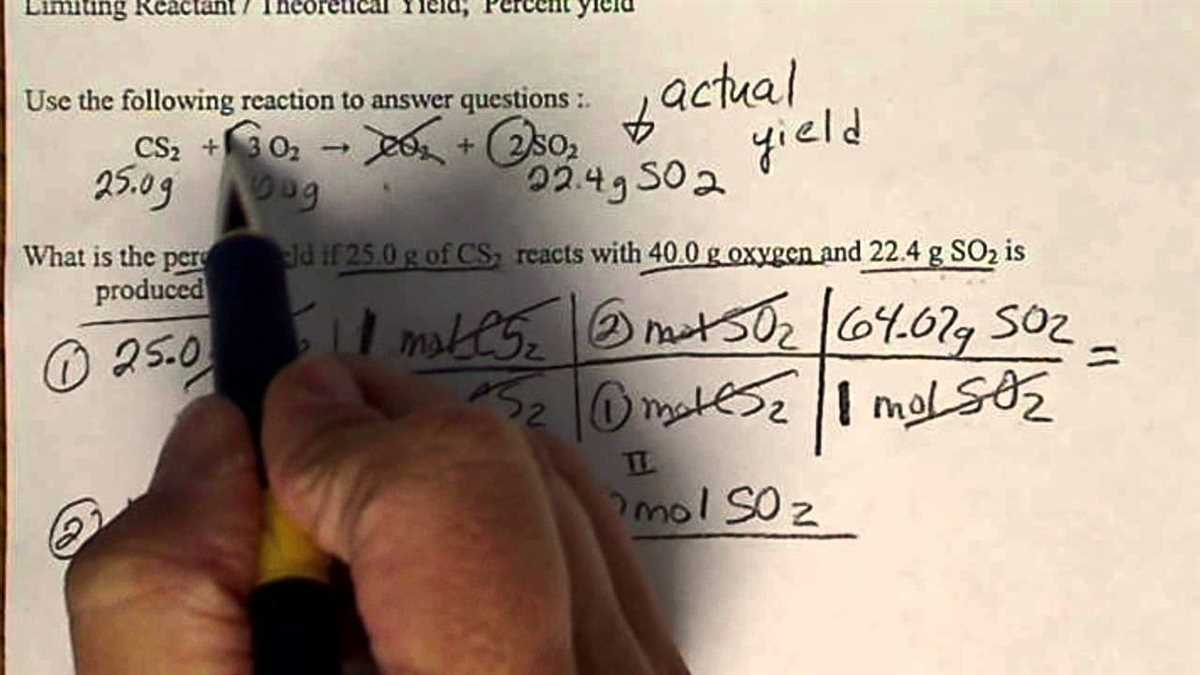
When it comes to understanding chemical reactions, one concept that is crucial to grasp is the idea of limiting reactants. In a chemical reaction, there are often multiple reactants involved, and some reactants may be present in excess while others may be used up completely. The limiting reactant is the one that gets used up first and limits the amount of product that can be formed.
The Gizmo Limiting Reactants is a valuable tool that helps students explore this concept in a hands-on and interactive way. By manipulating the amounts of reactants and observing the changes in the reaction, students can gain a deeper understanding of how limiting reactants affect the outcome of a chemical reaction.
This answer key provides a comprehensive guide to the Gizmo Limiting Reactants, allowing students to check their answers and ensure they are on the right track. It includes step-by-step explanations and solutions to the various problems and activities presented in the Gizmo, helping students develop their problem-solving skills and grasp the key concepts with clarity.
Gizmo Limiting Reactants Answer Key
In chemistry, the concept of limiting reactants is key to understanding chemical reactions. A limiting reactant is the substance that is completely consumed in a reaction, while the excess reactant is left over. The Gizmo Limiting Reactants Answer Key provides students with a comprehensive answer key to the Gizmo activity, which is an interactive simulation that allows students to explore limiting reactants in a hands-on way.
The Gizmo Limiting Reactants Answer Key includes step-by-step instructions on how to use the Gizmo, as well as detailed explanations of the concepts involved. It provides answers to all the questions and problems in the activity, allowing students to check their work and ensure they understand the material. The answer key also provides additional information and examples to help clarify any confusing concepts.
The Gizmo Limiting Reactants Answer Key is a valuable resource for both teachers and students. It allows teachers to quickly and easily assess student understanding and provide feedback. Students can use the answer key to review and reinforce their learning, ensuring they have a solid grasp of the material. By using the Gizmo and its answer key, students can gain a deeper understanding of limiting reactants and how they affect chemical reactions.
What are limiting reactants and why are they important?
In a chemical reaction, the limiting reactant is the substance that limits the extent of the reaction, preventing any further reaction once it is completely consumed. It determines the maximum amount of product that can be formed. The other reactant is called the excess reactant, as it is present in excess and is not completely consumed in the reaction.
Identifying the limiting reactant is crucial in understanding and predicting the yield of a reaction. It allows chemists to determine the theoretical yield, which is the maximum amount of product that can be obtained based on the amount of limiting reactant present. This information is essential for industries to optimize their production processes and minimize waste.
The importance of limiting reactants can be understood through an analogy. Imagine you are baking cookies, and you have a limited amount of flour, sugar, and butter. Each ingredient plays a specific role, but if you run out of flour before using up all the other ingredients, you won’t be able to make any more cookies. The flour in this case is the limiting reactant, and it determines the number of cookies you can bake.
Limiting reactants are also important in understanding reaction stoichiometry, which is the study of the quantitative relationship between reactants and products in a chemical reaction. By knowing the identity and the amount of limiting reactant, scientists can calculate the amount of product that will be obtained, as well as determine the amount of excess reactant remaining after the reaction.
In summary, limiting reactants are fundamental in chemistry as they determine the maximum amount of product that can be formed in a chemical reaction. They help scientists predict the yield of reactions and optimize production processes. Understanding and identifying limiting reactants is essential for industries and researchers alike.
How to Identify the Limiting Reactant in a Chemical Reaction
In a chemical reaction, the limiting reactant is the substance that will be completely consumed when the reaction reaches completion. It determines how much product can be formed and is an important concept in stoichiometry. Identifying the limiting reactant is crucial in determining the optimal conditions for a reaction and maximizing yield.
To identify the limiting reactant, you need to compare the ratios between the moles or masses of the reactants and the stoichiometric coefficient in the balanced chemical equation. The reactant with the smallest ratio is the limiting reactant.
Let’s take an example of the reaction between hydrogen (H2) and oxygen (O2) to form water (H2O) using the balanced chemical equation:
2H2 + O2 → 2H2O
In this equation, the stoichiometric coefficients indicate the ratio of moles required for the reaction. The ratio of H2 to O2 is 2:1, which means that 2 moles of hydrogen react with 1 mole of oxygen to produce 2 moles of water.
To determine the limiting reactant, you need to compare the number of moles or masses of the reactants. Let’s say you have 4 moles of H2 and 3 moles of O2. Using the stoichiometric ratio, you can calculate that 4 moles of H2 will require 2 moles of O2, and 3 moles of O2 will require 6 moles of H2. Here, you can see that there is an excess of O2 (6 moles required) compared to H2 (4 moles available). Therefore, H2 is the limiting reactant.
Once you have identified the limiting reactant, you can calculate the maximum amount of product that can be formed using the stoichiometric ratios. In this case, since the ratio of H2 to H2O is 2:2, and you have 4 moles of H2, the maximum yield of water will be 4 moles.
It is important to note that the limiting reactant can also be identified by comparing the masses of the reactants using their molar masses. The reactant with the smallest mass is the limiting reactant. This method is particularly useful when dealing with reactions in the laboratory, where the reactants are often measured by mass rather than moles.
Steps to complete a limiting reactant calculation
Completing a limiting reactant calculation involves several steps to determine which reactant will be completely consumed in a chemical reaction. This calculation is important because it allows us to determine the maximum amount of product that can be formed.
Here are the steps to complete a limiting reactant calculation:
- Write out the balanced chemical equation: Begin by writing out the balanced chemical equation for the reaction. Make sure the equation is balanced so that the number of atoms of each element is the same on both sides.
- Determine the moles of each reactant: Convert the given masses of each reactant to moles using the molar mass of each substance. This can be done by dividing the mass of each reactant by its molar mass.
- Calculate the moles of product: Use the stoichiometry of the balanced equation to determine the number of moles of the product that can be formed from each reactant. This can be done by multiplying the moles of each reactant by the appropriate mole ratio.
- Identify the limiting reactant: Compare the moles of product that can be formed from each reactant. The reactant that produces the smaller amount of product is the limiting reactant. This is because it will be completely consumed in the reaction, limiting the amount of product that can be formed.
- Calculate the amount of excess reactant: Once the limiting reactant is identified, calculate the amount of excess reactant that remains after the reaction is complete. This can be done by subtracting the moles of limiting reactant used from the initial moles of reactant.
- Determine the amount of product formed: Finally, use the moles of limiting reactant to calculate the amount of product that can be formed. This can be done by multiplying the moles of limiting reactant by the mole ratio of the product to the limiting reactant.
By following these steps, you can accurately determine the limiting reactant in a chemical reaction and calculate the maximum amount of product that can be formed.
Common misconceptions and tips for solving limiting reactant problems
Limiting reactant problems in chemistry can be challenging and often lead to common misconceptions. One common misconception is that the reactant with the smallest given mass is always the limiting reactant. However, this is not always the case. It is important to remember that the actual amount of each reactant must be considered, not just the given mass.
Another misconception is that the limiting reactant is always completely consumed in the reaction. While it is true that the limiting reactant is completely consumed, this does not mean that the other reactant is completely used up as well. The excess reactant may still have some remaining after the reaction is complete.
When solving limiting reactant problems, it is helpful to follow a step-by-step approach. First, write out the balanced chemical equation for the reaction. Next, calculate the molar masses of the reactants. Then, convert the given quantities of each reactant to moles. After that, determine the stoichiometric ratio between the reactants by examining the coefficients in the balanced equation. Use this ratio to calculate the amount of product that can be formed from each reactant. Finally, compare the results to identify the limiting reactant.
To avoid mistakes, it is important to carefully track units throughout the calculations and pay attention to significant figures. It is also helpful to practice with different examples of limiting reactant problems to become familiar with the process and gain confidence in solving them accurately.
- Remember: The limiting reactant is not always the reactant with the smallest given mass.
- Be aware: The limiting reactant is completely consumed, but the excess reactant may still have some remaining.
- Follow a step-by-step approach: Write the balanced equation, calculate molar masses, convert to moles, determine stoichiometric ratio, calculate product amounts, and compare results.
- Track units and significant figures: Pay attention to units and significant figures throughout the calculations.
- Practice: Practice with different examples to gain familiarity and confidence in solving limiting reactant problems accurately.
Examples of Limiting Reactant Calculations
In chemistry, limiting reactants refer to the reactants that are completely consumed in a chemical reaction, thereby limiting the extent of the reaction. To determine the limiting reactant, one must compare the molar ratios of the reactants and the balanced chemical equation.
Example 1:
Consider the reaction between hydrogen gas (H2) and oxygen gas (O2) to form water (H2O). The balanced chemical equation is:
2H2 + O2 → 2H2O
If 4 moles of hydrogen gas and 2 moles of oxygen gas are available for the reaction, we can determine the limiting reactant as follows:
- Convert moles to the same units for comparison:
- 4 moles of H2 → 4 moles H2
- 2 moles of O2 → 2 moles O2
- Compare the molar ratios of the reactants:
- 4 moles H2 : 2 moles O2 → 2:1
- Determine the limiting reactant:
- The ratio of H2 to O2 is 2:1, so for every 2 moles of H2, we need 1 mole of O2. Since we have 4 moles of H2 and only 2 moles of O2, O2 is the limiting reactant.
Example 2:
Consider the reaction between methane (CH4) and oxygen gas (O2) to form carbon dioxide (CO2) and water (H2O). The balanced chemical equation is:
CH4 + 2O2 → CO2 + 2H2O
If 2 moles of methane and 6 moles of oxygen gas are available for the reaction, we can determine the limiting reactant as follows:
- Convert moles to the same units for comparison:
- 2 moles of CH4 → 2 moles CH4
- 6 moles of O2 → 6 moles O2
- Compare the molar ratios of the reactants:
- 2 moles CH4 : 6 moles O2 → 1:3
- Determine the limiting reactant:
- The ratio of CH4 to O2 is 1:3, so for every 1 mole of CH4, we need 3 moles of O2. Since we have 2 moles of CH4 and 6 moles of O2, CH4 is the limiting reactant.
Understanding and identifying the limiting reactant is crucial in predicting the amount of product that can be formed in a chemical reaction. By calculating the limiting reactant, chemists can optimize reactions to maximize the yield of desired products.