthesis is a crucial aspect of organic chemistry, as it involves the creation of new molecules from simpler starting materials. The process of synthesizing organic compounds can be complex and challenging, requiring a deep understanding of reaction mechanisms, functional group transformations, and various techniques. To successfully master organic synthesis, it is essential to adopt effective strategies and employ helpful tips that can enhance your skills in planning and executing synthetic routes.
1. Plan your synthesis carefully: Before starting any organic synthesis, it is crucial to have a clear and detailed plan. Identify the target molecule and break it down into smaller, more manageable fragments. Consider the functional groups present in the target molecule and devise a strategy to introduce or modify these groups in the synthesis.
2. Understand reaction mechanisms: Organic synthesis heavily relies on understanding the mechanisms of various reactions. Familiarize yourself with common reaction types such as nucleophilic substitution, elimination, addition, and oxidation/reduction. This knowledge will help you predict the outcome of reactions and select appropriate starting materials and reagents.
- 3. Build a solid knowledge base: Regularly review and study organic chemistry concepts, including functional group chemistry, stereochemistry, and spectroscopic techniques. This foundation will help you make informed decisions during synthesis planning and troubleshooting.
- 4. Utilize protecting groups: Protecting groups are temporary modifications used to protect specific functional groups during a synthesis. By selectively blocking reactive sites, protecting groups allow for the manipulation of other functional groups without undesired side reactions. Mastering the use of protecting groups can greatly expand your synthetic toolbox.
- 5. Employ retrosynthetic analysis: Retrosynthetic analysis involves working backward from a target molecule to identify potential starting materials. Break down the target molecule into simpler fragments and analyze which reactions can be performed to assemble these fragments. Retrosynthetic analysis helps in devising a stepwise synthetic pathway.
Navigating spectroscopy in organic chemistry lab
Spectroscopy plays a crucial role in organic chemistry lab, allowing researchers to identify and analyze different compounds. One common technique used in spectroscopy is nuclear magnetic resonance (NMR) spectroscopy. NMR spectroscopy provides information about the structure, connectivity, and environment of atoms in a molecule. It is particularly useful in determining the presence and positions of functional groups, which can be critical in organic synthesis.
When performing NMR spectroscopy, it is essential to understand the basic principles of the technique. The NMR spectrum is obtained by subjecting the sample to a strong magnetic field and then applying radiofrequency radiation to cause transitions between energy levels of atomic nuclei. The resulting NMR spectrum consists of peaks corresponding to different nuclei in the molecule, each with its own chemical shift and intensity.
Interpreting an NMR spectrum requires knowledge of the chemical shifts and coupling patterns of different functional groups. For example, the chemical shift of a proton depends on the electronegativity and surrounding atoms. Aromatic protons typically appear at lower chemical shifts, while aliphatic protons appear at higher chemical shifts. Additionally, the number, spacing, and splitting patterns of peaks can provide information about the connectivity and coupling between neighboring protons.
Understanding spectroscopy in organic chemistry lab not only helps in compound identification but also in solving complex structural problems. By carefully analyzing NMR spectra and correlating the information obtained with other spectroscopic techniques like infrared (IR) spectroscopy or mass spectrometry (MS), researchers can confidently determine the structure of unknown compounds. This knowledge is valuable not only in the lab but also in various fields such as drug discovery, environmental analysis, and materials science.
Safety protocols and precautions in the organic chemistry lab
The organic chemistry lab is a highly specialized environment where the handling of chemicals and equipment requires strict safety protocols and precautions. These measures are in place to ensure the well-being of the lab personnel and to prevent accidents or mishaps that could compromise experimental results or cause harm.
Personal protective equipment (PPE) is a crucial aspect of laboratory safety. All individuals working in the organic chemistry lab must wear appropriate PPE, which often includes gloves, goggles, lab coats, and closed-toe shoes. PPE serves as a physical barrier between the chemist and potentially hazardous materials, minimizing the risk of direct contact or exposure.
Chemical handling and storage in the lab must be done with extreme caution. Chemicals should always be labeled appropriately and stored in designated areas to avoid confusion or accidental mixing. Each chemical should have a safety data sheet (SDS) readily available for quick reference on its properties, hazards, and safe handling practices.
Proper ventilation and fume hoods are essential in an organic chemistry lab. Many organic compounds can emit toxic vapors, and it is crucial to have adequate airflow to prevent their accumulation. Fume hoods are designed to draw and exhaust harmful gases and fumes away from the lab personnel, providing an additional layer of protection.
Emergency procedures and equipment should be familiar to all lab members. Knowing the location and proper use of fire extinguishers, eyewash stations, and emergency showers can be vital in the event of accidents or chemical spills. Regular emergency drills and training sessions should be conducted to ensure everyone is prepared for any unexpected situations.
In conclusion

Following safety protocols and precautions is of utmost importance in the organic chemistry lab. By wearing appropriate PPE, handling and storing chemicals responsibly, maintaining proper ventilation, and being knowledgeable about emergency procedures, lab personnel can minimize risks and create a safe working environment for themselves and others.