Energy is a fundamental element of our daily lives, powering our homes, cities, and industries. As the global demand for energy continues to rise, it is crucial to explore and develop new energy concepts and solutions to meet our growing needs. In recent years, there has been a growing interest in renewable energy sources, such as solar and wind power, as well as advancements in energy storage technologies.
One of the key challenges in the energy sector is finding answers to questions related to efficiency and sustainability. How can we optimize the generation and distribution of energy to minimize waste and environmental impact? How can we ensure a stable and reliable energy supply while reducing our dependence on fossil fuels? These are complex questions that require innovative thinking and interdisciplinary approaches.
One promising concept that has gained traction in recent years is the idea of a “smart grid”. A smart grid is an intelligent electricity network that utilizes advanced communications, sensors, and automation to monitor and control the flow of electricity. This concept holds the potential to optimize energy distribution, reduce energy losses, and integrate renewable energy sources more efficiently into the grid.
Another area of focus is energy storage. The intermittent nature of renewable energy sources like solar and wind poses a challenge for their widespread adoption. Energy storage technologies, such as batteries and fuel cells, offer a solution by storing excess energy during periods of high production and releasing it during times of high demand. Developing more efficient and affordable energy storage systems is crucial for a sustainable energy future.
In conclusion, finding answers to our energy needs requires constant innovation and collaboration between scientists, engineers, and policymakers. Exploring new energy concepts, such as smart grids and energy storage, will play a vital role in shaping our energy landscape and ensuring a sustainable future for generations to come.
What is Energy?
Energy is the ability to do work or cause change. It can exist in many different forms, such as mechanical, thermal, chemical, electrical, and nuclear. Energy is constantly being transferred and transformed from one form to another, but it can neither be created nor destroyed according to the law of conservation of energy.
Mechanical energy refers to the energy possessed by an object due to its motion or position. Thermal energy is the energy associated with the motion of particles in a substance and is related to its temperature. Chemical energy is stored in the bonds between atoms and is released during a chemical reaction. Electrical energy is the energy associated with the flow of electric charge. Nuclear energy is released during nuclear reactions, such as the fusion of atoms in the sun.
Energy is essential for all forms of life and plays a crucial role in various processes and activities. It powers our homes, fuels our vehicles, and enables us to perform tasks. Renewable energy sources, such as solar and wind, are becoming increasingly important in light of environmental concerns and the finite nature of fossil fuels. Understanding and harnessing different forms of energy is crucial for sustainable development and addressing the energy challenges of the future.
Definition of Energy
Energy can be defined as the ability or capacity of a system to do work or produce a change. It is a fundamental concept in physics that is used to explain various phenomena and processes in the natural world. Energy can exist in different forms, such as kinetic energy, potential energy, thermal energy, electrical energy, and chemical energy.
Kinetic energy is the energy an object possesses due to its motion. The faster the object moves, the greater its kinetic energy. This energy can be transferred from one object to another and can be used to do work.
Potential energy is the energy stored within an object as a result of its position or condition. This energy can be converted into kinetic energy when the object moves or changes its position.
Thermal energy is the energy associated with the temperature of an object or a system. It is a form of kinetic energy due to the random motion of particles within the object or system.
Electrical energy is the energy associated with the flow of electric charge. It is a form of energy that is commonly used to power electrical devices and systems.
Chemical energy is the energy stored within chemical compounds. It is a form of potential energy that can be released during a chemical reaction, such as combustion.
In summary, energy is a fundamental concept that describes the ability of a system to do work or produce a change. It can exist in various forms and can be converted from one form to another. Understanding energy is crucial for understanding how the natural world works and for developing sustainable energy sources and technologies.
Types of Energy

Energy is a fundamental concept in physics and is often described as the ability to do work or cause change. There are several different types of energy, each with its own characteristics and applications.
1. Kinetic Energy: Kinetic energy is the energy possessed by an object due to its motion. The faster an object moves, the more kinetic energy it has. For example, a moving car has kinetic energy because of its motion.
2. Potential Energy: Potential energy is the energy stored in an object based on its position or condition. There are several types of potential energy, including gravitational potential energy, elastic potential energy, and chemical potential energy. An object at the top of a hill has gravitational potential energy because of its position.
- Gravitational Potential Energy: Gravitational potential energy depends on an object’s height and mass. An object at a higher elevation has more gravitational potential energy. For example, a roller coaster at the peak of a hill has a high gravitational potential energy.
- Elastic Potential Energy: Elastic potential energy is stored in objects that can be stretched or compressed, like a spring. When a spring is stretched or compressed, it stores potential energy. For example, a stretched rubber band has elastic potential energy.
- Chemical Potential Energy: Chemical potential energy is stored in chemical bonds and can be released during a chemical reaction. For example, food contains chemical potential energy that our bodies convert into kinetic energy for movement.
3. Thermal Energy: Thermal energy refers to the internal energy of an object due to the motion of its atoms and molecules. It is associated with temperature and is responsible for the sensation of heat. For example, when you touch a hot object, you feel the transfer of thermal energy.
4. Electrical Energy: Electrical energy is the energy carried by electric charges, such as electrons. It can be generated through various processes, including electromagnetic induction and chemical reactions. Electrical energy powers a wide range of devices, from light bulbs to computers.
5. Light Energy: Light energy, or radiant energy, is a form of electromagnetic radiation that stimulates the sense of sight. It is produced by objects that emit or reflect light, such as the sun, light bulbs, and glowing objects. Light energy is vital for vision and plays a crucial role in many natural processes.
These are just a few examples of the different types of energy. Understanding the various forms of energy is essential for comprehending the world around us and developing sustainable energy sources for the future.
The Laws of Energy

The laws of energy are fundamental principles that govern the behavior and interactions of energy in various systems. These laws are based on observations and experiments conducted by scientists over the years, and they help us understand how energy can be transferred, transformed, and conserved.
1. The Law of Conservation of Energy:
This law states that energy cannot be created or destroyed; it can only be transferred or transformed from one form to another. In other words, the total amount of energy in a closed system remains constant. This principle is often referred to as the first law of thermodynamics. It signifies that energy is a fundamental quantity that is conserved throughout any process or interaction.
2. The Law of Energy Transformation:
This law states that energy can be converted from one form to another. For example, potential energy can be converted into kinetic energy, or electrical energy can be transformed into heat energy. However, during these transformations, some energy is often lost in the form of heat or other less useful forms of energy. This principle highlights the concept of energy efficiency and the importance of minimizing energy losses in various systems.
3. The Law of Energy Transfer:
This law states that energy can be transferred from one object to another through various mechanisms such as heat transfer, work transfer, or radiation. Heat transfer involves the transfer of thermal energy from a source to a recipient, while work transfer occurs when a force acts on an object to move it. Radiation refers to the transmission of energy through electromagnetic waves. Understanding how energy is transferred is crucial for designing efficient energy systems and optimizing energy usage.
4. The Law of Entropy:
This law, also known as the second law of thermodynamics, states that the entropy, or the measure of disorder in a system, tends to increase over time in an isolated system. In simpler terms, it implies that processes occurring in nature tend to move towards a state of higher disorder. This law has important implications for energy conversion processes, as it sets limits on the efficiency of converting energy from one form to another.
In conclusion, the laws of energy provide a framework for understanding the behavior and properties of energy. These laws enable scientists and engineers to develop efficient energy systems, improve energy conservation efforts, and make informed decisions regarding energy usage and management.
The First Law of Thermodynamics

The First Law of Thermodynamics, also known as the law of energy conservation, states that energy cannot be created or destroyed in an isolated system. Instead, the total energy within the system remains constant. This concept is essential in understanding the principles of energy transfer and transformation.
The First Law of Thermodynamics can be expressed mathematically as follows:
Esystem = Q – W
Where Esystem is the internal energy of the system, Q is the heat supplied to the system, and W is the work done by the system.
The equation shows that the change in internal energy of the system is equal to the heat added to the system minus the work done by the system. This means that if energy is added to a system in the form of heat, it can increase the internal energy. Conversely, if work is done by the system, it can decrease the internal energy.
The First Law of Thermodynamics has wide-ranging applications in various fields, including physics, chemistry, and engineering. It is fundamental in understanding how energy is conserved and transformed in different processes, such as in the operation of heat engines, chemical reactions, and electrical circuits. By applying this law, scientists and engineers can analyze and predict the behavior of energy in different systems, leading to advancements in technology and the development of sustainable energy solutions.
The Second Law of Thermodynamics
The Second Law of Thermodynamics is a fundamental principle in physics that deals with the concept of energy and its transformations. It states that the total entropy of an isolated system will always increase over time. Entropy can be understood as a measure of disorder or randomness within a system.
This law can be explained using various concepts and principles, such as heat transfer, energy conservation, and the efficiency of energy conversion processes. It is closely related to concepts like thermal equilibrium, heat engines, and the direction of spontaneous processes.
Key Concepts
- Entropy: The measure of the level of disorder or randomness within a system.
- Isolated System: A system that does not exchange matter or energy with its surroundings.
- Heat Transfer: The transfer of thermal energy from a region of higher temperature to a region of lower temperature.
- Energy Conservation: The principle that energy cannot be created or destroyed, only transformed from one form to another.
- Efficiency: The ratio of useful energy output to the total energy input in a process or system.
- Thermal Equilibrium: The state where two objects or systems are at the same temperature and there is no net heat transfer between them.
- Heat Engines: Devices that convert heat energy into mechanical work, such as steam engines or car engines.
- Spontaneous Processes: Processes that occur naturally without the need for external intervention or energy input.
Understanding the Second Law of Thermodynamics is crucial in many fields, including engineering, chemistry, and environmental science. It provides insights into the limitations and possibilities of energy conversion processes and helps in the design and optimization of various systems and devices.
In conclusion, the Second Law of Thermodynamics states that the total entropy of an isolated system will always increase over time. This law is based on fundamental concepts like entropy, heat transfer, energy conservation, and the efficiency of energy conversion processes. It has wide-ranging applications in different scientific and engineering disciplines.
Energy Conversion

The process of energy conversion involves transforming energy from one form to another. This is a fundamental concept in physics, as energy can exist in different forms such as mechanical, thermal, electrical, and chemical energy. Energy can also be converted from one form to another, depending on the system and the processes involved.
In many practical applications, energy conversion is necessary to make energy usable for specific purposes. For example, in power plants, energy is converted from thermal energy (generated by burning fossil fuels or through nuclear reactions) into electrical energy. This conversion process involves the use of generators and turbines to produce electricity.
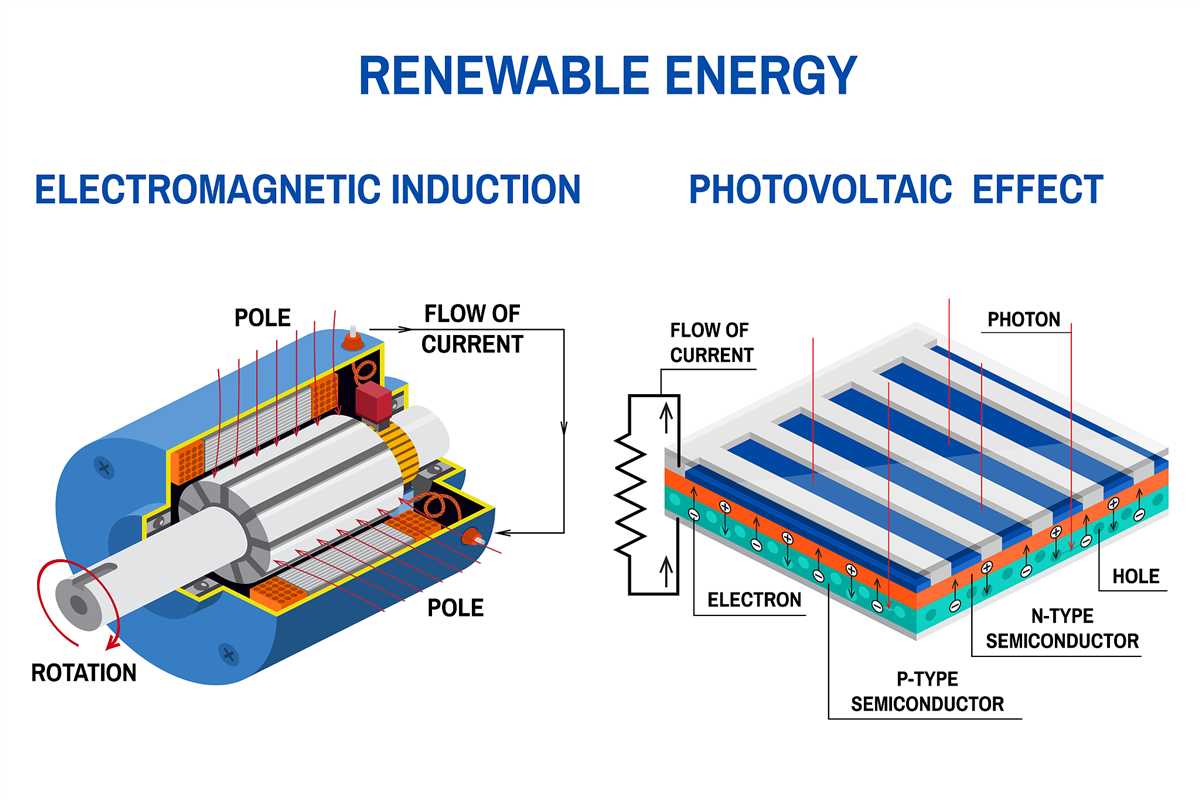
Another common example of energy conversion is in the use of solar panels to generate electricity. Solar panels convert sunlight (radiant energy) into electrical energy through the photovoltaic effect. This process involves the use of semiconductor materials that absorb the photons from sunlight and release electrons, creating an electric current.
Energy conversion is also essential in everyday devices such as cars and batteries. In a car, the chemical energy stored in gasoline is converted into mechanical energy by the engine. In batteries, the chemical energy is converted into electrical energy, which can then be used to power various electronic devices.
Overall, energy conversion plays a vital role in our modern society, enabling the efficient use of different forms of energy for various applications. Understanding the principles of energy conversion is crucial for developing sustainable and renewable energy sources and improving energy efficiency.