
In the study of chemistry, understanding the nature of elements and compounds is crucial. Elements are pure substances composed of only one type of atom. They cannot be broken down into simpler substances. On the other hand, compounds are substances composed of two or more elements chemically combined in definite proportions. They can be broken down into simpler substances through chemical reactions.
When studying elements and compounds, it is important to understand their properties and how they interact with each other. This understanding allows scientists to predict and explain the behavior of different substances. To aid in this understanding, worksheets are often used to provide practice and reinforce knowledge.
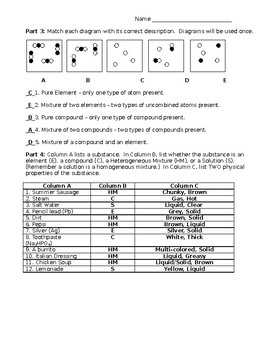
2.3 Elements and Compounds Worksheet Answers is a resource that provides the answers to various questions and problems related to elements and compounds. This worksheet covers topics such as identifying elements and compounds, naming compounds, and balancing chemical equations. By using this resource, students can check their understanding and ensure they are on the right track.
Overall, the study of elements and compounds is fundamental to the field of chemistry. It provides the foundation for understanding the composition, structure, and behavior of matter. Utilizing resources like the 2.3 Elements and Compounds Worksheet Answers can help students enhance their learning and solidify their understanding of this important topic.
Understanding Elements and Compounds: 2.3 Worksheet Answers
Introduction
In the study of chemistry, it is essential to understand the concepts of elements and compounds. Elements are the simplest form of matter that cannot be broken down into simpler substances through chemical means. Compounds, on the other hand, are substances that consist of two or more different elements chemically bonded together. In this worksheet, we will explore various questions related to elements and compounds to enhance our understanding of these fundamental concepts.
Worksheet Answers
1. What is an element?
An element is a pure substance that is composed of only one type of atom. It is the simplest form of matter and cannot be broken down into simpler substances through chemical means.
2. What is a compound?
A compound is a substance that consists of two or more different elements chemically bonded together. The elements in a compound are present in fixed proportions and can be separated into their constituent elements through chemical reactions.
3. How are elements and compounds different?
- Elements consist of only one type of atom, while compounds consist of two or more different elements.
- Elements cannot be broken down into simpler substances, while compounds can be separated into their constituent elements.
- Elements have unique chemical symbols, while compounds have chemical formulas.
4. Give an example of an element and a compound.
- An example of an element is oxygen (O). It consists of only one type of atom.
- An example of a compound is water (H2O). It consists of two different elements, hydrogen (H) and oxygen (O), chemically bonded together.
5. How can you determine the number of elements in a compound?
The number of elements in a compound can be determined by analyzing the formula of the compound. Each element in the formula is represented by its chemical symbol. By counting the different chemical symbols, we can determine the number of elements present in the compound.
6. Can compounds exist in different forms?
Yes, compounds can exist in different forms known as allotropes. Allotropes are different structural arrangements of the same elements in a compound. For example, carbon can exist as diamond, graphite, or fullerenes, which are all different forms of the compound composed of carbon atoms.
Conclusion
Understanding elements and compounds is crucial in the study of chemistry. Elements are the building blocks of matter, while compounds are formed by the chemical bonding of two or more elements. By answering the questions in this worksheet, we have gained a deeper understanding of these fundamental concepts and their distinguishing characteristics.
The Difference Between Elements and Compounds
Elements and compounds are two fundamental types of substances found in nature. They are both composed of atoms, but there are significant differences between the two.
Elements are pure substances that cannot be broken down into simpler substances by any chemical means. They are the building blocks of all matter and are represented by symbols on the periodic table. Each element has its own unique set of properties, such as atomic number, atomic mass, and chemical reactivity. Examples of elements include hydrogen, oxygen, carbon, and gold.
Compounds, on the other hand, are substances that are composed of two or more elements chemically bonded together. Unlike elements, compounds can be broken down into their constituent elements through chemical reactions. Compounds have unique properties that are different from the elements that make them up. For example, water is a compound made up of hydrogen and oxygen, and it has different properties than its constituent elements.
In summary, the main difference between elements and compounds lies in their composition and ability to be broken down. Elements are pure substances that cannot be broken down, while compounds are composed of two or more elements chemically combined together. Understanding the difference between elements and compounds is crucial in understanding the nature of matter and how substances interact with each other.
Characteristics of Elements
Elements are the building blocks of matter. They cannot be broken down into simpler substances by chemical reactions. Each element is unique and has its own set of characteristics that distinguish it from other elements.
Atomic Number: The atomic number of an element is the number of protons in the nucleus of its atom. It uniquely identifies an element and determines its position on the periodic table. The atomic number also determines the number of electrons in a neutral atom of the element.
Atomic Mass: The atomic mass of an element is the average mass of all the isotopes of that element found in nature. It is expressed in atomic mass units (amu) or grams per mole (g/mol). The atomic mass is determined by the number of protons and neutrons in the nucleus of the atom.
Physical State: Elements can exist in different physical states, namely solid, liquid, or gas, depending on the temperature and pressure. For example, oxygen is a gas at room temperature, while gold is a solid.
Chemical Properties: Each element has its own characteristic set of chemical properties, which determine its behavior in chemical reactions. These properties include reactivity, oxidizing or reducing power, and ability to form compounds with other elements.
Symbol: Elements are represented by symbols, usually derived from their names. These symbols are used to identify elements in chemical equations and formulas. For example, the symbol for oxygen is O, and the symbol for carbon is C.
Period and Group: Elements are organized in the periodic table based on their atomic number. The periodic table is divided into periods (horizontal rows) and groups/families (vertical columns). Elements in the same group have similar properties, while elements in the same period have the same number of electron shells.
In conclusion, elements have unique characteristics such as atomic number, atomic mass, physical state, chemical properties, symbol, and position in the periodic table. These characteristics help identify and differentiate elements from one another.
Properties of Compounds
A compound is a substance composed of two or more elements chemically bonded together. The properties of compounds are different from the properties of the individual elements that make up the compound. These properties are a result of the chemical bonds and interactions between the atoms in the compound.
Chemical Composition: Compounds have a fixed chemical composition, meaning they always contain the same ratio of elements. For example, water (H2O) always has two hydrogen atoms for every one oxygen atom. This fixed composition gives compounds their unique properties.
Melting and Boiling Points: Compounds usually have higher melting and boiling points compared to the elements they are made of. This is because the chemical bonds in compounds are stronger than the forces between individual atoms.
Electrical Conductivity: Compounds can be either conductors or insulators of electricity. Ionic compounds, which are composed of positively and negatively charged ions, can conduct electricity when dissolved in water or melted. However, covalent compounds, which share electrons between atoms, do not conduct electricity.
Solubility: Compounds have different solubilities in different solvents. Some compounds dissolve easily in water, while others are insoluble. The solubility of a compound depends on the nature of its chemical bonds and the polarity of the solvent.
Reactivity: Compounds often have different chemical reactivity compared to the elements they are made of. This is because the chemical bonds in compounds are different from the bonds between individual atoms. Compounds can react with other substances to form new compounds through chemical reactions.
Physical Properties: Compounds have a wide range of physical properties, including color, density, odor, and texture. These properties can vary depending on the arrangement of atoms and the types of chemical bonds present in the compound.
Overall, compounds exhibit unique properties that are a result of their chemical composition and the interactions between their constituent elements. Understanding these properties is important in various fields of science, such as chemistry and materials science.
Identifying Elements and Compounds

In chemistry, understanding the difference between elements and compounds is crucial. Elements are the building blocks of matter, and they cannot be broken down into simpler substances through chemical reactions. They are represented by symbols on the periodic table, such as carbon (C) or oxygen (O). Compounds, on the other hand, are formed when two or more elements chemically combine in a fixed ratio.
One way to identify elements is by their unique physical and chemical properties. Each element has its own set of characteristics, including melting and boiling points, density, and reactivity. These properties can be used to distinguish between different elements and determine their presence in a substance. For example, hydrogen (H) is highly flammable, while gold (Au) has a high melting point and is resistant to corrosion.
To identify compounds, one must analyze the composition of a substance and determine the elements present. This can be done through techniques such as spectroscopy or chemical analysis. In spectroscopy, the substance is subjected to different wavelengths of light, and the resulting spectrum can reveal the elements and compounds present. Chemical analysis involves various tests and experiments to identify the elements in a compound.
In conclusion, identifying elements and compounds is essential in chemistry. By understanding the unique properties of elements and analyzing the composition of substances, scientists can determine the presence and nature of different substances. This knowledge is crucial for various applications, from understanding the composition of materials to developing new drugs and technologies.
The Periodic Table as a Tool
The Periodic Table is a valuable tool for scientists and chemists alike. It is a system to organize and classify elements based on their chemical properties and atomic structure.
One of the key features of the Periodic Table is its ability to provide information about the various properties of elements. Each element is represented by a symbol and is placed in a specific location on the table. The arrangement of the elements is based on their atomic number, which refers to the number of protons in an atom’s nucleus. This arrangement allows scientists to easily identify and compare elements based on their properties.
The Periodic Table also displays the elements’ atomic mass. This is the average mass of an atom of that element, taking into account the different isotopes and their abundance. It is represented by a decimal number below the element’s symbol. This information is crucial for chemists in determining the stoichiometry of chemical reactions.
Furthermore, the Periodic Table helps scientists understand the periodic trends and patterns that exist among the elements. For example, elements in the same group (vertical column) often have similar chemical properties, while elements in the same period (horizontal row) exhibit similar energy levels. These trends can be used to make predictions about the behavior and reactivity of elements.
In conclusion, the Periodic Table is an indispensable tool in the field of chemistry. It provides a visual representation of elements and their properties, allowing scientists to study, classify, and predict the behavior of various substances. Its organization and arrangement have revolutionized our understanding of the building blocks of matter.
Practical Applications of Elements and Compounds
The study of elements and compounds is not only important for understanding the basic building blocks of matter, but it also has numerous practical applications in everyday life. Here are some examples:
